Abstract
Macrophage migration inhibitory factor (MIF) is a key pro-inflammatory cytokine in immuno-inflammatory diseases. For the first time, we examined the expression of MIF in
Introduction
Groupers are considered as luxury consumption in China, Southeast Asian countries, the Atlantic Ocean, and Mediterranean Sea. A rapid expansion of commercial grouper aquaculture activities were practiced in most parts of world, which resulted in many disease outbreaks.
1
Macrophage migration inhibitory factor (MIF) plays a critical role in several autoimmune inflammatory diseases in mammals, birds, amphibians, and fish.3–5 MIF is the first molecule to get to the inflammation site. MIF measured the degree of cellular inflammation. 6 The mechanism of fish MIF in response to bacterial infection is unknown. MIF has already been considered as a new therapeutic target and a biomarker in the diagnosis and treatment of human multiple septicemia, which prompts us to investigate the role of MIF in immunity against fish vibriosis.
Moreover, in humans, mammals, and zebrafish, MIF protein and mRNA were expressed in many infectious and inflammatory diseases.5,7 To date, MIF genes have been measured in sea lamprey (
MIF can promote the secretion of inflammatory cytokines tumor necrosis factor (TNF)-α, interleukin (IL)-6, and IL-12.3,4 MIF further co-stimulates T and B lymphocytes and upregulates the production of IL-6, interferonc, and TNF-α by a feed-forward, positive feedback loop. TNF-α, in cooperation with IL-1, plays an important role in inflammatory diseases.8,9 To our knowledge, there are no studies on the functions of MIF in groupers. Little is known about the relationship between inflammatory diseases and MIF.
For the first time, the MIF mRNA expression of several tissues in
Materials and methods
V. parahaemolyticus
Fish pathogenicity experiments
Healthy
RNA extraction and reverse transcription
Total RNA was extracted from tissue specimens as described above using TRIzol (Invitrogen, Carlsbad, CA, USA) following the manufacturer’s instruction. First-strand cDNA was synthesized from 2 mg of total RNA using a Revert Aid Mu-MLV cDNA synthesis kit according to the instructions of the manufacturer. The RNA were stored at −80°C.
Real-time PCR
Five groupers from each replicate of each treatment were pooled. RNA was isolated as described above. Real-time PCR analysis was performed using the StepOnePlus™ Real-Time PCR System (ABI, USA). Quantitative PCR analysis was then performed to examine the mRNA level of MIF in the tissues following the manufacturer’s instructions. The reactions were carried out in a total reaction volume of 10 µL. The primers used for PCR was as follows: forward primer: 5’-TMATGCCGATGTTYRTCVGTGARCAC-3’, reverse primer:5’-CTGGAATGATCTTYTDTTTC-3’. The primers for β-actin (used as an internal reference gene) were 5’-CGAGAAATCGTTCGTGAC-3’ (forward) and 5’-TGCTGTTGTAGGTGGTCTCGT-3’ (reverse). The mRNA level of MIF gene was normalized using β-actin. The amplification reactions consisted of 35 cycles. In each case, denaturing, annealing, and extension steps were 20 s at 94°C, 30 s at 58°C, and 30 s at 72°C, respectively. Data analysis was performed using the relative standard curve method. All results shown were representative of at least five groupers. All results shown are representative of at least five groupers.
Quantification of MIF, TNF-α, and IL-1 tissue levels
MIF, TNF-α, and IL-1 levels in the tissues were detected by Enzyme Linked Immunosorbent Assay (ELISA) in duplicates using a commercial kit (Wuhan EIAab Science Co., Ltd., PR China) according to the manufacturer’s instructions. The kits include Epinephelus MIF ELISA Kit (Catalog no. E0698f), Epinephelus TNF-α ELISA Kit (Catalog no. CSB-E13254Fh), and Epinephelus IL- 1 ELISA Kit (Catalog no. CSB-E13259Fh). Commercial antibodies include anti-TNF-α antibody and anti- Epinephelus IL-1 antibody (Wuhan EIAab Science Co., Ltd., China). Tissue specimens from healthy and diseased
Statistical analysis
Results are reported as means ± SE. Statistical analysis of MIF tissue distribution was carried out with GraphPad Prism 5. 0 software. The Relative Expression Software Tool (REST 2008, version 2) was used to calculate the relative expression of mRNA target genes in real-time fluorescence quantitative PCR using the Pair Wise Fixed Reallocation Randomization Test. The mathematical model used was based on the mean crossing point deviation between the sample and the control group, normalized by the mean crossing point deviation of the reference genes. Specific amplification efficiencies were included in the correction of the quantification ratio. All data are presented as relative mRNA expression and the tissue MIF, TNF-α, and IL-1 expression as means ± standard deviation (SD). The Kruskal–Wallis H test was conducted to compare the tissue MIF, TNF-α, and IL-1 expression between control groups and treatment groups. The analyses were performed with SPSS version 13.0 (SPSS Inc., Chicago, IL, USA). One-way analysis of variance (ANOVA) was applied to compare different groups.
Results
Clinical signs
The diseased

Expression of MIF in fish tissues in response to V. parahaemolyticus infection
To determine the function of MIF, we first screened mRNA expression in the tissues of

MIF expression in nine selected tissues and organs of
Tissue MIF, TNF-α, and IL-1 expression in V. parahaemolyticus infected E. awoara and controls
MIF, TNF-α, and IL-1 levels in the tissues of
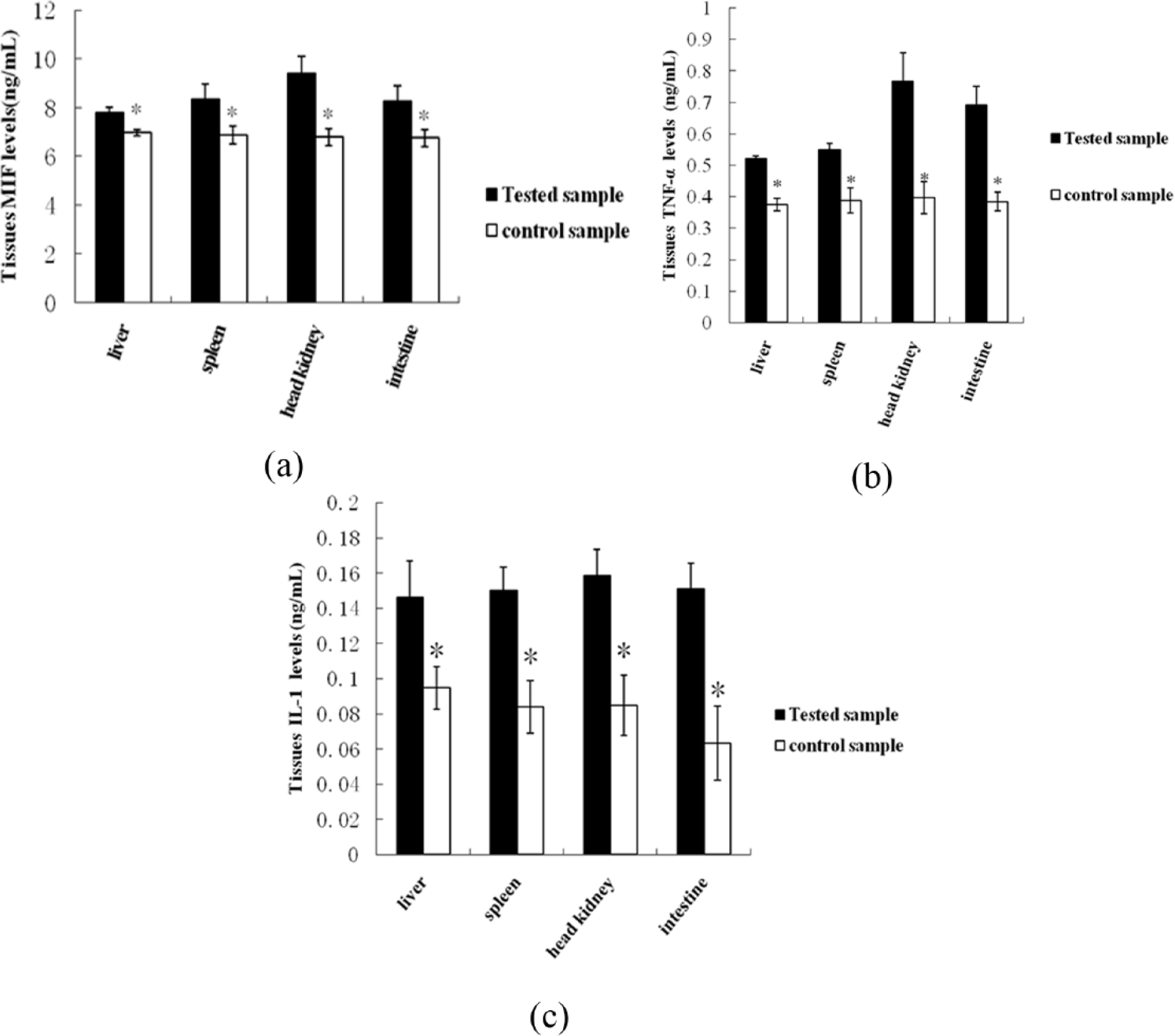
Tissue levels of MIF, TNF-α, and IL-1 of
A difference in TNF-α levels in tissues was noted between treatment group and controls. The concentrations of TNF-α in head kidney, intestine, spleen, and liver were 0.768 ± 0.09 ng/mL, 0.691 ± 0.06 ng/mL, 0.549 ± 0.02 ng/mL, and 0.521 ± 0.01 ng/mL in the infected group, and 0.397 ± 0.05 ng/mL, 0.384 ± 0.03 ng/mL, 0.389 ± 0.04 ng/mL, and 0.374 ± 0.02 ng/mL in controls. There was an obvious difference in TNF-α levels in the infected group compared with controls (
In our series, expressions levels of IL-1 in infected fish were higher than those in non-infected groups. The IL-1 content in the tissues of the head kidney, intestine, spleen, and liver of
The results showed that MIF, TNF-α, and IL-1 levels in the liver were significantly higher than those in spleen, head kidney, intestine, and muscle of diseased fish (Figure 3).
Furthermore, circulating MIF, TNF-α, and IL-1 levels in
Discussion
For the first time, we detected MIF expression in tissues of
In our study, tissue levels of MIF, TNF-α, and IL-1 were significantly increased in the tissues of
Footnotes
Declaration of Conflicting Interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the Science Foundation of the Fujian Province, China (#2016J01167, #2016NZ0001-3, Jimei University research〔2015〕19), the National Natural Science Foundation of China (General Program #31502194). Regional Demonstration of Marine Economy Innovative Development Project under contract No.12PYY001SF08, 14PYY050SF03 and 14CZP032HJ06.
