Abstract
Sepsis is a fatal infectious disease accompanied by multiple organ failure. Immune dysfunction and inflammatory response play an important role in the progression of the disease. Tripterygium glycoside (TG) has immune suppression and anti-inflammatory effects. Here, we investigated the effects of TG on cecal ligation and puncture (CLP)-induced sepsis. Septic mice model was induced by cecal ligation and puncture(CLP), after administration of TG, specimens are collected at designated time points. Histopathology changes of lung tissues and Kidney tissues were observed under light microscope, magnetic microbeads were used to isolate splenic CD4+CD25+ regulatory T cells (Tregs), and phenotypes were then analyzed by flow cytometry. ELISA method was employed to detect the concentrations of plasma TNF-α, IL-6, and IL-10. Nuclear p-NF-κB and Cytoplasmic IkB-a was detected by western blot. TG administration significantly alleviated lung and kidney inflammatory injury and improved the survival of septic mice. Furthermore, the suppressive function of regulatory T cells was enhanced and plasma expression of IL-10 was increased following TG treatment. The NF-B signaling pathway and secretion of plasma TNF-α and IL-6 was notably inhibited in septic mice treated with TG. TG exerts protective effects through improving regulatory T cells and attenuating pro-inflammatory cytokines in septic mice.
Introduction
Sepsis is a dysregulated response to severe infection characterized by life-threatening organ failure and is the leading cause of mortality worldwide. Multiple organ failure is the central characteristic of sepsis and is associated with poor outcome of septic patients. 1 Despite progress in antibiotic therapy, ventilator management, resuscitative strategies, and blood glucose maintenance, sepsis remains the leading cause of death in the intensive care unit (ICU). 2
The immune response in sepsis can be characterized by a cytokine-mediated hyper-inflammation phase in which most patients survive, and a subsequent hypo-inflammation (immune paralysis or immunosuppression) phase. 3 Regulatory T cells (Tregs) are a component of adaptive immunity that suppresses the responses of other effector T cell subsets. This helps to maintain tolerance to self-antigens and suppress autoimmune disease. 4 The enhanced suppressive function of CD4+CD25+ Tregs is associated with fatal outcomes in patients with septic shock.5,6 Forkhead/winged helix transcription factor p3 (FOXP3) is specifically expressed in CD25+CD4+ Tregs and controls their development and function. 7 Previous studies have demonstrated that the suppressive function of Tregs restrains inflammatory responses in diverse diseases, including sepsis.8,9
Tripterygium glycoside (TG), an active ingredient of the widely used Chinese herb Tripterygium wilfordii Hook F, has immunosuppressive and anti-inflammatory effects.10–12 It has been used extensively to treat autoimmune and inflammatory diseases, including rheumatoid arthritis, systemic lupus erythematosus, and nephrotic syndrome.10,13 However, few studies have been reported on the role of TG in sepsis. The present study explores the effects of TG on immune status and inflammatory response in septic mice.
Methods
Reagents and kits
TG was purchased from Zhejiang Deende Pharmaceutical Co., Ltd. (Zhejiang, China). Mouse creatinine and urea nitrogen detection kits were provided by Nanjing Jiancheng Biology Engineering Institute (Nanjing, China). NE-PER Nuclear and Cytoplasmic Extraction Kits were provided by Thermo Scientific (Grand Island, USA). The rabbit polyclonal antibody against murine NF-κB p65 and IκB-a was purchased from Santa Cruz Biotechnology (Santa Cruz, USA).
Mouse CD4+ T Cell Isolation Kit were purchased from Miltenyi Biotec GmbH (Bergisch Gladbach, Germany). The antibodies used for flow cytometric analysis, including fluorescein isothiocyanate (FITC)-conjugated anti-mouse CD4, allophycocyanin (APC)-conjugated anti-mouse CD25, phycoerythrin (PE)-conjugated anti-mouse CD25 and PE-conjugated anti-mouse Foxp3, were purchased from eBioscience (San Diego, CA). Anti-mouse CD3 and CD28 were purchased from eBioscience (San Diego, CA). Enzyme-linked immunosorbent assay (ELISA) kits for murine IL-10, IL-6, and TNF-α were purchased from Biosource (Worcester, MA). The Cell Counting kit-8 (CCK-8) was purchased from Dojindo Lab (Kumamoto, Japan).
Experimental design
In the survival experiment, BalB/c mice (n = 75) were randomly divided into the following five groups (n = 15/group): Sham, CLP, and CLP and TG (1, 5, 10 mg/kg). All mice were provided with food and water ad libitum and were monitored every 6 h. The mice were monitored for 7 days to analyze the long-term effects of TG. The limitation of this study is that the power calculation and justification of the sample size did not provide.
All mice in the TG groups received TG (1, 5, 10 mg/kg) daily via intragastric administration after CLP introduction. All mice in the Sham group received an equal volume of normal saline daily until the end of the experiment. 40 animals were randomly divided into the following five groups, eight mice in each group: the Sham group (abbreviation: Sham), CLP group (abbreviation: CLP), CLP+TG 1 mg/kg group (abbreviation: TG 1 mg), CLP+TG 5 mg/kg group (abbreviation: TG 5 mg), and CLP+TG 10 mg/kg group (abbreviation: TG 10 mg).
Mouse model of sepsis
The cecal ligation and puncture (CLP) method is a commonly used procedure for modeling sepsis in vivo. 14 BALB/c mice were anesthetized with amobarbital sodium (0.05 g/kg) and placed supine on a warming pad at 37°C with the abdomen shaved. Under sterile conditions, the abdominal skin and muscle were opened and the cecum exposed. the cecum was prolene suture ligated over its distal portion and the cecum distal to the ligature was punctured twice with an 18-gauge needle to allow the cecal contents to be expressed intraperitoneally, The wound was closed and the animal allowed to recover from anesthesia. The sham group receiving a laparotomy but no CLP.
Magnetic isolation of splenic CD4+ T
The spleen was immediately obtained after the mice were sacrificed. Splenocytes were isolated aseptically using a cell mesh. CD4+ T cells were enriched via depletion of cells expressing CD8a, CD11b, CD45R, CD49b, and Ter-119 from splenocytes using a CD4+ T Cell Isolation Kit.
Flow cytometry
To assess CD4+CD25+ Treg and Foxp3 expression, cells were stained with anti-mouse CD4-FITC and CD25-APC antibodies for 30 min at 4 °C in the dark. Intranuclear Foxp3 was simultaneously detected using cells treated with 1 mL of freshly prepared fixation/permeabilization working solution for 1 h at 4°C. After washing the cells twice with permeabilization buffer, the cells were stained using an anti-mouse/rat Foxp3-PE antibody for 30 min at 4°C in the dark. Following two more washing steps, the cells were analyzed via flow cytometry.
Proliferation assay
Isolated CD4+ T cells were co-incubated for 72 h with 1 mg/mL of soluble anti-CD3 and anti-CD28 antibodies in a humidified environment with 5% CO2 at 37°C. CCK-8 solution (10 μL) was added to each well, followed by incubation for 4 h at 37°C with 5% CO2. The absorbance values of the samples were measured at 450 nm using a microplate reader. The optical density at 450 nm represents the amount of cell proliferation.
Serum cytokine assay
Blood samples were harvested at specified time points. The concentration of IL-10, IL-6, and TNF-α were assessed in serum samples by quantitative sandwich immunoassays performed on a fully automated ELISA machine using ELISA kits. Estimation of each cytokine was performed in accordance with the manufacturers’ instructions.
Assessment of renal function
Renal functions were assessed by blood urea nitrogen (BUN) and serum creatinine (Scr) levels at Nanjing Jian-Cheng Biology Engineering Institute.
Western blotting
The nuclear and cytoplasmic proteins of lymphocytes were prepared using a NE-PER Nuclear and Cytoplasmic Extraction Kit. The proteins were then transferred to polyvinylidene fluoride (PVDF) membranes. The membranes were blocked with 5% non-fat milk in TBST buffer and probed with rabbit anti-rat antibodies against NF-κB p65, IκB-a, lamin B, and β-actin overnight at 4°C. Then, the membranes were exposed to the horseradish-peroxidase-conjugated secondary antibody for 2 h at room temperature. Signals were detected using an ECL kit and X-ray films.
Histopathological examination
The kidneys and lungs were collected at the indicated time points. They were fixed in 10% neutral-buffered formalin, embedded in paraffin, sectioned, and stained with hematoxylin and eosin (H&E). Lung injury was scored from 0 to 4 (normal to severe) based on alveolar edema, hemorrhage, and neutrophil infiltration. The total score for each sample was calculated. Kidney injury was scored from 0 to 3 (normal to severe) based on glomerular epithelial hyperplasia, tubular dilatation, protein cast formation, and inflammatory cell infiltration.
Statistical analysis
Data are expressed as mean (M) ± standard error of mean (SEM). Quantitative data were analyzed using the one-way analysis of variance (ANOVA) method. The survival rate was determined using the Kaplan-Meier estimator. Statistical analyzes were performed with SPSS 22.0 software. p < 0.05 was considered statistically significant.
Results
The effect of TG on the survival of mice after CLP
To clarify the protective response of TG against CLP-induced mortality, 75 mice were randomly divided into five groups (Sham, CLP, TG 1 mg, TG 5 mg, and TG 10 mg). Mice were assessed for 7 days. The survival rate was significantly improved by 60% in TG 10 mg-treated mice when compared with the CLP mice (Figure 1; n = 15; p < 0.05).

Effect of TG on the survival of mice after CLP. 75 BalB/c mice were randomly divided into three groups (sham group, CLP group, TG 1 mg group, TG 5 mg group, TG 10 mg group). The mice were monitored daily for 7 days. The survival rate was analyzed by Kaplan-Meier survival analysis and compared by the log-rank test. #p < 0.05 versus sham; *p < 0.05 versus CLP; $p < 0.05 versus CLP; &p > 0.05 versus CLP.
TG alleviated tissue injury in septic mice
Mice were sacrificed on post-operation days 1, 3, and 5. The induction of CLP led to severe tissue injury, exhibiting hemorrhage, alveolar edema, and neutrophil infiltration (Figure 2). In addition, the CLP group exhibited tubular epithelial necrosis, tubular dilatation, protein cast formation, and inflammatory cell infiltration (Figure 3). Lung and renal histological scores were increased in the CLP group when compared with the Sham group. However, the extent of tissue injury was alleviated in the TG-treatment groups (1, 5, and 10 mg/kg). These data suggest that TG alleviated inflammatory tissue injury in septic mice.
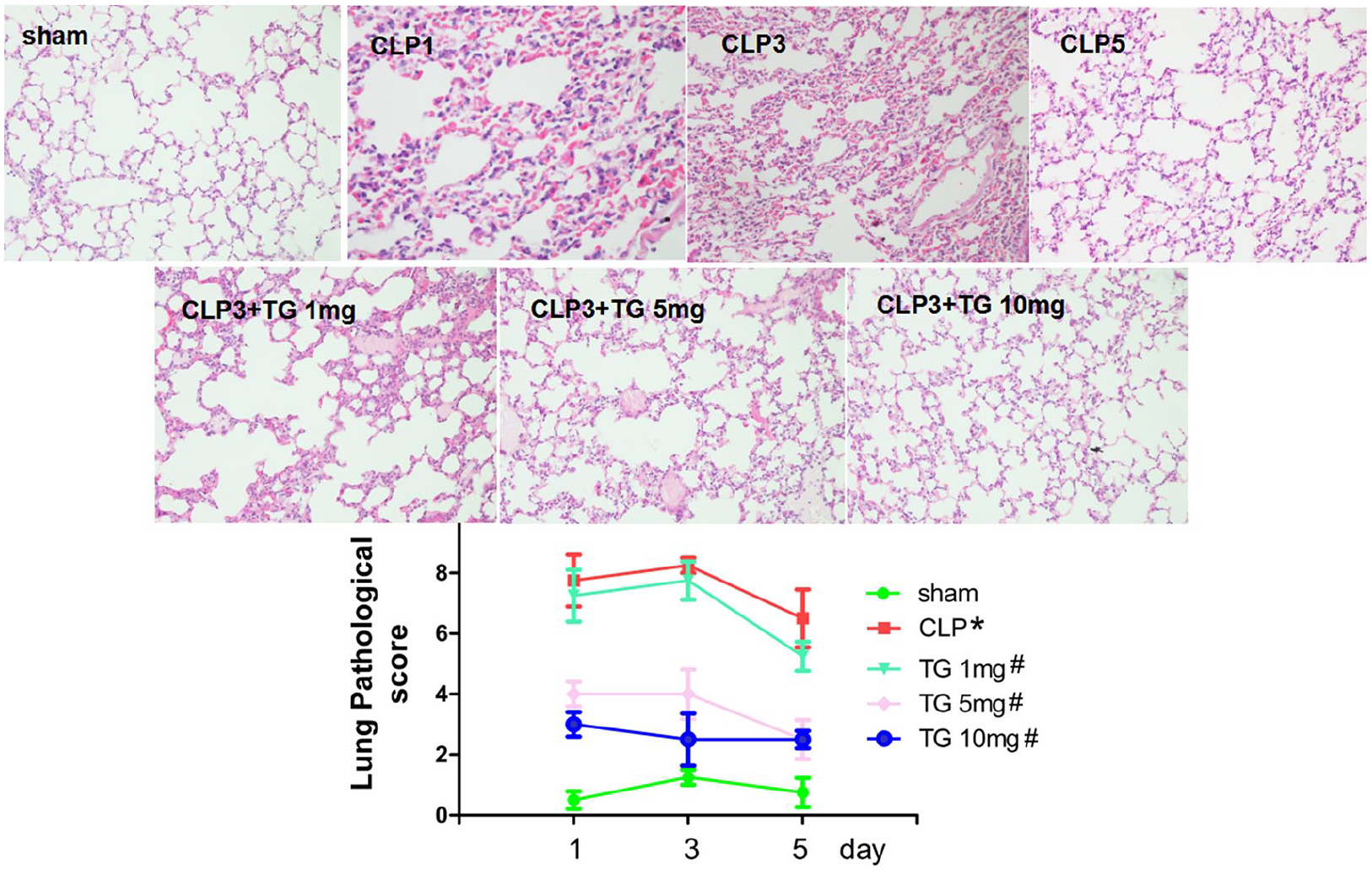
TG alleviated lung injury in septic mice. BalB/c mice were sacrificed on post-operation days 1, 3, and 5. Inflammatory lung injury was evaluated by histological observation and scores. Pathological images represent the results of a representative experiment on day 3 after operation. *p < 0.05 versus sham; #p < 0.05 versus CLP.

TG alleviated kidney injury in septic mice. BalB/c mice were sacrificed on post-operation days 1, 3, and 5. Inflammatory kidney injuries were evaluated by histological observation and scores. Images represent one representative experiment on day 3 after the operation. *p < 0.05 versus sham; #p < 0.05 versus CLP.
TG improved regulatory T cells in septic mice
CD4, CD25, and Foxp3 expression was analyzed in splenic lymphocytes via flow cytometry. A representative sample is shown in Figure 4. There was an increase in the percentage of CD4+CD25+ and CD4+Foxp3+ T cells in septic mice. In addition, the mean fluorescence intensity (MFI) of CD4+Foxp3+ T cells was increased in the septic mice. These results indicated that the phenotypic expression of regulatory T cells was increased in sepsis. Interestingly, the percentage of CD4+CD25+ and CD4+Foxp3+ T cells was increased further in septic mice following TG administration. Furthermore, the MFI of CD4+Foxp3+ T cells in the TG 5 mg and TG 10 mg groups was increased when compared with the CLP group.

TG enhanced the phenotypic expression of Tregs in septic mice. (a) BalB/c mice were randomly divided into five groups. Mice were sacrificed on post-operation day 3, and their spleens were harvested. CD4, CD25 and Foxp3 expression was analyzed in splenic lymphocytes. CD4+ T cells gated by R2 were isolated from lymphocytes (LYM) and divided into CD4+Foxp3+ (R4). In addition, CD4+CD25+ T cells gated by R3 were isolated from LYM. CD4+CD25+ and CD4+Foxp3+ T cells are displayed via histogram. (b, c) The percentage of CD4+CD25+ T cells in the LYM cell subpopulation was analyzed. A representative sample is shown. The percentage of CD4+CD25+ T cells was increased in septic mice (*p < 0.05 vs Sham-treated mice; n = 8 mice per group). In the TG 5 mg and TG 10 mg groups, the percentage of CD4+CD25+ Tregs was increased when compared with the CLP group (#p < 0.05). (d) Histogram overlay showing the expression of CD4+CD25+ T cells versus isotype control. (e, f) The percentage of CD4+Foxp3+ T cells in the CD4+ T cell subpopulation was analyzed. A representative sample is shown. The percentage of CD4+Foxp3+ T cells was increased in septic mice (*p < 0.05 vs Sham-treated mice). In the TG 5 mg and TG 10 mg groups, the percentage of CD4+Foxp3+ T cells was increased when compared with the CLP group (#p < 0.05). (G) Histogram overlay showing the expression of CD4+Foxp3+ T cells versus isotype control. (H) The mean fluorescence intensity (MFI) of CD4+Foxp3+ T cells was analyzed via flow cytometry. The MFI of CD4+Foxp3+ T cells was increased in septic mice (*p < 0.05 vs Sham-treated mice). In the TG 5 mg and TG 10 mg groups, the MFI of CD4+Foxp3+ T cells was increased when compared with the CLP group (#p < 0.05).
TG decreased the proliferation activity of splenic CD4+ T cells
Since TG improved regulatory T cells in septic mice, we examined proliferation activity of splenic CD4+ T cells. As shown in Figure 5. The proliferation level in the CLP group was decreased compared with the sham group. Treatment with TG (5 mg and 10 mg) could further decreased the proliferation levels of splenic CD4+ T cells in sepsis.

TG decreased the proliferation activity of splenic CD4+ T cells. After different treatment, the splenic CD4+ cells were separated. The proliferation activity of splenic CD4+ T cells was measured using CCK-8 assay. The proliferation level in the CLP group was decreased compared with the sham group (*p < 0.05 vs sham group). Administration of TG (5 mg and 10 mg) could further decreased the proliferation levels of splenic CD4+ T cells in sepsis. (#p < 0.05 vs CLP group).
TG increased the expression of anti-inflammatory cytokines in septic mice
As shown in Figure 6, The anti-inflammation cytokine IL-10 protein concentrations in the CLP group were notably higher than those of the sham group (p < 0.05). The treatment with TG could further increase the serum IL-10 levels.

TG increased serum levels of IL-10 in septic mice. The levels of IL-10 was assessed 1, 3, and 5 days post-CLP treatment by using specific ELISA. The levels of IL-10 were significantly higher in the CLP groups on days 1, 3, and 5 compared to the sham group (p < 0.05). The treatment with TG could further increase the levels of IL-10 compared to the CLP group (p < 0.05).
TG inhibited the concentration of serum proinflammatory cytokines in septic mice
The concentration of serum TNF-α and IL-6 was increased in the CLP group when compared with the Sham group; TNF-α and IL-6 concentrations peaked on CLP treatment day 1, followed by a gradual decrease on days 2–5 (Figure 7). Treatment with TG significantly decreased the concentration of serum TNF-α and IL-6 in mice, indicating an anti-inflammatory effect of TG in sepsis.

The effects of TG on the serum levels of TNF-a and IL-6 during sepsis. Blood samples were taken at the indicated time points. ELISA was applied to detect the serum levels of TNF-α and IL-6. The results showed that in the CLP group, the levels of TNF-α and IL-6 significantly increased compared with those in the sham group (p < 0.05). In the TG group, the levels of TNF-α and IL-6 decreased compared with those in the CLP group (p < 0.05).
TG inhibited the NF-κb signal pathway
To confirm whether or not NF-κB was involved in the anti-inflammatory effects of TG in sepsis, Western blot was performed to evaluate the expression of IκB-α in the cytosol and NF-κB p65 in the nucleus of lymphocyte. As shown in Figure 8, CLP significantly induced the down-regulation of IκB-α in the cytosol and up-regulation of NF-κB p65 in the nucleus of lymphocyte. However, TG treatment reversed the changes in these two proteins. These results indicated that TG inhibited sepsis induced inflammatory cytokine production and NF-κB activation.

TG inhibited NF-κB activation in septic mice. Nuclear p-NF-κB and Cytoplasmic IkB-a was detected by western blot. Quantitative expressions of IkB-a normalized against actin and NF-kB p65 normalized against lamin B. Data were expressed as means ± SD from three independent experiments.*p < 0.05 versus sham groups. #p < 0.05 versus TG group.
Discussion
Sepsis, a condition characterized by dysregulated inflammation and immunity, has been a critical problem in the intensive care unit for decades.15,16 Numerous therapeutic strategies aimed at reducing mortality in these patients have been developed; however, they have not had significant impact. The pathogenesis and pathophysiology of sepsis are very complicated. The proinflammatory response can drive early mortality in the first days of sepsis, while a compensatory anti-inflammatory response can induce organ failure, immune suppression, and mortality weeks later. 17 However, new insights from genomic analyzes of tissue samples from septic patients have confirmed a persistent and simultaneous inflammatory and anti-inflammatory state, which is driven by the dysfunction of the innate and adaptive immunity. This ultimately culminates inpersistent organ injury and death.18–20 Several studies have shown that T cell exhaustion and higher percentages of regulatory T cells (Tregs) are vital for sepsis-induced immunosuppression.21–23 In the present study, we performed a CLP-induced polymicrobial sepsis model. In accordance with previous studies,24,25 we observed elevated proinflammatory factors and tissue damage in sepsis mice. Moreover, the percentage of regulatory T cells was increased and immunosuppressive function was enhanced in septic mice.
TG has been used to treat inflammatory conditions in China, such as rheumatoid arthritis, various skin disorders, chronic nephritis and ankylosing spondylitis. Dextran sulfate sodium-induced IL-6 expression is inhibited by triptolide, the main extract from Tripterygium wilfordii glycosides, in ulcerative colitis. 26 Xiao and his colleagues have revealed that treatment with Tripterygium wilfordii Polycoride is anti-inflammatory in ulcerative colitis via a reduction in proinflammatory factor expression in the NOXs-ROS-NLRP3 signaling pathway. 27 Another study has found that TG inhibites the inflammatory mediators in interleukin-1β-stimulated cells. 28 Furthermore, TG could markedly reduces serum IL-6, IL-8, and TNF-α in type II collagen-induced arthritis rats. 29 TG upregulates the anti-inflammatory cytokine, IL-37, via ERK1/2 and p38 MAPK signaling pathways. 30 The results of our study, confirmed the anti-inflammatory effects of TG; we found significant inhibition of the NF-B signaling pathway and serum TNF-α and IL-6 in septic mice treated with TG.
Recently, the relationship between TG and Tregs has attracted attention. TG treatment improves the signs and symptoms of patients with ankylosing spondylitis, possibly via the upregulation of CD4+CD25+CD127low Tregs and the downregulation of IL-17 in the peripheral blood. 31 In addition, TG upregulates the expression of IL-10 and Foxp3, and downregulates the expression of TNF-α in rheumatoid arthritis. 32 In this previous study, the authors hypothesized that the CD4+CD25− T cells can be converted into Treg. In line with their study, we have shown that TG enhanced the suppressive function of Tregs. TG also decreased the proliferation activity of splenic CD4+ T cells, two possible reasons are that TG can enhance immunosuppressive function of Treg and TG has toxic effect. The NF-κB transcription factor family, which are inducible regulators of innate and adaptive immunity, 33 are associated in Treg development. 34 Therefore, we speculate that the enhanced suppressive function of Tregs is associated with the NF-κB signaling pathway.
Due to its anti-inflammatory and immunoregulatory effects, tripterygium glycoside is used in the clinical treatment of rheumatoid arthritis, restenosis following endovascular, Graves ophthalmopathy and so on.35–37 At the same time, tripterygium glycoside has many side effects included leucopenia, erythroblastopenia, granulocytopenia, nausea and so on. Therefore, tripterygium glycoside is not widely used in the clinical treatment of sepsis. Although there are a few studies on animal models, there are still many problems to be solved before clinical application.
Conclusion
In conclusion, the findings of the present study indicated that TG is protective against sepsis. The underlying mechanism of this effect may be via the upregulation of CD4+CD25+ Treg expression. These cells promote the expression of IL-10 and Foxp3, and inhibit the NF-κB signal pathway and the secretion of serum TNF-α and IL-6. Our results indicated that administration of TG is a possible therapy for the abrogation of sepsis development. Further studies are required to fully elucidate the precise mechanisms of this beneficial effect.
Footnotes
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation. All animal experiments have been followed with the ARRIVE guidelines and been carried out in accordance with the U.K. Animals (Scientific Procedures) Act, 1986 and associated guidelines, EU Directive 2010/63/EU for animal experiments, or the National Institutes of Health guide for the care and use of Laboratory animals (NIH Publications No. 8023, revised 1978).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Writing assistance
Ethics approval
Ethical approval for this study was obtained from Laboratory Animal Ethics Committee of Wenzhou Medical University (ID:WZDY20170524).
