Abstract
This study was designed to investigate the hepatoprotective potentials of the Wu-Zi-Yuan-Chung-Wan (WZYCW) using an animal model of carbon tetrachloride (CCl4) induced liver injury. CCl4 induced chronic liver hepatotoxicity in adult Sprague-Dawley rats. Excluding the control group, all of the rats with chronic liver fibrosis received 0.4% CCl4 (1.5 mL/kg of body weight, ip) twice per week for 8 weeks. WZYCW (20, 100, and 500 mg/kg) and silymarin (200 mg/kg) were administered five times per week for 8 weeks. After 8 weeks, the rats were sacrificed, blood samples were obtained, and liver histological examinations were performed for subsequent assays. These results suggest that WZYCW considerably reduced Glutamic Oxaloacetic Transaminase (GOT), Glutamic Pyruvic Transaminase (GPT), Triglyceride (TG); and cholesterol activity; and the levels of malonaldehyde (MDA), nitric oxide (NO), and transforming growth factor-β1 (TGF-β1) in the liver. WZYCW also increased the level of superoxide dismutase (SOD), catalase (CAT), glutathione peroxidase (GPx), and glutathione reductase (GR) in liver tissue. WZYCW produced hepatoprotective and antifibrotic effects. This is the first study to demonstrate that WZYCW expressed hepatoprotective activity against CCl4 induced acute hepatotoxicity in rat. In addition, the primary compound of WZCYW was analyzed using HPLC. The major peaks of WZCYW, including schizandrin. The results indicate that WZYCW not only enhances hepatic antioxidant enzyme activities and inhibits lipid peroxidation but also suppresses inflammatory responses in CCl4 induced liver damage. Our findings provide evidence that WZYCW possesses a hepatoprotective activity to ameliorate chronic liver injury.
Introduction
Liver is the key organ of metabolism and excretion. It is often exposed to a variety xenobiotics and therapeutic agents. Until today, people have not yet found an actual curative therapeutic agent for liver disorder. Carbon tetrachloride (CCl4) has been widely used in animal models to investigate chemical toxin-induced liver damage. 1 Carbon tetrachloride (CCl4) is commonly used to study hepatotoxicity in animal models. 2 CCl4 can be metabolizedi not the highly reactive trichloromethyl radical and then trigger lipid peroxidation. 3 Therefore, blocking the lipid peroxidation can protect liver against CCl4-induced injury. 4 In this study, the hepatoprotective activity of the ethanol extract of Wu-Zi-Yuan-Chung-Wan (WZYCW) was investigated on CCl4-induced chronic liver injury in mice.
Wu-Zi-Yuan-Chung-Wan (abbrev. WZYCW), a traditional Chinese medicine with five herbal ingredients, was first described in Shung-Jie-Lou which is an ancient Chinese book, was composed of Plantago asiatica L., Lycium chinense L., Cuscuta chinensis Lam., Rubus idaeus Hu., and Schizandra chinensis Baill., Lycium chinense and Cuscuta chinensis possess germination and kidney function-enhancing effects. 5 Recent years, herbal medicines as a resource for liver diseases have been attracted by the world wide scientists, 6 among, which WZYCW is one of the potential herbs. It possesses anti-aging effect via improving symptoms of aging caused by kidney function decay, regulating the sex hormone level, decreasing LPO level in the blood, and elevating the activity. 7 Silymarin, an effective agent for liver protection, was used as a positive control. In addition, our previous study demonstrated WZYCW possessed anti-inflammatory activity. 8 The anti-inflammatory mechanisms of WZYCW in λ-carrageenan induced paw edema model may be increasing the antioxidant enzyme. Therefore, intake of oxygen radical scavengers (antioxidants and phytochemicals) from herb drugs be a good defense mechanism for hepatoprotection.
Inflammation is initiated by CCl4-induced hepatotoxicity, followed by the release of proinflammatory mediators including inducible NO synthase (iNOS) and TNF-α. TNF-α, which is produced by Kupffer cells, can induce immune responses by activating T cells and macrophages and further stimulate the secretion of other inflammatory cytokines and the production of NO. 9 Superoxide dismutase (SOD), glutathione peroxidase (GPx) and glutathione reductase (GR) are anti-oxidative enzymes which are easily inactivated by reactive oxygen species (ROS) and lipid peroxides which are caused by CCl4. 10 When ROS overwhelm the cellular antioxidant defense system, whether through an increase in ROS levels or a decrease in the cellular antioxidant capacity, oxidative stress occurs. Therefore, an increase in antioxidant activity and the inhibition of ROS are positively correlated with hepatic protection. From the above results, it is found that there is a close relationship between antioxidant enzymes and inflammation.
To sum up, the physiological function of WZYCW on oxidative stress-mediated hepatotoxicity has not been investigated yet. In the present study, CCl4-induced acute and chronic liver injury in rats was introduced as an animal model to investigate the protective effect of WZYCW. Silymarin was used as a positive control to against the CCl4-induced hepatic damage in rats. Serum AST and ALT was measured to observe the liver function in different treatment groups, and the SOD, GPx, GR, and Catalase activities were determined in rat liver tissue. Histological study was conducted to observe the morphological changes among different groups. To elucidate the underlying mechanism of WZYCW protection against chronic liver damage induced by CCl4.
Materials and methods
Materials and chemicals
CCl4 was purchased from Merck Co. GOT, GPT, TG, Cholesterol, SOD, GPx, and GR were purchased from Roche, UK. Nitrate/Nitrite colorimetric assay kit was purchased from Cayman, USA. Human TGF-β1 cytoset was purchased from Biosource, USA. Silymarin, sodium phosphate, thiobar-bituric acid (TBA) 1, 1, 3, 3-Teraethoxypropane (TEP) and butylated hydroxytoluene (BHT) were purchased from Sigma-Aldrich, USA. CCl4 was dissolved in olive oil as 50% (v/v) solution. Silymarin was suspended in 2% CMC (carboxymethylcellulose). Silymarin, an effective agent for liver protection, was used as a positive control. Schizandrin was purchased from Wako Pure Chemical Industries, Ltd. (Osaka). Polyvinylidene fluoride (PVDF) membrane filter was obtained from Millipore Corp (Bedford, MA, USA).
Plant material and crude extract preparation
According to the 8th edition Chinese pharmacopeia record, the Wu-Zi-Yuan-Chung-Wan was composed of Plantago asiatica L., Lycium chinense L., Cuscuta chinensis Lam., Rubus idaeus Hu., and Schizandra chinensis Baill at the ratio of 1:4: 4: 2: 0.5. After mixing the five herbal medicines, the 1.8 kg mixture was extracted with 10L 75% ethanol for four times. Ethanol was removed by distillation under reduced pressure and the remaining solution was lyophilized to yield the crude ethanol extract of WZYCW which was then stored in 20% refrigerator. The yield ratio of WZYCW was 22.2%. For the pharmacological tests, the extract was dissolved in saline solution prior to its use. The filtrates were collected and concentrated with a rotary evaporator under reduced pressure. The concentrated extract was then lyophilized and weighted. All of the herbal medicines were purchased from Shin-Long Drug store in Taichung city and identified by Dr. Kuo, Chao-Lin, leader of the Department of Chinese Pharmaceutical Sciences and Chinese Medicine Resources. The voucher numbers of TCMs of WZYCW are D1995001 (Dunqua).
HPLC analysis of WZYCW
The hot water extract of WZYCW was prepared by dissolving it in MeOH (5.0 mg/ml). Sample and standard solutions of schizandrin were filtered through a 0.22 m membrane filter prior to HPLC injection. HPLC was performed on a Shimadzu HPLC system (Japan) equipped with Shimadzu LC-20AT pump, Shimadzu SIL-20 auto sampler, and Shimadzu SPD- M20A detector (wavelength: 190–800 nm). The HPLC profile of the hot water extract of WZYCW was performed using a RP-18 column (SUPELCO, Discovery HS C18, 4.6 × 250 mm, 5 nm) at a flow rate of 1.0 mL/min, detected at UV 254 nm. The injection volume was 10 L. The mobile phase was composed of solvent (A), 0.1% TFA water solution, and solvent (B), methanol. The solvent gradient was as follows: 0–90 min from 5% B to 70% B; 90–100 min from 70% B to 100% B. A flow rate of 1.0 mL/min was used. By diluting the stock solution, a series of standard solutions (schizandrin) were prepared with concentrations of 2, 1, 0.5, 0.25, 0.1, 0.05, 0.025, 0.01, and 0.005 mg/ml and used to calculate the concentration of examined compounds.
Experimental animals
Sprague-Dawley rats, weighting between 230 and 260g, were obtained from the Animal Center of the College of Medicine in National Taiwan University. They were housed in standard cages at a constant temperature of 22 ± 1°C, relative humidity 55 ± 5% with 12 hours light-dark cycle (08:00 to 20:00) for at least 1 week before the experiment. Animals used in this study were housed and cared in accordance with the NIH Guide for the Care and Use of Laboratory Animals. 11 The experimental protocol was approved by the Committee on Animal Research, China Medical University. The animal use protocol listed below has been reviewed and approved by the Institutional Animal Care and Use Committee (IACUC, Protocol No: 104-243).
Animals and study design
A total of 60 Sprague-Dawley rats were randomly divided into six groups of 10 each: for the control group and the CCl4 group, rats were orally administered 1% carboxymethyl cellulose (CMC). For the silymarin group, rats were orally administered silymarin (200 mg/kg in 1% CMC). For the WZYCW groups, rats were orally administered WZYCW (20, 100, and 500 mg/kg in MeOH (5.0 mg/ml). Theses oral administrations were conducted using a feeding tube with 100 µL/10 g Body Weight every day for eight consecutive weeks. After 1 week of the administration of the silymarin and experimental drugs, 0.4% CCl4 (1.5 ml/kg of body weight) was started to inject intraperitoneally into all rats except for rats in the control group at one hour before the administration of the experimental drugs for every 3.5 days (twice a week) and eight consecutive weeks. Thus, there were sixteen times of CCl4 treatment for the five CCl4 treated groups. The control mice received an equivalent volume of olive oil. One week after the last administration of the experimental drugs, the rats were sacrificed under anesthesia (Ketamine + Medetomidine; 75 mg/kg + 0.5 mg/kg, ip) and their blood was collected from the heart.
Serum biochemistry
The blood was centrifuged at 3000 rpm (Beckman GS-6R, Germany) at 4°C for 30 min to separate the serum. Moreover, AST, ALT, triglyceride, and cholesterol were measured using spectrophoto-metric diagnostic kits.
Antioxidant enzymes activity measurements
SOD enzyme activity was determined according to the method as previously described at room temperature.12,13 One hundred micro liters of tissue extract was added to 880 µl carbonate buffer (0.05 M, pH 10.2, 0.1 mM EDTA). Twenty micro liters of 30 mM epinephrine (in 0.05% acetic acid) was added to the mixture and measured at 480 nm for 4 min on a Hitachi U 2000 spectrophotometer. The enzyme activity was presented as the amount of enzyme that inhibits the oxidation of epinephrine by 50% which is equal to 1 unit.
GPx enzyme activity was determined according to the method as previously described at 37°C.13,14 A reaction mixture was composed of 500 µl phosphate buffer, 100 µl 0.01 M GR (reduced form), 100 µl 1.5 mM NADPH and 100 µl GR (0.24 units). One hundred micro liters of the tissue extract was added to the reaction mixture and incubated at 37◦C for 10 min. Then 50 µl of 12 mM t-butyl hydro peroxide was added to 450 µl tissue reaction mixture and measured at 340 nm for 180 s. The molar extinction coefficient of 6.22×10−3M−1 cm−1 was used to determine the enzyme activity. One unit of activity is equal to the mM of NADPH oxidized / min per mg protein. GR enzyme activity was determined following the method as previously described.13,14 Fifty micro liters of NADPH (2 mM) in 10 mMTris buffer (pH 7.0) added in a cuvette containing 50 µl of GSSG (20 mM) in phosphate buffer. One hundred micro liters of tissue extract was added to the NADPH-GSSG buffered solution and measured at 340 nm for 3 min. The molar extinction coefficient of 6.22×10−3M−1 cm−1 was used to determine GR enzyme activity. One unit of activity is equal to the mM of NADPH oxidized / min per mg protein.
Catalase activity was measured by the method of Goth. 15 A 0.1 mL of supernatant was added to cuvette containing 1.9 mL of 50 mM phosphate buffer (pH 7.0). Reaction was started by addition of 1.0 mL of freshly prepared 30 mM H2O2. The rate of decomposition of H2O2 was measured spectrophoto metrically at 240 nm. Activity of catalase was expressed as U/mg of protein.
MDA assay
As MDA level is usually used to elucidate the level of lipid peroxidation in liver, the effects of WZYCW on CCl4-induced MDA production were examined. MDA was evaluated by the thiobarbituric acid reacting substance (TRARS) method.12,13 Briefly, MDA reacted with thiobarbituric acid in the acidic high temperature and formed a red-complex TBARS. The absorbance TBARS was determined at 532 nm.
NO assay
The free radical NO is a highly active nitrogen species produced by both parenchymal and nonparenchymal liver cells from l-arginine through NO synthase. Nitrate was converted to nitrite by using nitrate reductase. Nitrite subsequently reacted with sulfanilic acid to produce diazonium ions, which reacted with N-(1-naphthyl) ethylenediamine to produce a chromophoric azo derivative (purplish red), which was recorded at 540 nm. The sum of nitrite and nitrate obtained in this procedure are represented as μM. NO was measured according to the previous method. 14
TGF-β1 assay
TNF-α, is critical cytokines in inflammatory responses, and its level is increased during the development of liver damage. TNF-α, which is produced by Kupffer cells, can induce immune responses by activating T cells and macrophages and further stimulate the secretion of other inflammatory cytokines and the production of NO Hepatic. TGF-β1 levels were determined by ELISA (Quantikine MB100, R&D Systems). The liver was homogenized in buffer containing 20 mM Tris-HCl (pH 7.5), 2 M NaCl, 1 mM EDTA, 1 mM PMSF, and 0.1% Tween-80. Latent TGF-β1 in the liver homogenate (200 µl) was activated to the immuno reactive form by 40 µl of 1 M HCl, and neutralized by 26 µl of 1.2 M NaOH and 0.5 M HEPES. 16 The liver homogenate (50 µl) was subsequently incubated with monoclonal anti- TGF-β1 antibody, polyclonal antibody against TGF-β1 conjugated to horseradish peroxidase, and the substrates (H2O2 and tetramethyl benzidine). The level of TGF-β1 was measured at 450 nm and corrected at 540 nm.
Histological analysis
The results of hematoxylin and eosin histological analyses showed that CCl4 induced histological changes including increased hepatic cells cloudy swelling, cytoplasmic vacuolization, lymphocytes infiltration, hepatocellular, and necrosis. All animals were subjected to necropsy at the end of the experiment. The liver was observed grossly and excised, blotted, and weighed. The weights of the livers were also measured and presented as the percentage to the body weight. Tissues were fixed in 10% buffered formaldehyde solution and embedded in paraffin. The paraffin wax were cut into 2 µm sections, stained with hematoxylin and eosin, and then examined under the light microscopy. The liver injury lesions were evaluated using the Knodell method17,18 and the degree of liver fibrosis according to the Ruwart and Maedeh method.19,20
Statistical analysis
All data were expressed as mean ± SEM. Statistical analyses were performed using one-way ANOVA followed by Scheffe’s multiple range tests by SPSS software. 21 The criterion for statistical significance was p < 0.05. Histological analysis was carried out using non-parametric Kruskal-Wallis test followed by Mann-Whitney U-test. The criterion for statistical significance was p < 0.05.
Results
Qualification of the WZYCW extraction procedure
The reference compound (schizandrin) within the water extracted from WZYCW was identified using high-performance liquid chromatography (HPLC) and was used as an indicator of the extraction quality of each batch (Figure 1). Using HPLC quantification, the schizandrin content was calculated as 0.125 mg/mL of WZYCW.

The HPLC profile of WZYCW: (a) the chemical profile of the WZYCW was performed using a RP-18 column and detected at UV 254 nm (see materials and methods) and (b) HPLC chromatograms of the reference compound (schizandrin, tR = 80.7 min).
Effects of WZCYW and silymarin on serum GOT activity in CCl4-treated rats at Weeks 1, 3, 6, and 8
As shown in Table 1, serum GOT activity in rats injected with CCl4 twice per week for 8 weeks increased substantially compared with that in the normal rat (***p < 0.001). After WZCYW and silymarin were administered during the CCl4 treatment period, a dose-dependent decrease in serum GOT activity and an increase in the standard level of activity reaching 75% were observed in rats treated with 500 mg/kg for 8 weeks compared with those exhibited in the normal and CCl4 control groups (***p < 0.001).
Effects of WZCYW and Silymarin on serum got activity in CCl4-treated rats at weeks 1, 3, 6, and 8.

Values are mean ± SEM (n = 10).
Indicates significant difference from the control group (###p < 0.001).
Indicates significant difference from the CCl4 group (*p < 0.05, **p < 0.01, ***p < 0.001).
Effects of WZCYW and silymarin on serum GPT activity in CCl4-treated rats at Weeks 1, 3, 6, and 8
As shown in Table 2, serum GPT activity in rats injected with CCl4 twice per week for 8 weeks increased substantially compared with that exhibited in the normal rat (###p < 0.001). After treatment with WZCYW (20, 100, and 500 mg/kg) and silymarin (200 mg/kg), serum GPT activity decreased in the CCl4-treated rats at Weeks 1, 3, 6, and 8 compared with that in the normal and CCl4 control groups (***p < 0.001).
Effects of WZCYW and Silymarin on serum GPT activity in CCl4-treated rats at weeks 1, 3, 6, and 8.
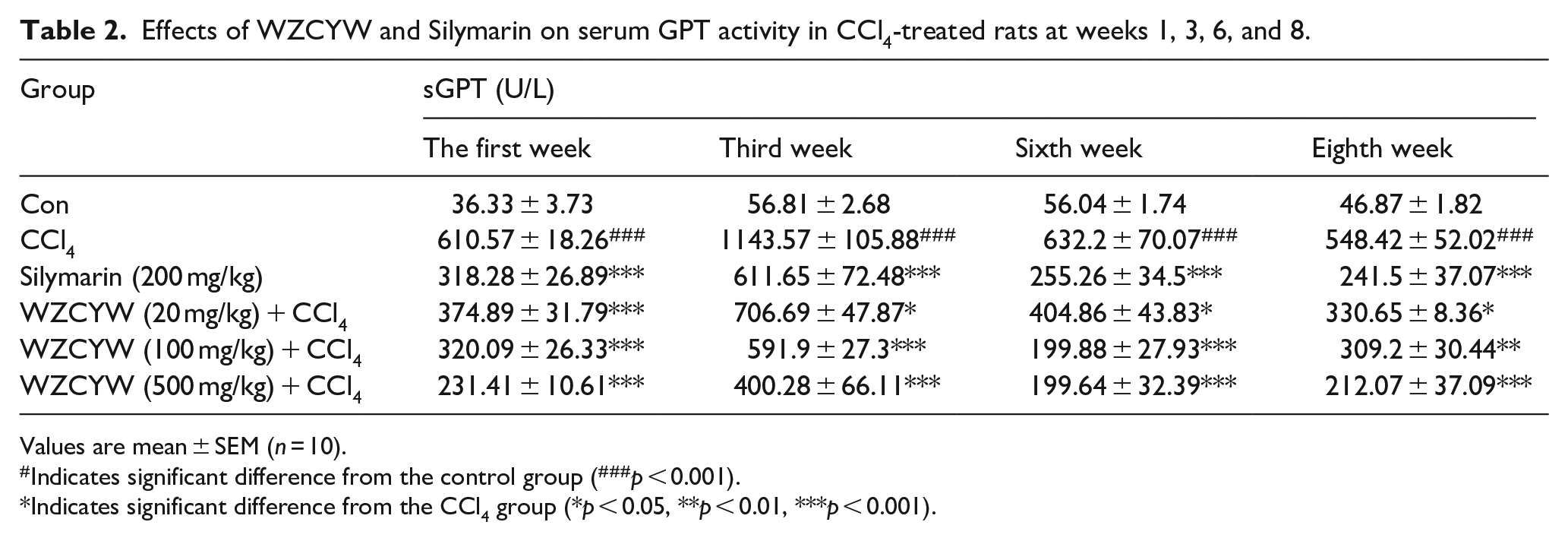
Values are mean ± SEM (n = 10).
Indicates significant difference from the control group (###p < 0.001).
Indicates significant difference from the CCl4 group (*p < 0.05, **p < 0.01, ***p < 0.001).
Effects of WZCYW and silymarin on serum TG activity in CCl4-treated rats at Weeks 1, 3, 6, and 8
As shown in Table 3, serum TG activity in rats injected with CCl4 twice per week for 8 weeks increased substantially compared with that in the normal rat (###p < 0.001). After treatment with WZCYW (20, 100, and 500 mg/kg) and silymarin (200 mg/kg), decreased serum TG activity in the CCl4-treated rats was observed at Weeks 1, 3, 6, and 8 compared with that in the normal and CCl4 control groups (***p < 0.001).
Effects of WZCYW and Silymarin on serum TG activity in CCl4-treated rats at weeks 1, 3, 6, and 8.

Values are mean ± SEM (n = 10).
Indicates significant difference from the control group (#p < 0.05, ##p < 0.01,###p < 0.001). *Indicates significant difference from the CCl4 group (*p < 0.05, **p < 0.01, ***p < 0.001).
Effects of WZCYW and silymarin on serum cholesterol activity in CCl4-treated rats at Weeks 1, 3, 6, and 8
As shown in Table 4, serum cholesterol activity in rats injected with CCl4 twice per week for 8 weeks increased substantially compared with that in the normal rat (###p < 0.001). After treatment with WZCYW (20, 100, and 500 mg/kg) and silymarin (200 mg/kg), the serum cholesterol activity in the CCl4-treated rats at Weeks 1, 3, 6, and 8 weeks exhibited a decline compared with that in the normal and CCl4 control groups (***p < 0.001).
Effects of WZCYW and Silymarin on serum cholesterol activity in CCl4-treated rats at weeks 1, 3, 6, and 8.

Values are mean ± SEM (n = 10).
Indicates significant difference from the control group (###p < 0.001).
Indicates significant difference from the CCl4 group (*p < 0.05, **p < 0.01, ***p < 0.001).
Effects of WZCYW and silymarin on antioxidant enzyme activity in CCl4-treated rats
As shown in Table 5, a considerable increase (p < 0.01) in liver antioxidant enzyme activity was observed in the CCl4-treated rats compared with that exhibited in the normal control group. Prefeeding with 500 mg/kg b.w. of WZCYW considerably enhanced (p < 0.001) the antioxidant enzyme activity (SOD, CAT, GPx, and GR) in the liver of the WZCYW-treated (CCl4+WZCYW) rats in a dose-dependent manner. Similarly, treatment with silymarin (200 mg/kg b.w.) considerably increased (p < 0.001) SOD, CAT, GPx, and GR activity in the liver.
Effects of WZCYW and Silymarin on the activity of antioxidant enzymes in CCl4-treated rats.

Values are mean ± SEM (n = 10).
Indicates significant difference from the control group (##p < 0.01, ###p < 0.001).
Indicates significant difference from the CCl4 group (*p < 0.05, **p < 0.01, ***p < 0.001).
Effects of WZYCW and silymarin on NO levels in the liver of CCl4-intoxicated rats
The NO levels in various groups of rats are shown in Figure 2. In the CCl4 group, the NO level was 2.5-fold higher than that in the normal control group (p < 0.001). Treatment with WZCYW (100 and 500 mg/kg b.w.) reduced NO levels to 60% and 50% (p < 0.05, p < 0.01). In addition, treatment with silymarin (200 mg/kg b.w.) reduced serum NO levels to 57% (p < 0.01).
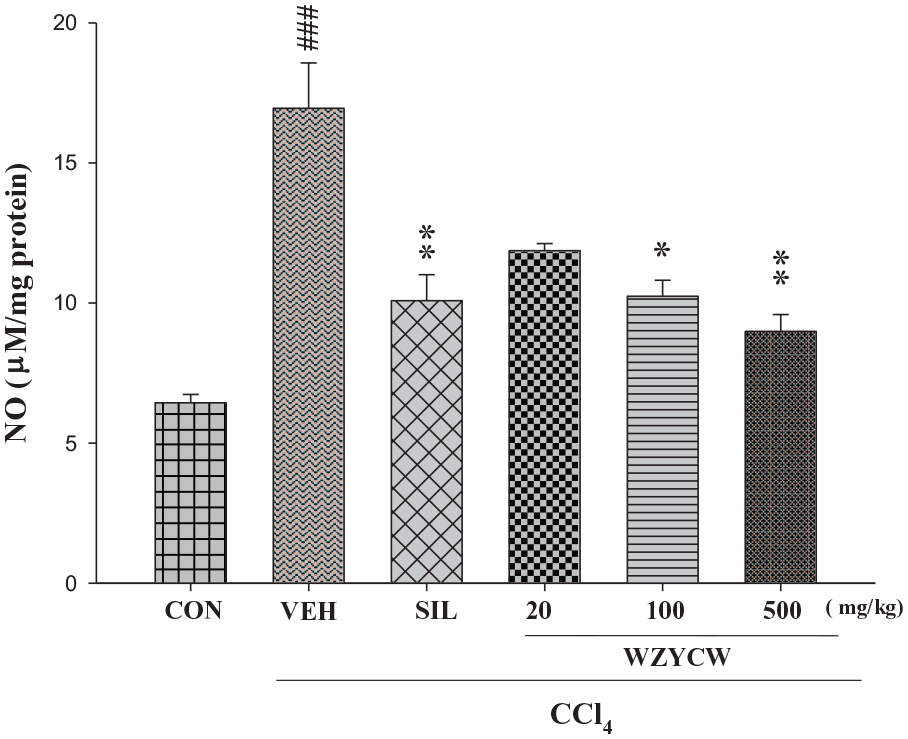
Effects of WZYCW and silymarin on liver NO level in CCl4-intoxicated rats. Each value represented as mean ± SEM (n = 10).
Effects of WZYCW and silymarin on the TGF-β1 level in the liver of CCl4-intoxicated rats
The TGF-β1 levels for various groups of rats are shown in Figure 3. For the CCl4 group, the TGF-β1 level was 1.5-fold higher than that in the normal control group (p < 0.001). Treatment with WZCYW (100 and 500 mg/kg b.w.) or silymarin (200 mg/kg b.w.) reduced the level of TGF-β1 by 22%, 35%, and 32% (p < 0.05, p < 0.001, and p < 0.01).

Effects of WZYCW and silymarin on liver TGF-β1 level in CCl4-intoxicated rats. Each value represented as mean ± SEM (n = 10).
Effects of WZYCW and silymarin on malondialdehyde (MDA) levels in the liver of CCl4-intoxicated rats
To evaluate the protective effect of WZYCW for CCl4-induced oxidative liver damage in rats, we identified the primary antioxidant enzymes in liver. Because of lipid peroxidation, CCl4 considerably (p < 0.01) increased MDA concentrations in the liver for the CCl4-treated group (Figure 4) compared with that for the normal control group. In addition, MDA concentrations in the liver were reduced in a dose-dependent manner through the preadministration of WZYCW.

Effects of WZYCW and silymarin on liver MDA level in CCl4-intoxicated rats. Each value represented as mean ± SEM (n = 10).
Effects of WZYCW and silymarin on the total protein levels in the liver of CCl4-intoxicated rats
The total protein levels in the liver for various groups of rats are shown in Figure 5. In the CCl4 group, the total protein level in the liver was reduced by 30% compared with that in the normal control group (p < 0.001). Treatment with WZCYW (100 and 500 mg/kg b.w.) and silymarin (200 mg/kg b.w.) increased the total protein level in the liver by 25%, 30%, and 22% (p < 0.05, p < 0.01, and p < 0.05).

Effects of WZYCW and silymarin on liver total protein level in CCl4-intoxicated rats. Each value represented as mean ± SEM (n = 10).
Effect of WZYCW and silymarin on liver weight in CCl4-intoxicated rats
Table 6 shows the liver weight of the rats in the CCl4 group (relative liver weight g/100 g of b.w.) compared to the substantial increase in liver weight observed in the normal control group. Treatment with WZCYW (100 and 500 mg/kg b.w.) or silymarin (200 mg/kg b.w.) reduced the liver weight (relative liver weight g/100 g of b.w.) of the CCl4-intoxicated rats.
Effect of WZYCW and Silymarin on liver weight in CCl4 intoxicated rats.

Values are mean ± SEM (n = 10).
Indicates significant difference from the control group (###p < 0.001).
Indicates significant difference from the CCl4 group (**p < 0.01).
Histologic liver injury score following various doses of WZYCW and silymarin for rats treated with CCl4
Our results showed that major changes in histology induced by CCl4 resulted in increased vacuolization, inflammation, and coagulative necrosis (Figure 6) when compared with the control group. Liver injuries were reduced by pretreatment with WZCYW (500 mg/kg b.w.), and the injury scores of vacuolization, inflammation, and hepatocellular necrosis significantly decreased (Table 7). Our histological findings also demonstrated that pretreatment with WZCYW significantly prevented CCl4-induced liver injury.

WZYCW improved chronic CCl4-induced rat liver damage, a histological examination using H&E staining: (a) control group, (b) CCl4, (c) CCl4+silymarin (200 mg/kg), and (d) CCl4+WZYCW (500 mg/kg) (100× magnification).
Quantitative summary of the protective effects of silymarin and WZYCW on CCl4-induced hepatic damage based on histological observations.
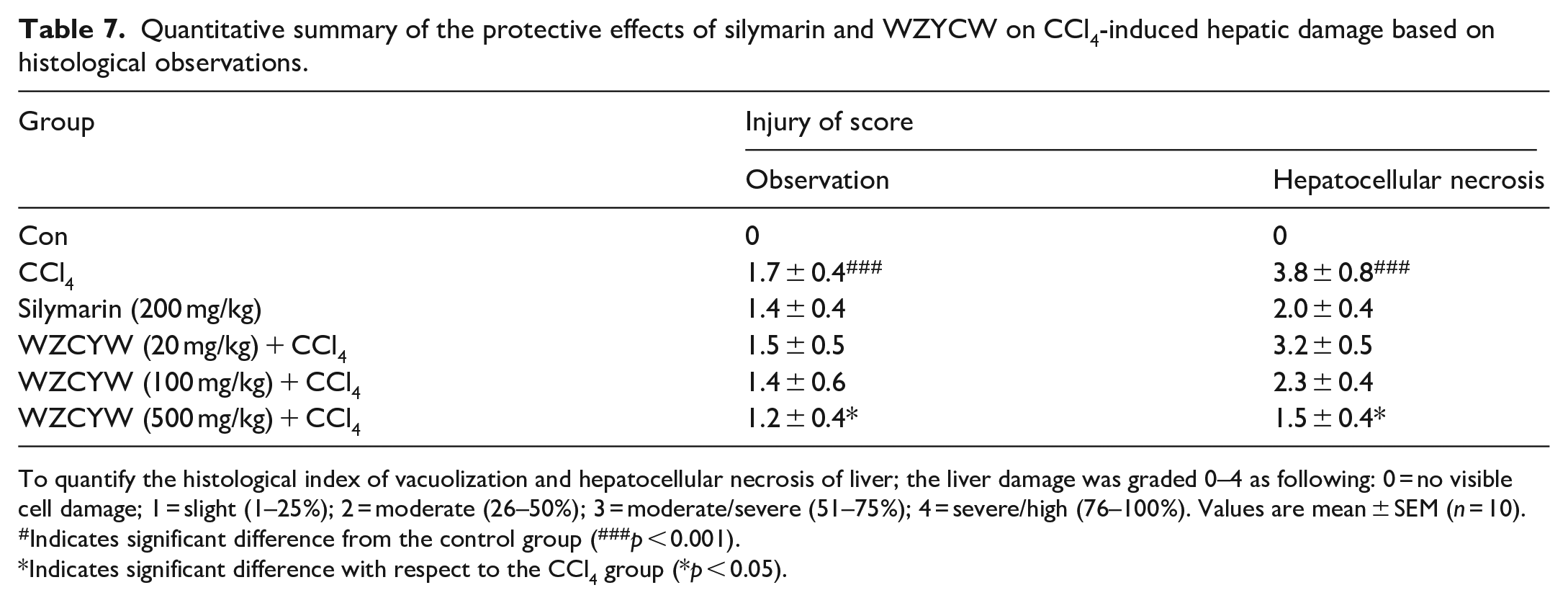
To quantify the histological index of vacuolization and hepatocellular necrosis of liver; the liver damage was graded 0–4 as following: 0 = no visible cell damage; 1 = slight (1–25%); 2 = moderate (26–50%); 3 = moderate/severe (51–75%); 4 = severe/high (76–100%). Values are mean ± SEM (n = 10).
Indicates significant difference from the control group (###p < 0.001).
Indicates significant difference with respect to the CCl4 group (*p < 0.05).
Discussion
Hepatotoxin CCl4 is commonly used to induce liver fibrosis in animal models.1,4 In this study, the hepatoprotective activity of the ethanol extract of WZCYW was investigated on CCl4-induced chronic liver injury in rats. Once liver damage has occurred, liver marker enzymes (alanine aminotransferase (ALT), and aspartate aminotransferase (AST)) and lipid profile (total triglyceride and cholesterol) will be increased. 22 Therefore, the levels of serum ALT, AST, cholesterol and triglyceride were measured in this study. In this study, as shown in Tables 1 to 4, the serum ALT, AST, and TG levels and cholesterol activity in rats injected with CCl4 twice per week for 8 weeks increased substantially compared with those of the normal rat (p < 0.01). The oral administration of WZCYW (20, 100, and 500 mg/kg) during the CCl4 treatment period resulted in dose-dependent decreases in the serum ALT, AST, and TG levels and cholesterol activity. These results clearly suggested that WZCYW possess protective properties against CCl4-induced liver injury.
In addition, liver biopsies were performed for examining the pathological changes. The most remarkable pathological characteristics of CCl4-induced hepatotoxicity are liver steatosis, cirrhosis, and necrosis, which are believed to result from the formation of reactive intermediates, such as trichloromethyl free radicals (CCl3+), metabolized by the mixed function cytochrome p450 in the endoplasmic reticulum.1,4 Oxidative stress-induced overproduction of free radicals causes the lipid peroxidation of the hepatocellular membrane, leading to a series of cellular event cascades involving a massive release of inflammatory mediators and cytokines.1,4 Thus, continuous administration of CCl4 results in extended oxidative stress and eventually chronic liver damage. Hepatic damage, such as liver fibrosis, cirrhosis, and atrophy, can initiate regeneration and, thus, increase liver weight. 23 Compared with the CCl4 group, the WZCYW extract groups exhibited increased weight gain without changes in feed consumption, which indicates that rat growth was not affected by herbal treatments.
Antioxidant enzymes (SOD, GPx, GR, and catalase) provide one form of protection against oxidative tissue damage. 24 SOD converts O2 into H2O2. GPx and catalase metabolize H2O2 into nontoxic products. The GSH antioxidant system comprises GSH and an array of functionally related enzymes and plays a fundamental role in the cellular defense against reactive free radicals and other oxidant species. Of the related enzymes, GR is responsible for GSH regeneration, and GPx and GST combine with GSH to decompose hydrogen peroxide and other organic hydroperoxides. 25 The results revealed increased MDA concentrations in the liver following CCl4 treatment, which suggests that oxidative damage to the liver also increased. In this study, treatment with WZCYW (500 mg/kg) resulted in the increased MDA levels and decreased SOD, GSH, GR, GPx, and catalase antioxidant enzymes levels returning to the standard control levels, which suggests that WZCYW may prevent lipid peroxidation by CCl4. According to the result of the histological examination, treatment with WZCYW or silymarin suppressed chronic hepatic damage and was consistent with improvements in the serum biological parameters of hepatotoxicity.
Regarding the expression of the transforming growth factor (TGF-β1) for the synthesis of extracellular matrices in most cytokines, the most crucial factor is fibrosis. 26 TGF-β1 stimulated hepatic stellate cells, resulting in numerous first, three type I collagen, which led to the occurrence of fibrosis, and the inhibition of TGF-β1 can reduce the generation of liver fibrosis. 27 The results indicated that the oral administration of WZCYW (100 and 500 mg/kg b.w.) and silymarin (200 mg/kg b.w.) can effectively reduce TGF-β1 levels in rat liver. This finding indicates that the antifibrosis mechanism of WZCYW might be related to inhibited TGF-β1 secretion, which reduces the activation of hepatic stellate cells, resulting in decreased liver fibrosis. Histopathological studies have reported consistent results. Disruption of the tissue architecture, fiber extensions, large fibrous septa formation, pseudolobe separation, and collagen produced by CCl4 treatment was observed in rats ameliorated after WZCYW administration, indicating that WZCYW can considerably protect hepatocyte from peroxidative damage caused by CCl4 (Figure 6).
Identifying the major compounds in herbs or herbal preparations may benefit efforts to understand their pharmacological activity and mechanisms. 28 The primary compound of WZCYW was analyzed using HPLC. The major peaks of WZCYW, including schizandrin (Figure 1), were identified. Schizandrin, is widely used as a tonic in traditional Chinese medicine.29,30 Previous studies have shown that schizandrin possesses numerous biological properties, 30 including hepatoprotective 31 and anti-inflammatory, 32 as well as suppressive effects against lipid peroxidation and potentiating effects on glutathione-mediated antioxidation. From the above results, we boldly infer that WZCYW has a liver protection effect that may be related to the content of schizandrin.
Conclusion
The present study clearly elucidated that WZCYW exhibited a hepatoprotective activity against CCl4-induced chronic liver injury in rats. The underlying mechanisms were likely the decreasing in MDA, NO, and TGF-β1 level through enhancing the activities of hepatic anti-oxidative enzymes such as CAT, GPx, GR, and SOD, and thereby the significant decrease of serum ALT, AST, triglyceride, and cholesterol. In addition, fibrosis of liver was significantly reduced by WZCYW. Therefore, WZCYW can be developed into pharmacological agents to prevent some liver disorders.
Footnotes
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation
Data availability
All relevant data are within the paper and its supporting information files.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical clearance certificate was obtained from the Research Ethical Clearance Committee (RECC) of the China Medical University, Taiwan. (Approval no: 04/2012/101-132).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by the National Science Council, Taiwan (NSC 101-2320-B-039-032-MY2), Taiwan Department of Health Clinical Trial and Research Center of Excellence (DOH100-TD-B-111-004) and Committee on Chinese Medicine and Pharmacy, Department of Health, Executive Yuan (CCMP102-RD-104, CCMP102-RD-019).
Significance statement
The present study is unique in demonstrating direct between WZCYW antifibrotic treatment and damaged liver in rat models. WZCYW is demonstrated in this study to exhibit a hepatoprotective effect against chemically induced liver injury, it can be potentially developed into a functional drug or even a pharmacological agent for the prevention of liver diseases. The findings will help researchers to pursue more refined studies of the hepatoprotective study.
