Abstract
The purpose of the study was to evaluate the effect of
Introduction
Intravenous thrombolysis with recombinant tissue plasminogen activator (rt-PA) is the gold standard treatment for acute cerebral stroke within the first 4.5 h.1,2 However, the treatment can lead to ischemia-reperfusion injury (R/I), which includes the blood–brain barrier (BBB) destruction, cerebral edema, and hemorrhagic transformation (HT). 3 The mechanisms of ischemia-R/I are complex, which include excitatory amino acids toxicity, calcium overload, inflammatory reaction, oxidative damage, and apoptosis.4–6 Among the above-mentioned mechanisms, inflammation is a major contributing factor of R/I. Recent studies showed that regulatory T cells (Tregs) play an important role in reducing inflammation, mitigating the permeability of BBB, and decreasing the cerebral lesion volume in the treatment of R/I after acute ischemic stroke. 7 The therapy that combines Tregs with rt-PA can ameliorate inflammation after reperfusion treatment in stroke rats model. 6 Hence, Tregs can be a promising treatment for ischemia-R/I.
In Chinese medicine, the main pathology of R/I after thrombolysis is qi deficiency and blood stasis.
8
Therefore, the main treatment focuses on synergizing qi and promoting blood circulation, which can be obtained by administrating the widely used Chinese medicines
Methods
Animals
The animal experiments were performed according to the procedures formulated by the Institutional Animal Care and Use Committee (IACUC) of Guangdong Provincial Hospital of Chinese Medicine, Guangzhou, China. This procedure was in accordance with the guidelines of European Community (EEC Directive in 1986; 86/609/EEC). Adult male Wistar rats (provided by Medical Laboratory Animal Center of Guangdong, Guangzhou, China) weighing 250–300 g and aged 3–4 months were maintained in the animal room (SPF, Animal Experimental Center, Guangdong Academy of Chinese Medicine, Guangzhou, China) at 22 ± 2°C and 55 ± 2% relative humidity on a 12-h light/dark cycle with free access to food and water.
According to Overgaard et al. 13 and Zhang et al., 14 the autologous embolus was prepared as follows: first, 20 μL of blood was withdrawn from the femoral artery and injected into a 20-cm PE-50 tube. Second, 1U thrombin was injected into the PE-50 tube. After 15 min, a 30- to 35-mm (1.0 μL) autologous clot was formed. Third, the clot was withdrawn into a Hamilton syringe. Finally, an embolus with 0.5 cm of length was ready for injection.
The MCAO models were made as follows: first, the animals were anesthetized with 0.35 mL/100 g 10% Chloral Hydrate by intraperitoneal injection. Second, the rat model was set in a supine position, followed by the sanitation of the skin. Afterward, an incision was made in the middle of the neck under an operating microscope. Third, the right common carotid artery (CCA), internal carotid artery (ICA), and external carotid artery (ECA) were exposed by blunt dissection. Fourth, a 15-mm long autologous embolus was carefully injected in a rapid manner into the ICA lumen. In the end, the skin was sutured to close the wound and the model was completed. The sham surgery was conducted in similar manner as the method described above, but without the injection of autologous embolus.
Experimental design
The animals were randomly divided into six groups, namely, sham (n = 72), MCAO (n = 72), thrombolysis (n = 72),

(a) The chemical structure of ligustrazine and (b) the experimental design. The pharmaceuticals were intraperitoneally injected immediately after ischemic stroke. rt-PA was intravenously injected for thrombolysis 3 h after ischemic stroke. Neurological assessment and TTC staining were conducted at 3, 6, and 24 h after thrombolysis induction. ELISA, qRT-PCR, and Western blot assays were conducted at 3 and 24 h after thrombolysis induction.
According to previous studies, saline was added into the

(a) TTC staining: the brain samples were subjected to TTC staining. (b) Histogram showing the infarct volumes of the sham, MCAO, thrombolysis,
Neurological assessment
We measure the neurological function of the rats at 3, 6, and 24 h after the induction of cerebral ischemia according to modified Bederson’s examination system as previously reported. 15 There are five levels of the neurologic examination grading system in which the scores are as follows: 0 indicates no obvious neurological deficit, 1 indicates disabled forelimb flexion, 2 indicates decreased resistance of disabled forelimb when pulling tail, 3 indicates moving to all directions spontaneously but circling to the disabled side when pulling tail, and 4 indicates circling to the disabled side spontaneously.
TTC staining
The TTC staining to measure the ischemic volumes was conducted as follows: (1) the brains from models were sliced into 2-mm slices using a rat-brain matrix (RWD, Life Science); (2) the sample slice was incubated at 37°C for 20 min in 2% 2, 3, 5-TTC solution (Sigma-Aldrich); (3) the sample sections were photographed and analyzed by a double-blind collaborator using the ImageJ software (NIH Program, Bethesda, MD, USA); and (4) as according to previous studies, 16 the ischemic volumes were calculated by summing the thicknesses of the brain slices multiplied by the infarction areas across different brain slices.
ELISA analysis
The levels of transforming growth factor beta 1 (TGF-β1), interleukin-10 (IL-10), interferon gamma (IFN-γ), and IL-17 in serum were assessed using ELISA kit. The peripheral blood was collected and centrifuged at 3000 r/min to prepare the serum. Further, the serums were processed according to the manufacturer’s instructions of the ELISA kit. At last, the prepared serums were assayed using a microplate reader (BIO-RAD, America). Each serum sample was tested three times to get the mean value.
Quantitative real-time PCR
The expression levels of Foxp3, TGF-β1, IL-10, IL-4, IL-lβ, IFN-γ, and IL-17 mRNA were assessed using TaqMan real-time PCR. The total RNA of the brain samples was isolated using Trizol reagents (Invitrogen). cDNA for the amplified templates was synthesized from the total RNA by means of a PrimeScript RT reagent kit (TaKaRa Bio; Shiga, Japan). The primers designed using the Premier 5.0 biological software and synthesized by Sangon Biotechnology Company (Shanghai, China) are shown in Table 1. RT-PCR was performed in a model 7300 sequence detector (PE Applied Biosystems; Chiba, Japan) using an SYBR Premix EX Taq II Kit (TaKaRa Bio; Shiga, Japan). The experiments were repeated twice.
The primers of RNA used in this study.

Western blot analysis
The brain samples were lysed in high-efficiency RIPA (radioimmunoprecipitation assay) lysate that contains 20 mmol/L Tris (pH 7.6), 0.2% SDS (sodium dodecyl sulfate), 1% deoxycholate, 1% Triton X-100, 0.11 IU (international units)/mL aprotinin, and 1 mmol/L phenylmethylsulphonyl fluoride (all purchased from Sigma-Aldrich). The protein samples, 5 µg each lane, were subjected to 8% SDS-PAGE (sodium dodecyl sulfate–polyacrylamide gel electrophoresis) and subjected to electrophoresis at 120 V. Subsequently, the isolated proteins were transferred to PDNF (parasite-derived neurotrophic factor) membrane in 300 mA. The following antibodies were used: Foxp3 (1:1000 dilution; Millipore), TGF-β1 (1:500 dilution; Abcam), IL-10 (1:1000 dilution; Abcam), and β-actin (1:6000 dilution; Sigma-Aldrich). The Western blots were quantified with ImageJ software (NIH Program, Bethesda, MD, USA). The experiments were conducted three times.
Flow cytometry
To prepare spleen cell suspensions, excised spleen with the size of 8 × 4 mm were cut, grind, and sieved into single-cell suspensions. After repeated suspending and washing, the cells were surface stained with fluorescein isothiocyanate (FITC)-labeled antibodies against rat CD4 together with allophycocyanin-labeled antibodies against rat CD25 at room temperature for 45 min. Then, the cells were washed, fixed, permeabilized with buffer (Cytofix/Cytoperm; eBioscience), and intracellularly stained with phycoerythrin (PE)-labeled antibodies against Foxp3 (eBioscience). The samples were examined using a flow cytometer (ACEA NovoCyte TM D2060R, ACEA Biosciences, USA) and analyzed using a software (CellQuest, Becton Dickinson, USA).
Statistical analyses
The data are presented as the means ± the standard deviation. The multiple-group comparisons were performed by one-way analyses of variance (ANOVAs) followed by Student–Newman–Keuls’ or Dunnett’s post hoc tests. The internal-group comparisons were performed by T tests. The bars in the icon indicate the means + the SEMs. The statistical analyses were conducted with SPSS software (21.0, Chicago, IL, USA). A
Result
The infarct volumes of each group
The result of TTC staining shows that the ischemic volumes in the MCAO, thrombolysis, ligustrazine,
Neurological assessment
To determine the neuroprotective effect of the combination therapy, we examined the neurological function for each group at 3, 6, and 24 h after thrombolysis, as shown in Figure 3. Observation at 3 and 6 h after thrombolysis showed no significant difference in neurological function between sham group (0,0), thrombolysis group (3.33 ± 0.52, 3.50 ± 0.55), MCAO group (3.45 ± 0.57, 3.56 ± 0.63),

Neurological deficit assessment among groups.
Combination drug treatment ameliorated inflammation response after thrombolysis
To explore the effect on ameliorating inflammatory response of the combination therapy, we examined the cytokine for each group at 3 and 24 h after thrombolysis, as shown in Figures 5–7. There were no significant difference in gene expression of TGF-β1, IL-10, IL-4, IL-1β, IFN-γ, and IL-17 between groups (
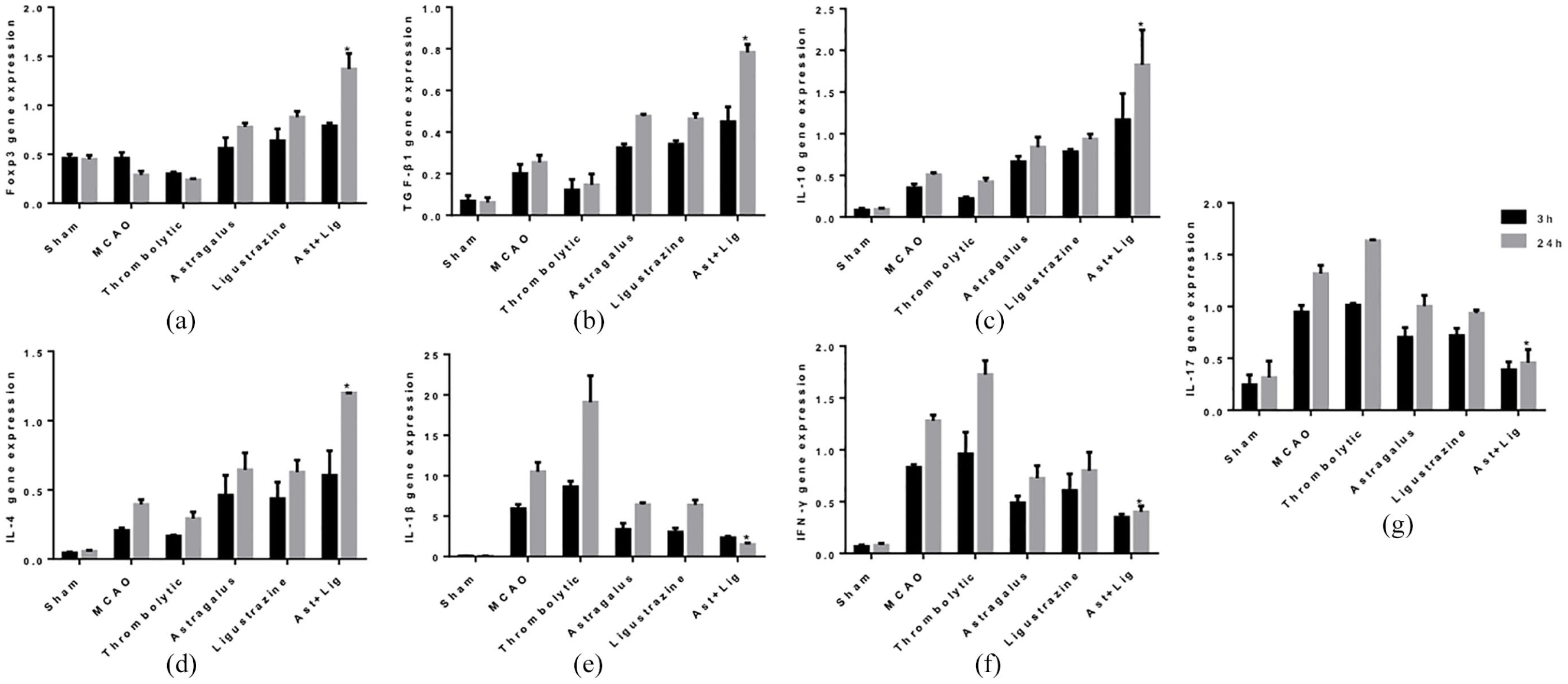
Gene expression assay of Foxp3, TGF-β1, IL-10, IL-4, IL-1β, INF-γ, IL-17 by RT-PCR. The histograms indicate the (a) Foxp3 gene expressions, (b) TGF-β1 gene expressions, (c) IL-10 gene expressions, (d) IL-4 gene expressions, (e) IL-1β gene expressions, (f) INF-γ gene expressions, (g) IL-17 gene expressions in the sham, MCAO, thrombolysis,
The Western blot result can be seen in Figure 5. There were no significant differences in TGF-β1 and IL-10 protein level between sham groups (
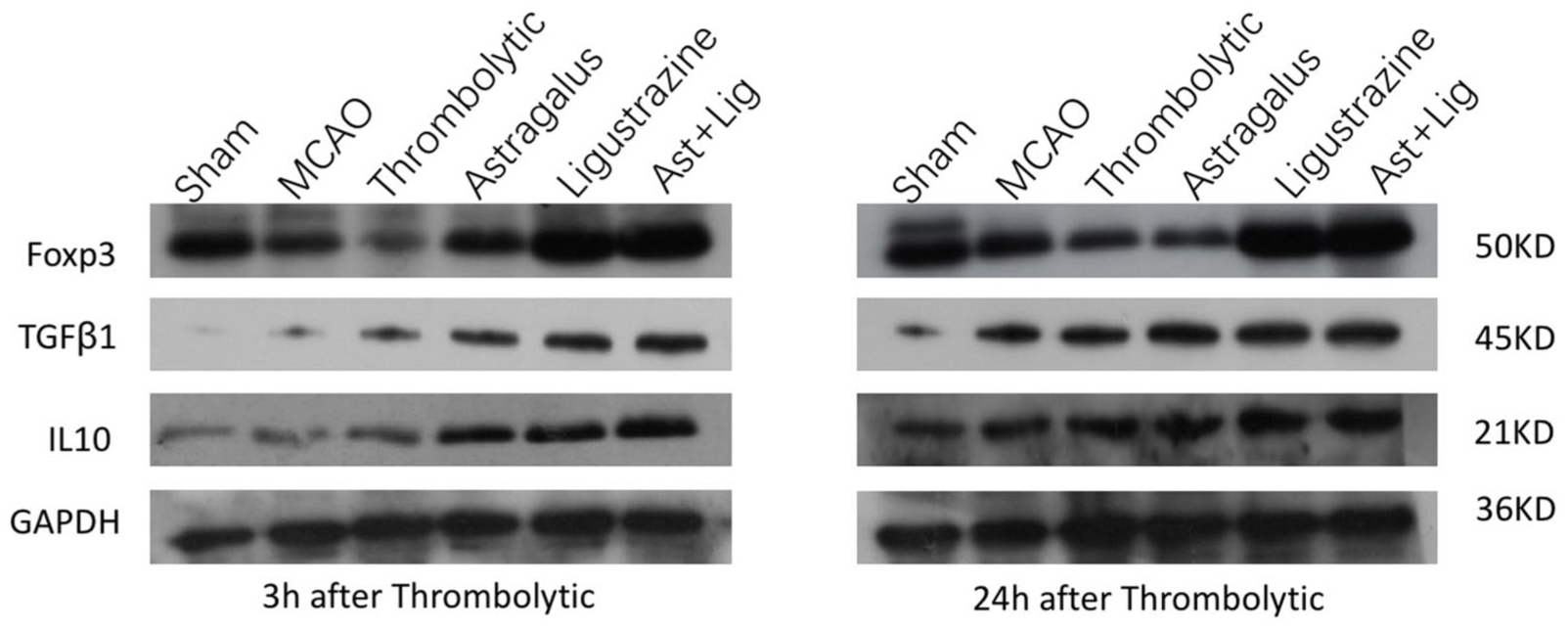
Western blot assay among groups: sham group, MCAO group, thrombolysis group,
The ELISA result can be seen in Figures 6 and 7. There were no significant differences in protein level of TGF-β1, IL-1β, IFN-γ, and IL-17 between sham groups (

(a) ELISA measurement of the TGF-β1 serum content level and (b) IL-1β serum concentration (n = 6 at each time point).

(a) ELISA assay of the IFN-γ serum concentration and (b) IL-17 serum concentration (n = 6 at each time point).
These results proved that the
Combination drug treatment upregulated Treg cells level in spleen after thrombolysis
To explore the effect on Treg cells of the combination therapy, we examined the Tregs level in the spleen for each group at 3 and 24 h after thrombolysis, as shown in Figure 8. The flow cytometry results showed that there was no significant difference in Tregs level between sham groups (
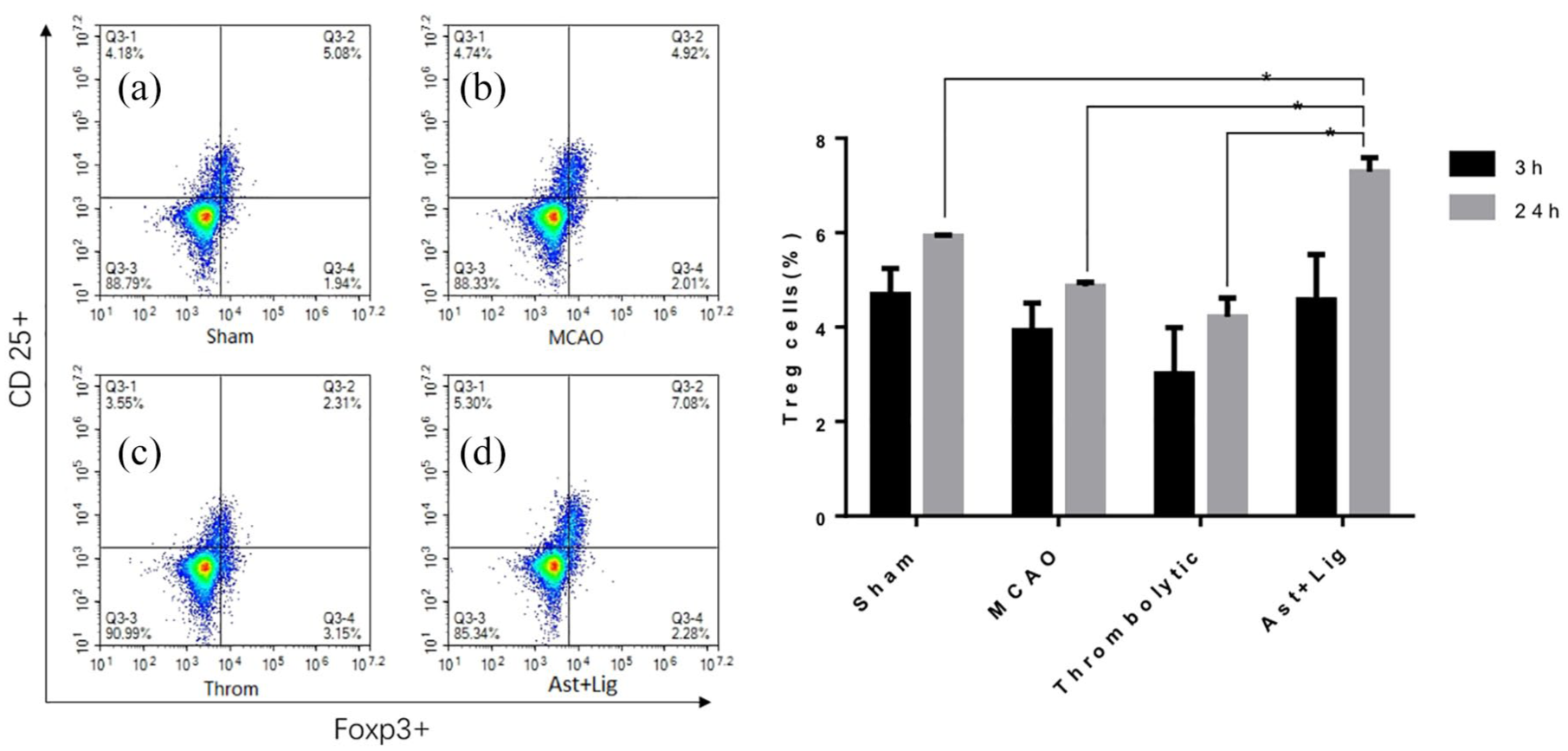
Treg cells measurement: the flow cytometry indicates the Tregs level of the spleen in the (a) sham, (b) MCAO, (c) thrombolysis, (d) Ast + Lig groups (n = 6 at each time point).
Discussion
R/I treatment with rt-PA in acute ischemic stroke patients can induce inflammation reaction which may result in the increased risk of BBB damage and hemorrhage transformation. In order to obtain safer application of rt-PA treatment, it is necessary to explore effective therapies to decrease these complications. Recent studies had proved that endogenous Tregs could protect against ischemic brain injury. 17 In addition, the administration of Tregs together with rt-PA thrombolysis could reduce cerebral hemorrhages in ischemic stroke animals. Tregs also have neuroprotective effect and alleviating BBB damage in models of cerebral R/I. 18 Therefore, upregulating endogenous Tregs would be a promising method for the treatment of R/I.
Previous studies have found that
As shown by the results, the rt-PA thrombolysis treatment may not obviously improve neurological function effectively within 24 h of intervention. The rt-PA thrombolysis treatment supplemented with
Next, we explored the correlation of the neuroprotective effects of the combination therapy with its anti-inflammation property. In the inflammatory-related cells and cytokines assay, pro-inflammatory cytokines IL-17, IFN-γ and IL-1β in the damaged brain clearly increased after rt-PA thrombolysis treatment. At the same time, the Tregs in the spleen and the related cytokines (TGF-β, IL-10) were markedly decreased. However, after the intervention of Ast + Lig combination therapy, Tregs and its related cytokines (TGF-β, IL-10) were significantly increased, meanwhile the IL-17, IFN-γ, and IL-1β significantly decreased. This combination therapy also increased the expression of Foxp3, the main transcription factor of Tregs, and maintained the Tregs immune tolerance function. These results are similar to the study of Arthur et al.,
25
who had found that the expression levels of pro-inflammatory cytokines tumor necrosis factor alpha (TNF-α), IL-1β and IFN-γ were reduced after Tregs depletion in the ischemic brain. Therefore, we could infer that the
The neuroprotective and anti-inflammatory effect mechanism of the combination therapy lies in two aspects. On one hand, previous study has proved that Tregs can protect rt-PA-mediated BBB damage in stroke mice.
7
Our previous study also found that the
There are some limitations of this study. We have conducted a parallel observation of the
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
The scheme was authorized by Ethics Committee of the 2nd affiliated Hospital, Guangzhou University of Chinese Medicine.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the National Natural Science Foundation of China (No. 81804038to R.P. and No. 81873262 to Y.H.), China Postdoctoral Science Foundation (No. A2-BBK429152K07 to R.P.), the Natural Science Foundation of Guangdong Province (No. 2014A030310457 to R.P.), the Project of Science and Technology Bureau of Guangdong Province (No. 2016A020226030 to R.P.), and the Special Research Foundation of Science and Technology for Traditional Chinese Medicine from Guangdong Hospital of Chinese Medicine (No. YN2016QJ17 to R.P.).
