Abstract
Several case-control studies have been performed in different populations to uncover the association between interleukin-1 beta gene, interleukin-1 receptor antagonist gene polymorphisms and urolithiasis. Here we decided to perform a literature review and meta-analysis to further estimate it. A systematic search was conducted in PubMed, Embase, Cochrane, clinicaltrials.gov, CNKI databases. To pool the effect size, odds ratios and 95% confidence intervals were used. Finally, five articles were included. Our results of literature review suggested that IL1RN IVS2 VNTR might be associated with the risk of urolithiasis. However, the results of meta-analysis suggested that IL-1beta -511C>T, IL-1beta +3954C>T, and IL1RN IVS2 VNTR might not be associated with the risk of urolithiasis. There were not enough data to fully confirm this association and the results should be interpreted with caution.
Introduction
Urolithiasis is a multi-factor urological disorder. It is the third most common urologic disease. The major factors of stone formation is urinary tract anomalies and metabolic abnormalities. Urinary tract anomalies include urinary tract malformation and urinary tract infection. Infection can induce deposition of calcium phosphate, prompt the urine salt crystal growth, and promote the stone formation. 1
As a potent proinflammatory cytokine, interleukin-1 (IL-1) is closely related to the inflammation caused by infection. IL-1 can regulate the expression of cell adhesion molecules, immunomodulatory molecules and inflammatory mediators. 2 IL-1 includes two different types. As an autocrine messenger, IL-1α is mainly expressed in cell or on cell surface. IL-1beta is usually released by cells, and exert its effects by acting on other cells. 3 By localizing bone marrow macrophages to inflammatory site and stimulating the differentiation of macrophages into activated osteoclasts, IL-1beta can induce formation of osteoclasts, bone resorption and hypercalciuria.4,5 Interleukin-1 receptor antagonist (IL-1RN), which can competitively occupy the IL-1 receptor on the cell surface and reduce inflammation, is the natural antagonist of IL-1. 3
Proteins encoded by IL-1 gene family include IL-1α, IL-1beta, IL-1 receptor and IL-1RN. IL-1 gene family are clustered on human chromosome 2q14 and are known to be polymorphic and in linkage disequilibrium. 6 It was found that IL-1beta has high expression in calcium oxalate SD rats model. 7 And higher IL-1beta level, lower IL-1RN level was associated with significantly higher risk of urolithiasis. 8 IL1-RN might change the intracellular signaling pathways and have association with adult urolithiasis. 4 IL-1beta and IL-1RN play complex opposing roles in inflammatory response. Thus polymorphisms of IL-1beta and IL-1RN might influence the stone formation. 9
Several case-control studies have been performed in different populations to uncover the association between IL-1 gene, IL-1RN gene polymorphisms and urolithiasis. Some of those studies showed that IL-1 gene, IL-1RN gene polymorphisms can increase the risk of urolithiasis. However, those studies had relatively small sample sizes and inconsistent conclusions, thus no certain conclusion can be drawn. Here we decided to perform a literature review and meta-analysis to further estimate it.
Materials and methods
Identification of eligible studies
Two independent investigators carried out systematic search separately in PubMed, Embase, Cochrane Library, clinicaltrials.gov, CNKI (China National Knowledge Infrastructure) databases. The following terms were used: “IL1” OR “IL 1” OR “IL-1” OR “Interleukin1” OR “Interleukin 1” OR “Interleukin-1” OR “IL-1β” OR “IL 1β” OR “IL1β” OR “IL-1beta” OR “IL 1beta” OR “IL1beta” OR “Interleukin-1β” OR “Interleukin 1β” OR “Interleukin1β” OR “Interleukin-1beta” OR “Interleukin 1beta” OR “Interleukin1beta” OR “IL-1α” OR “IL 1α” OR “IL1α” OR “IL-1alpha” OR “IL 1alpha” OR “IL1alpha” OR “Interleukin-1α” OR “Interleukin 1α” OR “Interleukin1α” OR “Interleukin-1alpha” OR “Interleukin 1alpha” OR “Interleukin1alpha” OR “IL-1RN” OR “IL 1RN” OR “IL1RN” OR “Interleukin-1RN” OR “Interleukin 1RN” OR “Interleukin1RN” AND “stone OR calculus OR calculi OR lithiasis OR Nephrolithiasis OR urolithiasis” AND “polymorphisms OR polymorphism,” without any limitation applied. The last search update was performed on July 6, 2020. We also manually searched references of related studies and reviews.
Inclusion and exclusion criteria
Studies selection must meet the following inclusion criteria: (1) evaluation of the association between IL-1 gene, IL-1RN gene polymorphisms and urolithiasis; (2) case-control study; (3) studies focusing on tissues of human beings. Exclusion criteria: (1) duplication of previous publications (When there were multiple publications from the same population, only the largest study was included); (2) comment, review and editorial; (3) study without detailed genotype data; (4) GWAS; (5) studies focusing on cell lines. Dissertation thesis were included in the analysis. We took experience and inspiration from the article we have published recently, 10 in which the inclusion and exclusion criteria was mature and rigorous.
Data extraction, methodological quality assessment, and statistics analysis
Data about the chemical composition of urinary calculi were extracted. The most important factor of Newcastle-Ottawa Scale (NOS) 11 in this meta-analysis was “age, gender and country.” Others were performed according to methods in our published article Qin et al. 10 We performed the analysis with the PRISMA checklists. 12 Evaluation of Hardy-Weinberg equilibrium (HWE), OR and 95% CIs, heterogeneity, sensitivity and publication bias were performed according to methods in our published article Qin et al. 10
Results
Characteristics of studies
A total of 65 articles were acquired from databases (PubMed = 22, Embase = 43, Cochrane = 0, clinicaltrials.gov = 0, CNKI = 0, other sources (from manually search) = 0). The selection process was shown in Figure 1. 37 records (34 records were not about IL-1, IL1RN and urolithiasis; three records were reviews without detailed genotype data) were excluded because their title and abstract could not meet the inclusion and exclusion criteria. One full-text articles were excluded because it is not case-control study. 13 Finally, five articles4,8,9,14,15 were included. In these five articles, two SNP of IL-1beta (IL-1beta −511C>T, IL-1beta +3954C>T) and one intervening sequence of IL1RN (IL1RN IVS2 VNTR) was studied. The characteristics of each study were shown in Table 1.

Flow chart of study selection.
Characteristics of studies included.
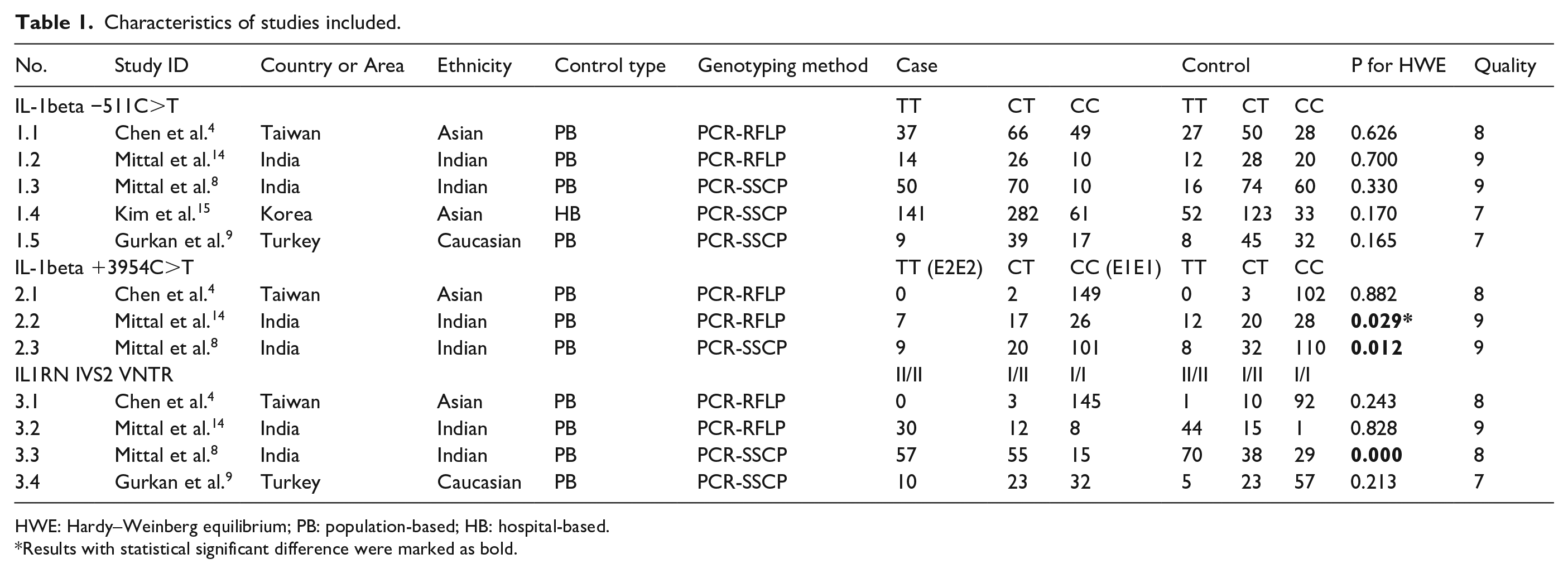
HWE: Hardy–Weinberg equilibrium; PB: population-based; HB: hospital-based.
Results with statistical significant difference were marked as bold.
In Chen et al.’s study, 4 152 patients (117 males and 35 females) aged between 23 and 76 years with recurrent calcium oxalate stone were included, and patients who showed symptoms of urinary tract infections during the period of stone treatment were excluded. In Mittal et al.’s study, 14 50 consecutive pediatric patients (age range = 2–14 years) with renal stones were included, and patients who had history of bowel disease, renal tubular acidosis and urinary tract anomalies were excluded. In Mittal et al.’s study, 8 130 patients (mean age 40.0 ± 11.5 years) were enrolled, and exclude any person with renal calcification and urinary-tract infection. In Gurkan et al.’s study, 9 80 patients (53 male and 27 female) aged between 25 and 61 (with an average of 42.9 ± 10.2 years) with recurrent stone disease were enrolled regardless of their family history, and who showed symptoms of urinary tract infections, pregnancy, vascular heart disease, acute or chronic infectious, immunologic conditions, history of malignancies, neoplastic, coagulation disorders or chronic renal failure during the period of stone treatment and the hypercalcemia, hyperuricemia, hyperoxaluria or hyperuricosuria were excluded. Kim et al.’s study 15 was lack of detailed data of the patients.
Paired control groups in the five articles consisted of people who had no history of stone disease. Different genotyping methods were utilized including PCR–RFLP and PCR-SSCP. Blood samples were used for genotyping in all studies. The control group of study No. 2.2, 2.3, and 3.3 had shown significant departure from HWE.
Overall analyses
Table 2 listed results of each genetic model. No statistically significant changes of urolithiasis risk was found in either genetic model of IL-1beta −511C>T (Figure 2), IL-1beta +3954C>T or IL1RN IVS2 VNTR.
Summary of pooled ORs in the meta-analysis.

OR: Odds ratio; CI: confidence interval.
Results with statistical significant difference were marked as bold. Unstable results in sensitivity analyses were marked as italic. Less than three studies were included in model (TT vs CC) and (TT vs CC + CT) of IL-1beta +3954C>T, so that sensitivity analyses could not be performed.

Forest plot with a random effects model for the association between IL-1beta –511C>T and urolithiasis in allelic comparison (T vs C). For each study, the estimate of OR and its 95% CI is plotted with a box and a horizontal line. Rhombus: pooled OR and its 95% CI.
Sensitivity analyses
In any comparison including more than two studies, sensitivity analyses were performed. Less than three studies were included in model (TT vs CC) and (TT vs CC+CT) of IL-1beta +3954C>T, so that sensitivity analyses could not be performed. In model (CT vs CC) and (CT+TT vs CC) of IL-1beta −511C>T, when study No. 1.1 was excluded, statistically different results were obtained. Other results showed stability in sensitivity analyses (Table 2, Supplemental Data).
Publication bias
Symmetry of funnel plot, p value of Begg’s test (pB) and p value of Egger’s test (pE) were evaluated in each genetic model of IL-1beta −511C>T and no significant publication bias was found. Owing to the scanty number of available studies in IL-1beta +3954C>T and IL1RN IVS2 VNTR, publication bias analyses could not be performed (Supplemental Data).
Discussion
In model (T vs C), (TT vs CC) and (TT vs CC+CT) of IL-1beta −511C>T, we found no associated with risk of urolithiasis, and the results showed stability in sensitivity analyses and no publication bias. No associated was found in model (CT vs CC) or (CT+TT vs CC) of IL-1beta −511C>T, either. However, the results lacked stability when study No. 1.1 was excluded.
In five models of IL-1beta +3954C>T, we found no associated with risk of urolithiasis either. However, publication bias analyses were not performed in five models and sensitivity analyses could not be done in model (TT vs CC) and (TT vs CC+CT).
No association with different risk of urolithiasis was found in five models of IL1RN IVS2 VNTR and the results showed stability in sensitivity analyses, however, publication bias analyses could not be done.
In Chen et al.’s study, 4 no significant difference in allele frequencies in IL-1beta −511C>T (p = 0.627) or IL-1beta +3954C>T (p = 0.403) between cases and controls was found by using Fisher’s exact test, but significant different result was found in IL1RN IVS2 VNTR (p = 0.005). In Mittal et al.’s study about pediatric cases and controls, 14 significant different result was also found in IL1RN IVS2 VNTR (p = 0.023), but not in IL-1beta -511C>T (p = 0.263) or IL-1beta +3954C>T (p = 0.694). In Mittal RD’s another study about adult cases and controls, 8 significant different result was found in IL1RN IVS2 VNTR (p = 0.033) and IL-1beta −511C>T (p < 0.001), but not in IL-1beta +3954C>T (p = 0.408). In Gurkan et al.’s study, 9 IL1RN IVS2 VNTR and IL-1beta −511C>T (p = 0.293) were studied and only IL1RN IVS2 VNTR showed a significant different result (p = 0.047). In Kim et al.’s study, 15 only IL-1beta −511C>T (p > 0.05) was studied but no significant difference was found.
To sum up, all four studies from three different areas (Taiwan, India, Turkey) pointed out the significant association between IL1RN IVS2 VNTR and urolithiasis, moreover, three studies of them were about adult and one study was about children. In 5 studies about IL-1beta −511C>T, only one study showed a significant different result. No significant difference was found in all three studies about IL-1beta +3954C>T. Our results of literature review suggested that: IL1RN IVS2 VNTR might be associated with the risk of urolithiasis. However, the results of meta-analysis suggested that IL-1beta −511C>T, IL-1beta +3954C>T and IL1RN IVS2 VNTR might not be associated with the risk of urolithiasis. There were not enough data to fully confirm this association and the results should be interpreted with caution.
Studies about the specific biological mechanisms of these gene polymorphisms are rare. IL-1beta −511C>T (rs16944) is a SNP located at promoter region of IL-1beta gene. Previous findings suggest that the presence of SNPs within the promoter region of IL-1 genes might regulate the expression levels of IL-1 protein. 16 IL-1beta +3954C>T is another SNP located in exon 5 at −3954, where it is responsible for a C to T transition. The region within the second intron of IL-1 receptor antagonist gene contains variable numbers of a tandem repeat (VNTR) of 86 basepairs and the number of repeats influences gene transcription and protein production. 8
To date, the number of available studies which can be included were small. All studies about IL-1beta −511C>T included only 881 cases/608 controls, pooled. IL-1beta +3954C>T 331 cases/315 controls. IL1RN IVS2 VNTR 390 cases/385 controls. Ethnicity of studies were not all the same. Human immune system is powerful and complex. Many unknown mechanisms need to uncover. In the meta-analysis, we could not perform the subgroup analyses owing to limited data. Publication bias analyses or sensitivity analyses could not be done in some groups. Study No. 2.2, 2.3, and 3.3 had shown significant departure from HWE. Ethnicity of studies were not all the same. Well-designed studies with larger sample size and more subgroups are required. Related studies published in other languages or unpublished were possibly missed. With limitations, we tried to provide some insights on the potential association between IL-1beta −511C>T, IL-1beta +3954C>T, IL1RN IVS2 VNTR and the risk of urolithiasis.
Conclusion
Our results of literature review suggested that: IL1RN IVS2 VNTR might be associated with the risk of urolithiasis. However, the results of meta-analysis suggested that IL-1beta −511C>T, IL-1beta +3954C>T and IL1RN IVS2 VNTR might not be associated with the risk of urolithiasis. There were not enough data to fully confirm this association and the results should be interpreted with caution.
Supplemental Material
PRISMA-2009-checklist-2- – Supplemental material for Association between interleukin-1 beta gene, interleukin-1 receptor antagonist gene polymorphisms and urolithiasis: A literature review and meta-analysis
Supplemental material, PRISMA-2009-checklist-2- for Association between interleukin-1 beta gene, interleukin-1 receptor antagonist gene polymorphisms and urolithiasis: A literature review and meta-analysis by Jiaxuan Qin, Jinchun Xing and Zonglong Cai in European Journal of Inflammation
Supplemental Material
Supplementary_data – Supplemental material for Association between interleukin-1 beta gene, interleukin-1 receptor antagonist gene polymorphisms and urolithiasis: A literature review and meta-analysis
Supplemental material, Supplementary_data for Association between interleukin-1 beta gene, interleukin-1 receptor antagonist gene polymorphisms and urolithiasis: A literature review and meta-analysis by Jiaxuan Qin, Jinchun Xing and Zonglong Cai in European Journal of Inflammation
Footnotes
Acknowledgements
Recently we (Jiaxuan Qin, Jinchun Xing) have published another paper titled “Association between CD40 rs1883832 and immune-related diseases susceptibility: A meta-analysis.” 10 The published paper has no relationship with this manuscript. The published paper and this manuscript used the same statistical method to study two totally different scientific questions. Owing to the strict report specification (we used PRISMA in both study) and the first author's writing habits (Jiaxuan Qin), similarity could be found in several sections between the published paper and this manuscript, which needs to be acknowledged here.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: (1) The Science Fund founded by the First Affiliated Hospital of Xiamen University for Young Scholars (Project Number: XYY2016011; Recipient: Jiaxuan Qin). (2) Youth Research Project of Fujian Provincial Health Commission (Project Number: 2019-2-47; Recipient: Jiaxuan Qin).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
