Abstract
To study the: (1) function of micro (mi)R-223 on H2O2-induced H9C2 cells; (2) relationship between miR-223 and insulin-like growth factor 1 receptor (IGF-1R); and (3) role of miR-223 on the phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT) pathway.
H9C2 cells were selected to establish the H2O2-injury model. Overexpression/low expression of miR-223 in H9C2 cells was constructed, respectively. Flow cytometry and western blotting were applied to measure the apoptosis, cell activity, and expression of related proteins. Dual-luciferase reporter gene assays (DLRGAs) were applied to test if miR-223 targeted IGF-1R. Overexpression/low expression of IGF-1R was constructed to test if miR-223 regulated IGF-1R expression negatively.
Increases in miR-223 expression were observed in H2O2-induced H9C2 cells. miR-223 absence improved H2O2-induced H9C2-cell apoptosis accompanied by an increase in B-cell lymphoma (Bcl)-2 expression and decrease in expression of Bax and cleaved caspase-3 (
miR-223 absence can ameliorate H2O2-induced cardiomyocyte apoptosis by targeting IGF-1R to regulate PI3K/AKT activity.
Keywords
Introduction
Oxidative stress injury (OSI) occurs in several cardiovascular diseases: myocardial ischemia, myocardial ischemia–reperfusion injury, and cardiac remodeling. 1 Reactive oxygen species (ROS) can be produced during these cardiovascular diseases. If the level of ROS exceeds that of antioxidant defenses, OSI occurs. 2 Subsequently, OSI can induce the necrosis and apoptosis of myocardial cells and, eventually, aggravate disease severity. 3 Necrosis usually occurs at the end stage of myocardial ischemia, whereas apoptosis usually proceeds through the entire pathologic process of cardiovascular diseases. 4 Therefore, alleviating OSI and reducing the apoptosis of myocardial cells are important for treatment of cardiovascular diseases.
Cardiomyocytes have an innate tendency toward apoptosis. Studies have shown that significant cell death occurs during differentiation of mouse and human embryonic stem cells toward cardiomyocytes, and a common biochemical pathway has been postulated.5,6 Remarkable differences have been found for the time of apoptosis and differentiation of cells. Akbari-Birgani et al.
5
found that, during apoptosis, expression of p53 and caspase-3/7 could be measured rapidly after cytochrome
miRNAs can regulate gene expression. The targeted relationship between miRNAs and genes has a network structure of interactions. 8 Several studies have demonstrated that miR-223 has a crucial role in cardiomyocyte apoptosis.9,10 Inhibition of miR-223 expression contributes to activation of expression of stromal interaction molecule 1 in phenylephrine-induced cardiomyocyte generation. 9 Silencing of miR-223-3p expression improved hypoxia-induced injury to H9C2 cells by preventing the apoptosis accompanied by an increase of B-cell lymphoma (Bcl)-2 expression and a decrease in expression of Bax and cleaved caspase-3. 10 However, how miR-223-3p controls apoptosis in cardiomyocytes requires more research.
Insulin-like growth factor 1 receptor (IGF-1R) is a target gene of miR-223-3p, which mediates expression of the phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT) pathway. 11 In addition, several studies have shown that IGF-1R participates in cardiomyocyte injury.12–14 IGF-1R has a constructive role in cardiovascular diseases because it prevents apoptosis of H9C2 cells and reduces expression of cleaved caspase-3/7. 14 Nevertheless, whether miR-223 affects cardiomyocyte apoptosis in hydrogen peroxide (H2O2)-induced cardiomyocytes is not known.
We investigated the effect of miR-223 on H2O2-induced cardiomyocyte apoptosis. We wished to ascertain the potential mechanism of action of miR-223 on H9C2 cardiomyocytes. The target relationship between miR-223 and IGF-1R was analyzed and the role of miR-223 on PI3K/AKT pathway was explored in H2O2-induced H9C2 cells. This study provides a theoretical basis for miR-223 application in the treatment of myocardial cell injury.
Materials and methods
Cell culture
Rat cardiomyocytes (H9C2 cells) were obtained from the Shanghai Cell Bank of the Chinese Academy of Sciences (Shanghai, China). Cardio-myocytes were cultured in Dulbecco’s modified Eagle’s medium (DMEM) (Gibco, Rockville, MD, USA) containing 10% fetal bovine serum (Sigma-Aldrich, Saint Louis, MO, USA) in an atmosphere of 5% CO2 (Thermo Fisher Scientific, Waltham, MA, USA) at 37°C. Cells at the logarithmic growth phase were used for experiments.
Model construction
H9C2 cells (105/mL) were inoculated in 96-well plates at 100 μL per well. After centrifugation, the supernatant was discarded. To induce oxidative stress, cells were treated with H2O2 (5, 20, 50, 100, 200, and 400 μmol/L) for 24 h. 15 Control groups were supplemented with DMEM. Cells were allowed to incubate at 37°C in an atmosphere of 5% CO2 for 24 h. Then, 10 μL of Cell Counting Kit-8 was added to the well and culture allowed for 4 h at 37°C. The optical density (OD) was measured at 450 nm. The H2O2 model with cell survival of ~50% was selected for further studies whereby

Cell transfection and grouping
Before induction of oxidative stress, H9C2 cells (density of 30%–50%) were transfected, respectively, with IGF-1R overexpression vector (IGF-1R), IGF-1R-negative control (IGF-1R-NC), IGF-1R silencing vector (si-IGF-1R), IGF-1R silencing negative control (si-NC), miR-223 mimic, negative control mimic (NC-mimic), miR-223 inhibitor, and negative control inhibitor (NC-inhibitor), all of which were obtained from RiboBio (Guangzhou, China), for 24 h. The IGF-1R fragment (20 ng; GenePharma, Shanghai, China) was cloned into plasmid pcDNA 3.1 (Thermo Fisher Scientific). The empty vector was used as the negative control.
Six groups were constructed: (1) control (no treatment); (2) model (H9C2 cells were treated with H2O2 (200 μmol/L) for 24 h); (3) miR-223 mimic (cells were transfected with miR-223 mimic for 24 h and then treated with H2O2 (200 μmol/L) for 24 h); (4) NC-mimic (cells were transfected with empty vectors of miR-223 mimic and treated with H2O2 (200 μmol/L)); (5) miR-223 inhibitor (cells were transfected with miR-223 inhibitor and treated with H2O2 (200 μmol/L)); and (6) NC-inhibitor (cells were transfected with miR-223 negative control inhibitor and then treated with H2O2 (200 μmol/L)). To determine if miR-223 could regulate apoptosis of H9C2 cells by targeting IGF-1R, the IGF-1R overexpression vector (IGF-1R), IGF-1R-negative control (IGF-1R-NC), IGF-1R silencing vector (si-IGF-1R), and IGF-1R silencing negative control (si-NC) were transfected (Figure 1).

The design of diagram of this study. Part 1 showed the effect of miR-223 on apoptosis of H2O2-induced H9C2 cells. Part 2 showed the effect of IGF-1R on apoptosis of H2O2-induced H9C2 cells. Effect of a serious of concentrations of H2O2 on the proliferation of H9C2.
Quantitative reverse transcription-polymerase chain reaction
After H2O2 treatment 24 h, H9C2 cells were suspended and collected. Total RNA was extracted using the TRIzol™ Plus RNA Isolation kit (Thermo Fisher Scientific). Reverse transcription (RT) was carried out with a TaqMan® RT kit (Qiagen, Stanford, VA, USA). Polymerase chain reaction (PCR) was undertaken under the following conditions: 94°C for 10 min, 94°C for 15 s, 60°C for 60 s, for 45 cycles. U6 was the internal reference. Results were analyzed using the 2−ΔΔCT (Livak method). Experiments were repeated thrice. The primer sequences (forward and reverse, respectively) were 5ʹ-GGCAGCACCCCATAAACTGTT-3ʹ and 5ʹ-AGTGCGTGTCGTGTCGTGGAG-3ʹ for miR-223; 5ʹ-CTCGCTTCGGCAGCACA-3ʹ and 5ʹ-AACGCTTCACGAATTTGCGT-3ʹ for U6.
Flow cytometry
After H2O2 treatment 24 h, an Annexin V-Fluorescein Isothiocyanate (FITC) kit and Propidium Iodide (PI) Apoptosis kit (Solarbio, Wuhan, China) were used to evaluate apoptosis. Cells from each group were collected and washed twice with 4°C sterile phosphate-buffered saline (PBS). Also, 1× binding buffer was used to adjust cells to 106 cell/mL. Next, 100 µL of the cell suspension was added to each tube. Then, 5 µL of Annexin V-FITC was added and mixed in a dark environment at room temperature. After 10 min, 5 µL of PI were added to the tube and culture allowed under the same condition. Then, PBS was added to make up the volume to 500 μL. Prepared samples underwent measurement by flow cytometry (BioRad, FACSCanto II, San Jose, CA, USA).
Western blotting
After H2O2 treatment for 24 h, total proteins were extracted. Samples (50 μg) were separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis using 10% gels (Beyotime, Guangzhou, China) and transferred to polyvinylidene difluoride (PVDF) membranes (Millipore, Billerica, MA, USA). PVDF membranes were blocked with 5% defatted milk for 1 h.
Next, 5% bovine serum albumin (BSA) was used to dilute the following primary antibodies: Bcl-2 (1:1100 dilution; catalog number, WBC011; R&D Systems, Minneapolis, MN, USA), Bax (1:1200; 33-6400; Thermo Fisher Scientific), cleaved caspase-3 (1:800; orb126608; Biorbyt, Shanghai, China), IGF-1R (1:1200; ab39675; Abcam, Cambridge, UK), PI3K (1:800; orb137259; Biorbyt), p-PI3K (1:800; orb338965; Biorbyt), AKT (1:900; orb213545; Biorbyt), p-AKT (1:900; orb222951; Biorbyt), and β-actin (1:1800; orb178392; Biorbyt). PVDF membranes were placed in primary-antibody solutions overnight at 4°C. After another three washes with TBST (Tris-buffered saline (TBS) (1 mL/L) and Tween-20, 10 min each time), the secondary antibody (1:1800; ab6721; Abcam) was added and incubation allowed for 2 h at 37°C. Electrochemiluminescence was used for detection. The density of each band was qualified by ImageJ (National Institutes of Health, Bethesda, MD, USA). β-actin acts as an internal reference gene.
DLRGA
Target genes for miR-223 were predicted using databases (TargetScan, miRanda, and miRDB). The wild-type and mutant-type of IGF-1R were amplified by overlapPCR. PmirGLO vectors and the target fragments of IGF-1R were recombined by XhoI and Xbal to construct pmirGLO-IGF-1R. H9C2 cells were seeded on 24-well plates (2 × 105 cells/well). The recombinant reporter vector was transfected into cells with Lipofectamine™ 2000 when the density reached 70%–80%. At 24 h after transfection, the DLRGA (Promega, Fitchburg, WI, USA) was undertaken.
Statistical analyses
Data were analyzed using SPSS v19.0 (IBM, Armonk, NY, USA). The variable is normally distributed and shows mean values ± standard deviation (SD). One-way analysis of variance (ANOVA) was carried out among multiple groups. The subsequent analysis was done using the Tukey test.
Results
Effects of H2O2 on the viability of H9C2 cells and miR-223 expression
H9C2 cells were treated with H2O2 (5, 20, 50, 100, 200, and 400 μmol/L), respectively. With an increase in the H2O2 concentration, percentage survival of cells decreased gradually (Figure 2(a)). Survival was 51.46% ± 2.62% when the H2O2 concentration was 200 μmol/L. Therefore, the injury model was established using H2O2 at 200 μmol/L. miR-223 expression was measured by quantitative reverse transcription-polymerase chain reaction (qRT-PCR) in groups. H2O2 treatment promoted significant expression of miR-223 in cardiomyocytes compared with that in the control group (

Effect of a serious of concentrations of H2O2 on the proliferation of H9C2 (a) and the expression of miR-223 in different groups (b). The experiments were repeated three times. The data were analyzed by the ANOVA and then followed by the Tukey test.
Inhibition of miR-223 expression inhibits apoptosis in H2O2-induced H9C2 cells
Apoptosis was upregulated after H2O2 treatment compared with that in control cells (

Inhibition of miR-223 suppressed the apoptosis of H2O2-induced H9C2 cells: (a) apoptosis rate in each group was analyzed by flow cytometry and (b) expression of Bcl-2, Bax, and cleaved caspase-3 in each group was analyzed by the western blot. β-actin acts as an internal reference gene. The experiments were repeated three times. The data were analyzed by the ANOVA and then followed by the Tukey test. The full length is presented in Supplement Document.
Inhibition of miR-223 expression enhances the activity of the IGF-1R/PI3K/AKT pathway in H2O2-induced H9C2 cells
Next, we investigated the underlying molecular pathways associated with miR-223 expression in regulation of H2O2-induced cardiomyocyte apoptosis. Western blotting was used to measure expression of IGF-1R/PI3K/AKT pathway-related proteins (Figure 4). Compared with the control group, H2O2 decreased expression of the proteins of IGF-1R, p-PI3K, and p-AKT in cardiomyocytes significantly (

Inhibition of miR-223-enhanced IGF-1R/PI3K/AKT activity in H2O2-induced H9C2 cells. Expression of IGF-1R, p-PI3K/PI3K, and p-AKT/AKT in each group was analyzed by the western blot. β-actin acts as an internal reference gene. The experiments were repeated three times. The data were analyzed by the ANOVA and then followed by the Tukey test. The full length is presented in Supplement Document.
miR-223 targets IGF-1R
We searched online databases (TargetScan, miRDB, and miRanda) and found that
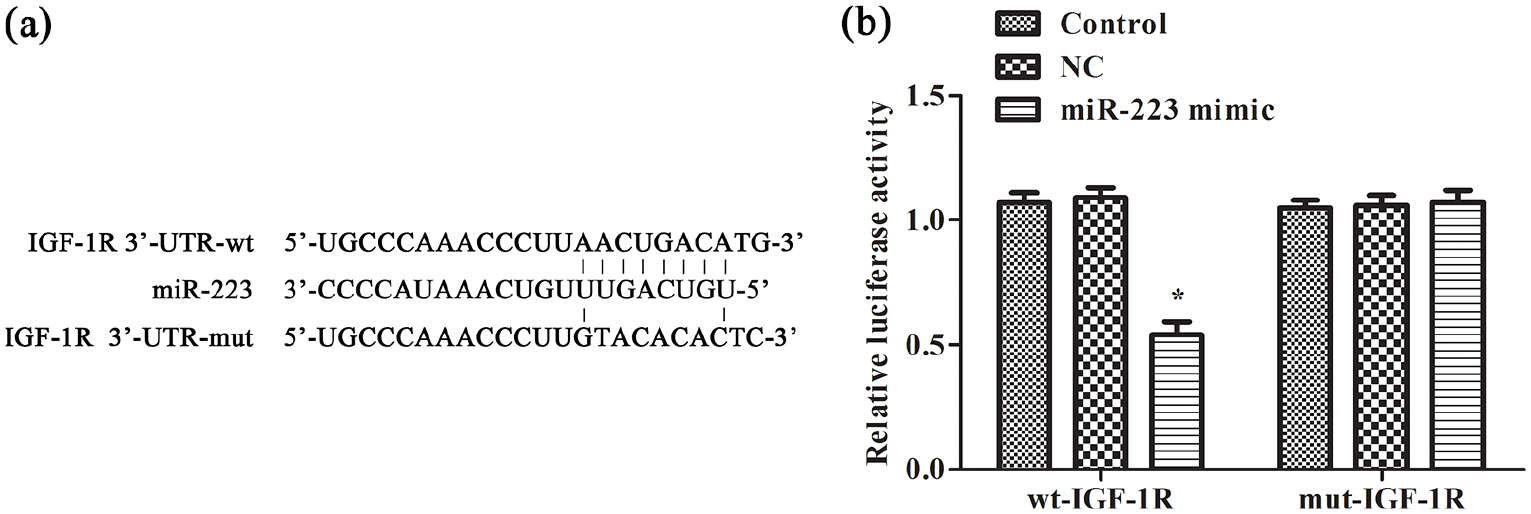
The targeting relation between IGF-1R and miR-223 was confirmed by dual luciferase reporter gene assay: (a) miR-223 and IGF-1R binding site and (b) dual luciferase reporter. The experiments were repeated three times.
IGF-1R regulates the activity of the PI3K/AKT pathway
To ascertain if IGF-1R could regulate the activity of the PI3K/AKT pathway in H2O2-induced cardiomyocytes, silencing of the IGF-1R vector was done. Expression of PI3K/AKT pathway–associated proteins was detected by western blotting (Figure 6). Expression of the proteins of IGF-1R, p-PI3K, and p-AKT was decreased significantly after silencing of IGF-1R expression (

Inhibition of IGF-1R downregulated PI3K/AKT pathway activities. Expression of IGF-1R, p-PI3K/PI3K, and p-AKT/AKT in each group was analyzed by the western blot. The experiments were repeated three times. The data were analyzed by the ANOVA and then followed by the Tukey test. The full length is presented in Supplement Document.
Inhibition of miR-223 expression restrains H2O2-induced H9C2-cell apoptosis via activation of the IGF-1R/PI3K/AKT pathway
Finally, we investigated if IGF-1R was involved in how miR-233 regulated H2O2-induced cardiomyocyte apoptosis. The IGF-1R overexpression vector, miR-223 inhibitor, and IGF-1R silencing vector were transfected to H9C2 cells, respectively (Figure 7). IGF-1R overexpression had a similar function to that of the miR-223 inhibitor: it significantly decreased the apoptosis of H2O2-induced cardiomyocytes compared with that in the model group (
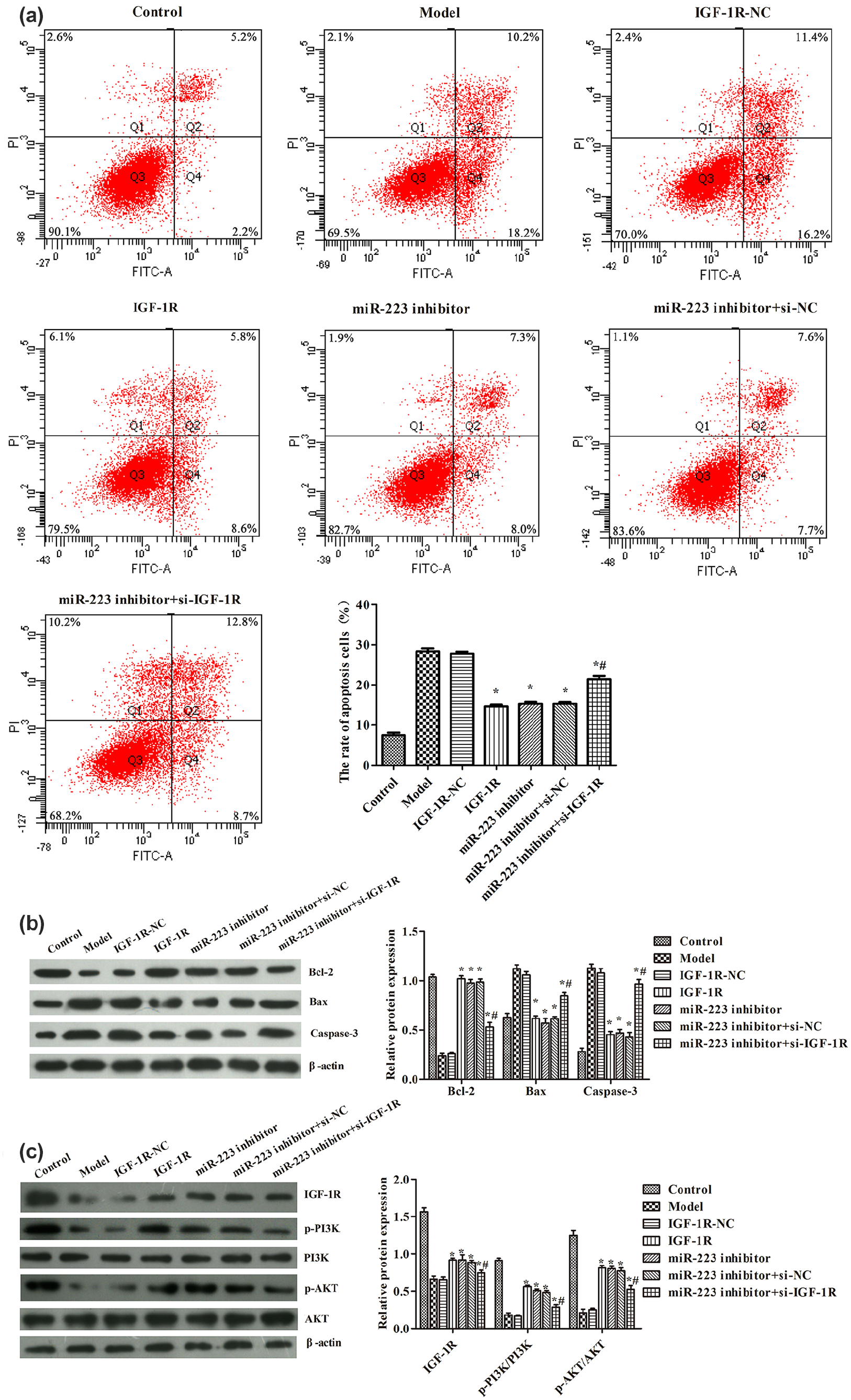
Inhibition of miR-223 suppressed H2O2-induced cardiomyocyte apoptosis via regulating IGF-1R/PI3K/AKT activity: (a) apoptosis rate in each group was analyzed by flow cytometry, (b) expression of Bcl-2, Bax, and cleaved caspase-3 in each group was analyzed by the western blot, and (c) expression of IGF-1R, p-PI3K/PI3K, and p-AKT/AKT in each group was analyzed by the western blot. β-actin acts as an internal reference gene. The experiments were repeated three times. The data were analyzed by the ANOVA and then followed by the Tukey test. The full length is presented in Supplement Document.
Discussion
High expression of miR-223 was shown in H2O2-induced H9C2 cells, and inhibition of miR-233 expression suppressed apoptosis accompanied by reduced expression of Bax and cleaved caspase-3. Concurrently, inhibition of miR-223 expression promoted the activity of the PI3K/AKT pathway. Furthermore, the target gene of miR-223 was confirmed by the DLRGA:
miRNAs, as a group of small non-coding RNAs, regulate several routes, including apoptosis. 16 Apoptosis is activated by two pathways: extrinsic/death receptor pathway or intrinsic/mitochondrial pathway. 7 In both pathways, caspase-9 is released first, followed by caspase-3/7. miR-223 has a negative role in hypoxia-induced cardiomyocyte apoptosis, in which silence of miR-223 improved hypoxia-induced H9C2 cell injury through preventing apoptosis accompanied by an increase of Bcl-2 expression and a decrease of Bax and cleaved caspase-3 expression.10,17 In this study, inhibition of miR-223 expression distinctly reduced H2O2-induced cardiomyocyte apoptosis, increased expression of the anti-apoptotic protein Bcl-2, and reduced expression of the pro-apoptotic proteins Bax and cleaved caspase-3. Our data revealed that miR-233 played a negative role in H2O2-induced cardiomyocyte apoptosis.
As one of the most frequently altered signaling networks, PI3K/AKT pathway plays a central role in cell growth, survival, and motility. 18 AKT promotes cell survival and protects against apoptosis through its ability to phosphorylate in cardiomyocytes.18,19 To further investigate the molecular role of miR-223 on expression of PI3K/AKT pathway, the expression of PI3K, AKT, and their phosphorylation was observed. We found that inhibition of miR-223 promoted the PI3K/AKT pathway activity in H2O2-induced H9C2 cells. These results suggest that inhibition of miR-223 suppressed the apoptosis of H2O2-induced H9C2 cells via PI3K/AKT pathway.
Gao et al. 11 showed that miR-223-3p targeted IGF-1R to mediate the PI3K/AKT pathway in regulation of mast cell apoptosis. IGF-1R has a constructive role in cardiovascular diseases because it prevents the apoptosis of H9C2 cells and reduces expression of cleaved caspase-3/7. 14 Our results are consistent with those of scholars showing that IGF-1R overexpression suppresses the apoptosis and activates the PI3K/AKT pathway in H2O2-induced cardiomyocytes. The targeting relationship between IGF-1R and miR-223 was confirmed by the DLRGA in H9C2 cells. Fan et al. 20 revealed that PI3K/AKT signaling can influence cardiomyocyte apoptosis by regulating various target proteins, such as Bcl-2 and Bax. Liu et al. 21 suggested that activation of PI3K/AKT signaling can decrease apoptosis in oxidative damage induced by H2O2. Those data are consistent with our experimental results.
Only in vitro experiments were carried out to reveal the role of miR-223 targeting IGF-1R to regulate the PI3K/AKT pathway in oxidative stress–induced apoptosis of myocardial cells. Other potential mechanisms of miR-223 targeting IGF-1R in oxidative stress–induced cardiomyocyte injury will be investigated in our future studies.
Conclusion
miR-223 showed high expression in H2O2-induced H9C2 cells. Inhibition of miR-233 expression suppressed the apoptosis of cells by targeting IGF-1R to increase the activity of the PI3K/AKT pathway. Our research revealed a basic theory on the mechanisms of myocardial cell apoptosis in cardiovascular diseases.
Supplemental Material
Supplyment_material – Supplemental material for Silencing of miR-223 expression inhibits the apoptosis of H2O2-induced cardiomyocytes by increasing the activity of the IGF-1R/PI3K/AKT pathway
Supplemental material, Supplyment_material for Silencing of miR-223 expression inhibits the apoptosis of H2O2-induced cardiomyocytes by increasing the activity of the IGF-1R/PI3K/AKT pathway by Qianqian Zhou, Yufen Li and Tianjin Gu in European Journal of Inflammation
Footnotes
Author contributions
Q.Z. participated in study design, data interpretation, and wrote the manuscript. Y.L. participated in study design, data collection and data analysis, and prepared the manuscript. T.G. participated in study design and data analysis.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplement material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
