Abstract
Objective
The objective is to examine the effect of tumor necrosis factor α (TNFα) on apoptosis and proliferation of rheumatoid arthritis synovial fibroblasts (RASFs) and to elucidate the regulatory roles of Kruppel-like factor 4 (KLF4) in TNFα-induced RASF apoptosis.
Methods
Changes in cell proliferation were measured using an 3-(4,5)-dimethylthiahiazo (-z-y1)-3,5-di- phenytetrazoliumromide (MTT) assay, and changes in cell apoptosis were detected by flow cytometry and Hoechst 33258 staining. Changes in the apoptosis-related protein caspase-3 and the apoptosis-related genes bcl-2/bax were measured by western blot and real-time PCR, respectively
Results
TNFα stimulation increased cell proliferation (p < 0.05), decreased cell apoptosis (p < 0.05), declined caspase-3 expression (p < 0.05), and upregulated bcl-2/bax level (p < 0.05) in RASFs. KLF4 gene silencing decreased cell proliferation (p < 0.05), increased cell apoptosis (p < 0.05), upregulated caspase-3 expression (p < 0.05), and downregulated bcl-2/bax level (p < 0.05) induced by TNFα in RASFs.
Conclusions
TNFα caused a decrease in RASF apoptosis, and KLF4 promoted resistance to TNFα-induced apoptosis and cell proliferation.
Introduction
Rheumatoid arthritis (RA) is a common autoimmune disorder characterized by synovial hyperplasia and inflammation resulting in joint cartilage and bone damage, ultimately leading to joint deformity and loss of function. Rheumatoid arthritis synovial fibroblasts (RASFs) can remain activated for an extended duration, and their phenotypic changes can increase the resistance of RASFs to apoptosis.1,2 Insufficient apoptosis promotes synovial hyperplasia, which prolongs RASF survival, further fueling synovial hyperplasia and cartilage damage.3–5 Tumor necrosis factor α (TNFα), a pro-inflammatory cytokine implicated in the pathogenesis and progression of RA, has been recognized as an important factor in inducing the resistance to apoptosis in RASFs. Therefore, decreased RASF apoptosis or resistance of RASF to apoptosis plays an important role in the manifestation of RA synovial hyperplasia.6,7
Kruppel-like factor 4 (KLF4) is a member of the Sp1/KLF zinc finger transcription factor family. KLF4 is involved in a variety of biological functions, including normal cell proliferation and differentiation, embryonic development, tumor cell cycle regulation, proliferation, apoptosis, differentiation, and migration.8–10 It has previously been reported that KLF4 is highly expressed in RA synovial tissues, playing an important regulatory role in RA synovial inflammation.11–13 However, the role of KLF4 in RASF apoptosis and RA synovial hyperplasia remains unclear. In the present study, the human synovial fibroblast cell line MH7A was used to examine the effects of TNFα on the proliferation, apoptosis, and apoptosis-related gene expression in RASFs. KLF4 gene silencing was conducted to elucidate the regulatory role of KLF4 in TNFα-induced RASF apoptosis.
Materials and methods
RASF culture
The human RASF cell line MH7A (Jennio, Guangxhou, China) was thawed and cultured in fresh Dulbecco’s modified Eagle’s medium (DMEM) (Life Technologies, Carlsbad, CA, USA) supplemented with 10% fetal bovine serum (FBS) (Life Technologies) which was replaced every 3 days. Upon reaching 80% confluency, the adherent cells were digested with 0.25% trypsin, split 1:2 into a fresh flask, and then cultured in DMEM +10% FBS.
KLF4 small interfering RNA transfection
Three pairs of KLF4 small interfering RNA (siRNA): klf4-homo-1286 (GGUCAUCAGCGUCAGCAAATT), klf4-homo-638 (GGAGAAGACACUGCGUCAATT), klf4-homo-1831 (CCACCCACACUUGUGAUUATT), and a scrambled control siRNA (scRNA) (sense oligonucleotide: 5’-UUC UCC GAA CGU GUC ACG UTT-3’) were designed and synthesized commercially by Genepharma Inc (Shanghai, China). KLF4 siRNA or control siRNA was transfected into MH7A cells using a Lipofectamine RNAiMax kit (Invitrogen). Cells were collected 48 h post-transfection and then the efficacy of gene silencing was determined by quantitative polymerase chain reaction (qPCR).
Detection of cell proliferation by MTT assay
Cells were seeded into the wells of a multiwell plate and treated with 20 μL MTT solution (5 mg/mL). After 4 h of culture, the culture medium was discarded and 150 μL DMSO added to each well. The plate was shaken for 10 min in the dark to allow the crystals to fully dissolve, and the absorbance (OD value) measured at 490 nm. The metabolism of cells was recorded relative to that of the normal control group, defined as 100%. Cell growth in the treated group (%) = (OD of treated group/OD of untreated group) ∗ 100%.
Detection of cell apoptosis by flow cytometry
Treated cells were digested by trypsin and then prepared as a suspension of individual cells. Apoptosis was measured using an annexin V-FITC cell apoptosis detection kit (Thermo scientific, Waltham, MA, USA), in which cells were stained with an annexin V antibody able to specifically bind phospholipid serine in the cell membrane. The 10× binding buffer was diluted to 1× using distilled water then placed on ice. Cells were washed twice in cold phosphate-buffered saline (PBS) (800–1000 r/min for 5 min), resuspended in 490 μL cold binding buffer (105–106 cells/mL), and then stained with 5 μL annexin V-FTIC and 5 μL propidium iodide. Cells were gently shaken and incubated for 10 min on ice in the dark. Cell apoptosis was measured by flow cytometry.
Change in cell apoptosis measured by Hoechst 33258 staining
The culture medium was removed from treated cells which were fixed in 0.5 mL fluorescent fixation solution for 15 min. The cells were washed 3 times in PBS (3 min each wash), stained with 0.5 mL Hoechst 33258 solution (Beyotime, Nantong, China) for 5 min, and then washed twice in PBS (3 min each wash). Fluorescence quenching and mounting solutions were added to a glass slide, onto which a cover slip was placed face down. Cell apoptosis was observed using a fluorescence microscope.
Western blot analysis
Treated cells were collected and lysed in radioimmune precipitation assay lysis buffer (Beyotime) for 30 min. Total protein was extracted and then quantified using a bicinchoninic acid assay (Beyotime). Equal quantities (20 μg) of protein sample were boiled, separated using 12% SDS-polyacrylamide gel electrophoresis (SDS-PAGE), and then transferred onto a polyvinylidene fluoride membrane (Millipore, MA, USA). The membrane was blocked with 5% skimmed milk for 2 h, incubated with primary antibody (anti-GAPDH monoclonal, 1:3000, Santa Cruz; rabbit anti-caspase-3 polyclonal, 1:1000, CST; rabbit anti-bax polyclonal, 1:1000, CST or rabbit anti-bcl-2 polyclonal. 1:1000, CST) overnight at 4°C, rinsed for 30 min, incubated with peroxidase-labeled secondary antibody for 1 h, stained with enhanced chemiluminescence (Millipore), and then visualized using a chemiluminescence imaging system. Grayscale values of the protein bands were calculated using Image J software.
Real-time PCR
Primer sequences of genes used in this study.

Statistical analysis
Data were statistically analyzed using GraphPad Prism 5 and expressed as mean ± standard deviation. Comparisons between two groups were analyzed using a t test, while one-way analysis of variance (ANOVA) was used to compare multiple groups.
p < 0.05 were considered statistically significant.
Results
Effects of TNFα on RASF proliferation and apoptosis
The MTT results indicated that RASF proliferation increased following TNFα stimulation, being most significant after 4 h and 12 h (p < 0.05) (Figure 1(a)). Fluorescence staining and flow cytometry indicated that RASF apoptosis declined after TNFα stimulation, being most significantly inhibited after 24 h (p < 0.05) (Figure 1(b) and (c)). Effect of TNFα on the proliferation and apoptosis in RASFs. The human RASFs were stimulated with 10 ng/mL TNFα for the indicated times. The cell proliferation was detected by MTT method (a), and the cell apoptosis was determined by Hoechst 33258 staining method (b) (×100) and flow cytometry (c). n = 3, ∗p < 0.05 vs 0 h group. TNFα: tumor necrosis factor α; RASFs: rheumatoid arthritis synovial fibroblasts; MTT: 3-(4,5)-dimethylthiahiazo (-z-y1)-3,5-di- phenytetrazoliumromide.
Effect of TNFα on apoptosis-related gene and protein expression in RASFs
Western blot analysis indicated that TNFα stimulation significantly downregulated caspase-3 protein expression in RASFs after 12 h (p < 0.05) but induced no change in the bcl2/bax ratio (Figure 2(a)). Quantitative PCR demonstrated that the bcl-2/bax ratio increased significantly, peaking 24 h after TNFα stimulation (p < 0.05) (Figure 2(b)). Effect of TNFα on the caspase-3 and bcl-2/bax expression in RASFs. The human RASFs were stimulated with 10 ng/mL TNFα for the indicated times. The expression of caspase-3 and bcl-2/bax were determined by western blotting (a) and real-time PCR (b). n = 3, ∗p < 0.05 vs 0 h group. TNFα: tumor necrosis factor α; RASFs: rheumatoid arthritis synovial fibroblasts; PCR: polymerase chain reaction.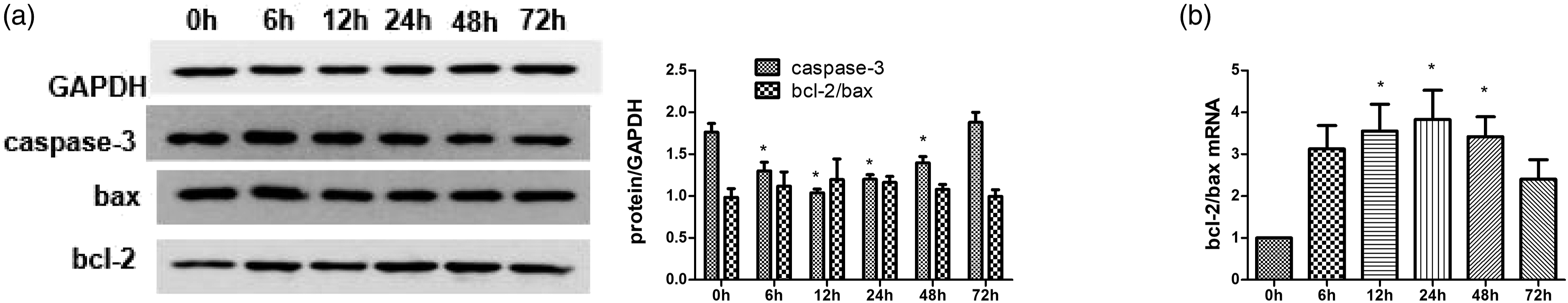
KLF4 siRNA sequence and concentration selection
The PCR results demonstrated that KLF4 mRNA was markedly downregulated in RASFs after transfection with klf4-homo-1286 (p < 0.05). The effect was most significant at a concentration of 20 nm. There was also a decreasing trend in KLF4 mRNA expression following transfection with klf4-homo-638 and klf4-homo-1831, although this change was not statistically significant. Therefore, the klf4-homo-1286 sequence was used for subsequent gene silencing experiments (Figure 3). KLF4 silencing was determined by quantitative PCR. The human RASFs were transiently transfected with three pairs of KLF4 siRNA (klf4-homo-638, 1286, 1831) or a scrambled control siRNA (scRNA) for 48 h, and KLF4 silencing was determined by real-time PCR. n = 3, ∗p < 0.05 vs the untreated control (Ctrl). KLF4: Kruppel-like factor 4; RASFs: rheumatoid arthritis synovial fibroblasts; PCR: polymerase chain reaction.
Effect of KLF4 silencing on RASF proliferation and apoptosis
The MTT assay demonstrated that TNFα-induced cell proliferation decreased significantly following transfection with KLF4 siRNA (p < 0.05) but not scRNA (Figure 4(a)). Fluorescence staining revealed that transfection with KLF4 siRNA, but not scRNA, promoted TNFα-induced cell apoptosis (Figure 4(b)). Likewise, flow cytometric analysis demonstrated that TNFα-induced cell apoptosis increased significantly after KLF4 siRNA transfection (p < 0.05) but did not change significantly following scRNA transfection (Figure 4(c)). Effect of KLF4 silencing on proliferation and apoptosis in RASFs induced TNFα. The human RASFs were transiently transfected with KLF4 siRNA or scrambled siRNA (scRNA) for 48 h and then incubated with 10 ng/mL TNFα for 2 h in the cell proliferation analysis and for 16 h in the cell apoptosis analysis. The cell proliferation was detected by MTT method (a), and the cell apoptosis was determined by Hoechst 33258 staining method (b) (×100) and flow cytometry (c). n = 3, ∗p < 0.05. KLF4: Kruppel-like factor 4; RASFs: rheumatoid arthritis synovial fibroblasts; TNFα: tumor necrosis factor α; MTT: 3-(4,5)-dimethylthiahiazo (-z-y1)-3,5-di- phenytetrazoliumromide.
Effect KLF4 silencing on apoptosis-related gene and protein expression in RASFs
Western blot analysis demonstrated that KLF4 siRNA transfection significantly increased TNFα-induced capase-3 protein expression and decreased bcl-2/bax mRNA expression (p < 0.05), whereas scRNA transfection had no significant effect on TNFα-induced capase-3 or bcl-2/bax expression (Figure 5(a)). Similarly, qPCR indicated that KLF4 siRNA transfection markedly decreased TNFα-induced bcl-2/bax mRNA expression (p < 0.05), while scRNA transfection had no significant impact on TNFα-induced bcl-2/bax expression (Figure 5(b)). Effect of KLF4 silencing on the caspase-3 and bcl-2/bax expression in RASFs. The human RASFs were transiently transfected with KLF4 siRNA or scrambled siRNA (scRNA) for 48 h and then incubated with 10 ng/mL TNFα for 2 h in the mRNA analysis and for 16 h in the protein analysis. Caspase-3 and bcl-2/bax mRNA and protein expression were determined by western blotting (a) and real time-PCR (b). n = 3, ∗p < 0.05. KLF4: Kruppel-like factor 4; RASFs: rheumatoid arthritis synovial fibroblasts; PCR: polymerase chain reaction.
Discussion
Although the pathogenesis of RA remains incompletely understood, many researchers believe that aberrant RASF apoptosis has an important role in RA synovial hyperplasia, and joint cartilage erosion and damage.1,3 RASF apoptosis has been reported to be lower in RA patients than in normal controls and osteoarthritis patients, a result of antiapoptotic protein expression, such as that of bcl-2, mcl-1, and flip in RA synovial tissues.2,14 In addition, the levels of antiapoptotic gene expression were positively correlated with the thickness of the RA synovial lining, demonstrating that insufficient RASF apoptosis or resistance to apoptosis is a key mechanism of RA synovial hyperplasia. 5 TNFα is a member of the TNF family and an important cytokine involved in a variety of acute and chronic inflammatory processes. TNFα is highly expressed in the synovial tissues of RA patients and plays an important role in RA pathogenesis. TNFα can upregulate bcl-2, mcl-1, flip, and sumo-1 gene expression, which inhibits mitochondria-mediated or death receptor–mediated RASF apoptosis. 3
The present study found that RASFs displayed low proliferation but high apoptosis in serum-free culture medium. However, TNFα stimulation increased RASF proliferation, decreased RASF apoptosis, and activated and upregulated caspase-3 expression, indicating that resistance to TNFα-induced apoptosis may be a key mechanism in RASF proliferation. Although both bax (proapoptotic) and bcl-2 (antiapoptotic) gene expressions in RASFs were elevated after TNF-α stimulation, we observed that the bcl-2/bax mRNA ratio increased significantly, demonstrating that resistance to TNFα induces RASF apoptosis by altering the bcl-2/bax balance. In this study, we failed to observe a steady increase in proliferation measured by the MTT method. We speculate that it may be due to the experimental error caused by the small number of repeated experiments.
KLF4 is a transcription factor with multiple biological functions and is involved in the regulation of normal and tumor cell proliferation and apoptosis.8,10,15 Recent studies have found that KLF4 expression is significantly upregulated in the synovial tissues of RA patients and from ex vivo animal models of RA, 11 and that TNFα can induce KLF4 upregulation in RASFs cultured in vitro. 13 However, whether KLF4 is involved in the regulation of RASF apoptosis while undergoing inflammatory cytokine stimulation has not been reported. In order to elucidate the role of KLF4 in TNFα-induced RASF proliferation and apoptosis, we transfected RASFs with KLF4 siRNA to examine the effect of KLF4 silencing on RASF cell proliferation and apoptosis. The results demonstrated that KLF4 silencing can decrease TNFα-induced cell proliferation and increase cell apoptosis in RASFs cultured in vitro. Further analysis revealed that KLF4 silencing reduced TNFα-induced capase-3 activation and expression in addition to the bcl-2/bax ratio. Together, these findings suggest that KLF4 can promote resistance to TNFα-induced RASF apoptosis and proliferation, and this may be associated with the increased bcl-2/bax gene expression ratio.
As a nuclear transcription factor, KLF4 mediates its biological function by regulating the transcription of downstream target genes. KLF4 can bind to KLF4-binding elements in the promoter regions of target genes and thereby regulates target gene transcription and expression.10,16 However, whether promoter binding allows KLF4 to regulate apoptosis-related gene transcription and RASF apoptosis requires additional investigation.
Because it was difficult for us to collect the synovial tissue of patients with active RA, we employed the immortalized RASF cell line MH7A in this study. However, the cell line MH7A, is not entirely consistent in the characteristics and functions as primary RASFs. In addition, we are unable simply to extrapolate our in vitro data to in vivo experiments of animal models of RA and establish their relevance to RASF proliferation and RA synovial hyperplasia. Further in vivo studies are required to elucidate the roles of KLF4 in RASF proliferation and apoptosis.
Conclusions
TNFα caused a decrease in RASF apoptosis, and KLF4 promoted resistance to TNFα-induced apoptosis and cell proliferation. This study indicates that KLF4 may be a key transcription factor in synovial hyperplasia in RA, and targeting of KLF4 in RASFs might be a novel strategy for RA.
Footnotes
Author contributions
NC performed the experiment and drafted the manuscript; WW, ZC, LK, WJ, and ZS performed the experiment; MX designed the study and the data; and LX conceived of study and drafted the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Joint Funds of Zhejiang Provincial Natural Science Foundation of China under Grant (LTY21H100001), Zhejiang Provincial Natural Science Foundation of China under Grant (LGD20H100002), Taizhou Science and Technology Project (1901ky76, 1902ky80), College Students' Innovation and Entrepreneurship Training Program Project of China (202010350054; 202110350052), and the Zhejiang Provincial Campus Student Research Training Program (2021R436020).
Ethical approval
Ethical approval for this study was obtained from Institutional Ethics Committee of Taizhou University (TZYXY2019-201).
