Abstract
The vitamin D receptor (VDR) plays an important role in the pathogenesis of systemic lupus erythematosus (SLE) mainly due to its anti-inflammatory and immunomodulatory activities. Interferon-inducible protein-10 (IP-10) is a chemokine found to be increased in SLE patients and correlated with severity of lupus nephritis. In this case control study, we investigated the connection between VDR expression and chemokine IP-10 in peripheral blood mononuclear cells (PBMCs) of SLE patients with or without renal involvement. 62 SLE patients and 30 healthy subjects were enrolled between 2014 and 2016. PBMC VDR mRNA and IP-10 mRNA were quantified by real-time PCR. PBMC VDR protein was measured by Western blotting. PBMC VDR mRNA and protein were downregulated, whereas IP-10 mRNA was upregulated in SLE patients with renal involvement. VDR mRNA levels were negatively correlated with systemic lupus international collaborating clinics (SLICC) renal activity scores (r = −0.423, P = 0.016) and IP-10 mRNA levels (r = −0.428, P = 0.008) in SLE with renal involvement. This study demonstrates that PBMC VDR was negatively correlated with SLICC renal activity and chemokine IP-10 in SLE patients with renal involvement which suggests that VDR downregulation may be used as an indicator of renal injury in SLE patients.
Keywords
Introduction
It is well known that systemic lupus erythematosus (SLE) is an inflammatory autoimmune disease characterized by immune cell-mediated injury to multiple organs including the kidney. Although considerable progress has been made in understanding the complicated clinical features and unpredictable progression of this disease, the pathogenesis of SLE remains unclear. 1 Recently, the role of vitamin D as an environmental factor in SLE has drawn considerable attention. 2 Studies have reported that vitamin D deficiency or insufficiency is common in SLE patients.3,4 The level of vitamin D was negatively correlated with disease activity and the levels of anti-dsDNA and anti-C1q antibodies, as well as related to renal injury and urinary protein levels in lupus patients.5,6 Vitamin D deficiency could play a significant role in the onset and progression of SLE. 7 The active form of vitamin D is 1α,25-dihydroxyvitamin D3 [1,25(OH)2D3]. Through activating the vitamin D receptor (VDR), 1,25(OH)2D3 exerts potent anti-inflammatory activity, which involves blockade of nuclear factor kappa B (NF-κB) activation and inhibition of pro-inflammatory cytokine production.8,9
Interferon-inducible protein-10 (IP-10) is a member of the CXC subfamily of chemokines. IP-10 plays an important role in inducing inflammatory responses as it participates in abnormal immune reactions by enhancing the Th1 response. 10 Previous studies confirmed that the level of IP-10 was increased in SLE patients and correlated with the disease activity of SLE and the severity of lupus nephritis (LN). 11 It has also been reported that VDR agonists could inhibit the production and release of IP-10 in many cells such as T cells, human thyroid cells, renal tubular cells, cardiomyocytes, and pancreatic cells.12–14 However, no previous study has investigated the relationship between VDR and cytokine IP-10 in SLE.
Our previous work has reported that VDR expression in renal tissues of LN patients was significantly downregulated and negatively correlated with the activity index of renal injury. 15 Therefore, we conducted this investigation to explore the connection between VDR and IP-10 in SLE patients with renal involvement to better understand the anti-inflammatory mechanism of VDR in SLE.
Materials and methods
Patients
In all, 62 patients initially diagnosed as SLE were recruited from the Department of Nephrology and Rheumatology in the Third Xiangya Hospital of Central South University between April 2014 and December 2016. At the same time, 30 healthy volunteers were recruited as control. All patients met the American College of Rheumatology (ACR) classification criteria of SLE. 16 SLE disease activity index (SLEDAI) was used to assess the disease severity. 17 Individuals with systemic diseases such as acute or chronic infectious diseases, hepatitis B virus surface antigen positive, diabetes, and other immunological diseases and pregnant and lactating women were also excluded. This study was approved by the Institutional Ethical Committee of the Third Xiangya Hospital of CSU (NO. 201403). All subjects signed informed consents.
Study subjects
The presence of renal involvement was determined by renal SLEDAI score 18 of SLEDAI-2000 including hematuria, proteinuria, pyuria, and urinary casts. Proteinuria: >3 g/day (11′), 1–3 g/day (5′), 0.5–1 g/day (3′); urine white blood cells ⩾5/high power field (1′); and urine red blood cells ⩾5/high power field (3′). Renal SLEDAI scores ranged from 0 (no renal manifestation) to a maximum score of 16; 62 SLE patients were divided into two groups on the basis of their renal SLEDAI scores at the time of enrollment: no renal manifestation group (score = 0, 30 patients) and with renal involvement group (scores ⩾1, 32 patients). The healthy control group was matched for age and gender without history of immunological diseases or family history, cardiovascular diseases, liver and kidney diseases, central diseases and other systemic diseases, and no recent history of infection.
Blood cell preparation
A volume of 5 mL of heparinized peripheral venous blood samples were collected from all subjects and diluted 1:1 with phosphate-buffered saline (PBS) buffer, and then, equal volume of lymphocyte separation medium was added. The liquid layer of white mist was separated by centrifugation at 2000 r/min for 20 min at 4°C and then mixed with another 10 mL PBS. After centrifugation at 1500 r/min for 10 min at 4°C and washed twice, the cells were harvested for extracting RNAs and proteins.
Reverse transcription quantitative polymerase chain reaction
Total RNAs were extracted from peripheral blood mononuclear cells (PBMCs) with the TRIzol reagent as described in our previous publication. 19 First strand cDNA was synthesized by using a Fermentas quantitative real-time polymerase chain reaction (qRT-PCR) kit according to the manufacturer’s instructions. Quantitative real-time PCR was carried out using a SYBR Green PCR Master Mix (Toyobo, Japan) on the 7300 Real Time PCR System (Eppendorf, Germany) to measure the expression levels of VDR and IP-10 messenger RNAs (mRNAs). PCR reactions were performed with pre-denaturation at 95°C for 2 min followed by denaturation at 95°C for 10 s, annealing at 58°C for 20 s, and extension at 72°C for 30 s with 40 cycles, followed by extension at 72°C for 5 min. Relative amounts of gene expressions were calculated using the 2−ΔΔCt formula. The PCR primers for VDR, IP-10, and β-actin were listed as follows:
VDR F: 5′-AGTGCAGAGGAAGCGGGAGATG-3′;
VDR R: 5′-CTGGCAGAAGTCGGAGTAGGTG-3′;
IP-10 F: 5′-GCCAATTTTGTCCACG-3′;
IP-10 R: 5′-CCTTTCCTTGCTAACTG`CT-3′;
β-actin F: 5′-CATCCTGCGTCTGGACCTGG-3′;
β-actin R: 5′-TAATGTCACGCACGATTTCC-3′.
Western blot
Total PBMC proteins were extracted from 62 random samples, including 17 subjects from each of the SLE groups with or without renal involvement and the control group according to the manufacturer’s instructions (Merck, USA). Proteins (30 μg) from each sample were then separated by sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto polyvinylidene fluoride (PVDF) membranes. The membranes were incubated overnight at 4°C with the primary antibody against VDR (sc-13,133; Santa Cruz Biotechnology). After three washes with Tris-buffered saline, 0.1% Tween 20 (TBST) buffer, the membranes were treated with secondary antibody for 2 h at room temperature. Protein contents were detected using an enhanced chemiluminescent reagent. The density of the bands was quantified by Image J software (NIH, USA). The relative expression of VDR protein was calculated by the ratio of gray value to the internal parameters.
Statistical analyses
Statistical analyses were carried out using SPSS22.0 software. All results were expressed as mean ± standard deviation. The difference between SLE patients with and without renal involvement was conducted using Student’s t-test and one-way analysis of variance for the SLE and the control groups. Spearman correlation analysis was used to confirm the correlations between the clinical and biochemical data. P value <0.05 was considered statistically significant.
Results
Clinical and demographic characteristics
There were no statistically significant differences between healthy control group and SLE groups with or without renal involvement regarding age and sex composition. The demographic characteristics of the control and the SLE groups with or without renal involvement are given in Table 1.
Clinical and demographic characteristics of the studied groups.

GFR: glomerular filtration rate; C3: complement factor 3; ESR: erythrocyte sedimentation rate; –: no data; ANOVA: analysis of variance; SLEDAI: Systemic Lupus Erythematosus Disease Activity Index.
Data are expressed as mean ± SD. Data are compared by chi-square test or one-way ANOVA.
P < 0.05 controls vs with renal involvement and no renal manifestation; #P < 0.05 no renal manifestation vs with renal involvement.
VDR expression in PBMCs of SLE with or without renal involvement
The relative mRNA (Figure 1(a)) and protein (Figure 1(b)) expression of VDR in the SLE patients with or without renal involvement were significantly lower than those in the control group (P < 0.01), and the SLE patients with renal involvement had much lower levels compared with those without renal involvement (P < 0.01).

VDR expression in PBMCs from SLE patients. VDR mRNA relative levels in healthy control (n = 30), SLE with renal involvement group (n = 32), and SLE without renal involvement group (n = 30), determined by real-time PCR. VDR protein was measured by (a) Western blotting and quantified by (b and c) densitometry. Dates are presented as the mean ± SD. **P = 0.001 vs control group; ##P = 0.005 vs SLE without renal involvement group.
IP-10 expression in PBMCs of SLE with or without renal involvement
To evaluate the association between VDR and IP-10, we measured the IP-10 mRNA levels (Figure 2). Compared with the control group, IP-10 mRNAs were significantly higher in SLE groups (P < 0.01), and the SLE patients with renal involvement had the highest IP-10 expression (P < 0.01).
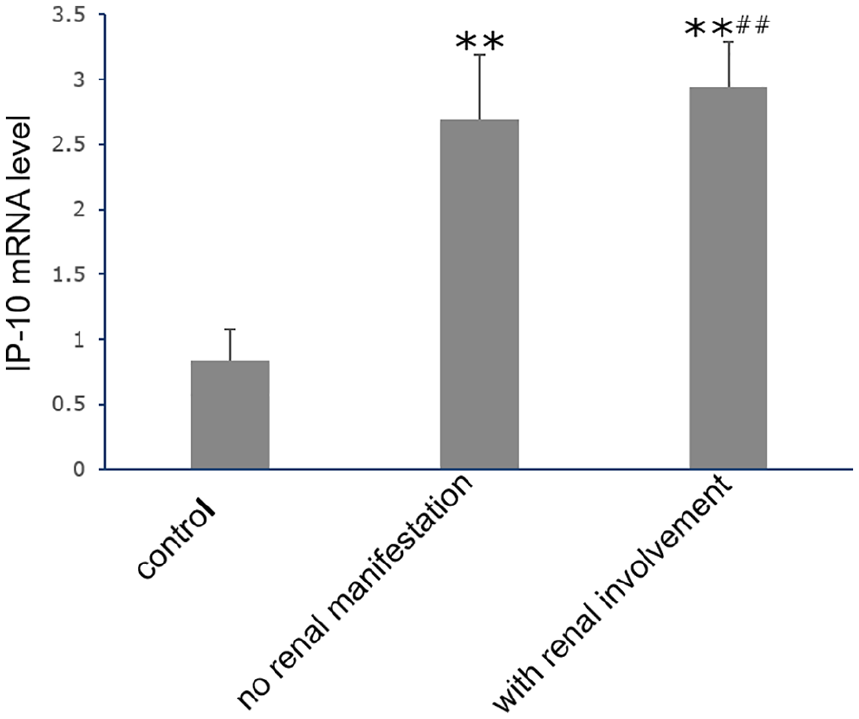
IP-10 mRNA level in PBMCs from SLE patients. IP-10 mRNA relative levels in healthy control (n = 30), SLE with renal involvement group (n = 32), and SLE without renal involvement group (n = 30), determined by real-time PCR. Dates are presented as the mean ± SD. **P = 0.001 vs control group; ##P = 0.015 vs SLE without renal involvement group.
Correlation between VDR mRNA and systemic lupus international collaborating clinics renal activity scores in SLE with renal involvement
The levels of VDR mRNAs were inversely correlated with the systemic lupus international collaborating clinics (SLICC) renal activity score (r = –0.423, P = 0.016) in the 32 patients with renal involvement (Figure 3).

Correlation between VDR mRNA and SLICC renal activity scores in SLE with renal involvement group. Scatter plot shows an inverse relationship between VDR mRNA level and SLICC renal activity score (r = –0.423, P = 0.016).
Correlation between VDR mRNA and IP-10 mRNA in SLE
The VDR mRNA levels were negatively correlated with the IP-10 mRNA levels in PBMCs (r = –0.428, P = 0.008) from the SLE patients with renal involvement (n = 32; Figure 4), but no such correlation was seen in SLE without renal involvement (n = 30).

Correlation between VDR mRNA and IP-10 mRNA in PBMCs of SLE patients with renal involvement group. Scatter plot shows an inverse relationship between VDR mRNA level and IP-10 mRNA level (r = –0.428, P = 0.008).
Discussion
In this study, we showed that PBMC VDR expression was significantly downregulated in SLE patients with renal involvement, which is consistent with our previous work. 15 IP-10 mRNAs were significantly elevated in SLE groups, but the patients with renal involvement had much higher IP-10 levels compared with those without renal involvement. We also found that the level of VDR mRNA was negatively correlated with SLICC renal activity score in the SLE patients with renal involvement. Moreover, VDR mRNA was inversely correlated with IP-10 mRNA in SLE patients with renal involvement, but not in SLE patients without renal involvement. Taken together, these findings suggest that the level of VDR in PBMCs could be used as a prediction of renal injury in SLE patients.
VDR is a ligand-dependent nuclear transcription factor that regulates many target genes with a variety of biological functions, including genes with inflammatory activities. As such, VDR has potent anti-inflammatory activities, and low VDR expression has been reported in various inflammatory diseases.20,21 Our previous work has reported the downregulation of VDR expression in renal tissues of LN patients, which was negatively correlated with renal activity and the severity of renal injury. 15 Based on this finding, we speculated that the VDR expression in PBMCs of SLE patients with renal involvement would show the same trend. Indeed, our data demonstrated that PBMC VDR expression was significantly downregulated in SLE patients, and those with renal involvement had the lowest levels. The downregulation of PBMC VDR may be caused by inflammation, as tumor necrosis factor (TNF)-α has been shown to promote microRNA-mediated VDR suppression. 22
Chemokines play important roles in immune response of SLE. Previous studies have confirmed that chemokines such as IP-10, MCP-1, and RANTES were related to the progress of SLE disease.23,24 Odler’s work found that plasma IP-10 was significantly higher in SLE with pulmonary involvement and plasma IP-10 might serve as a potential biomarker. 25 Our data also show that PBMC IP-10 mRNA was upregulated in SLE patients especially in those with renal involvement. This observation suggests that PBMC IP-10 may also be a potential biomarker for SLE with renal involvement. IP-10 has a strong chemotaxis effect on T cells, monocyte macrophages, and dendritic cells. It can selectively induce Th1 cells to express CXC3 receptor and promote Th1 response in SLE. 26 It is clear that VDR agonists can inhibit IP-10 expression in T cells, Langerhans cell, and pancreatic cells.9,27 For example, in patients with thyroiditis, the VDR agonist elocalcitol can inhibit the production of IP-10 in thyroid cells and turn Th1 immune response into Th2 response.12 But, it is unclear whether VDR agonist could reduce the expression of IP-10 in SLE with renal involvement. Our current study showed that PBMC VDR expression was inversely correlated with IP-10 in SLE with renal involvement, which suggests ligand-activated VDR may be able to inhibit the production of IP-10 in SLE.
This study has a number of limitations. This is a retrospective study with limited sample size; thus, studies with larger sample size are needed to confirm the finding. In addition, whether VDR indeed regulates the expression of IP-10 in SLE remains unknown. Further studies are required to clarify the mechanism underlying this potential regulation. Moreover, it is also interesting to address whether supplementation of VDR agonist can reduce IP-10 in SLE patients to alleviate the disease.
Conclusion
In summary, this work demonstrated that PMBC VDR was significantly downregulated and IP-10 was elevated in SLE groups with renal involvement. VDR was negatively correlated with SLICC renal activity and IP-10 in SLE patients with renal involvement. Thus, PMBC VDR and IP-10 could be used as potential indicators of renal injury activity in SLE patients. Future studies are needed to understand the mechanism whereby VDR regulates IP-10.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Ethical approval for this study was obtained from the Institutional Ethical Committee of the Third Xiangya Hospital of Central South University (Approval Number/ID: 201403).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the National Natural Science Foundation of China (No. 81870498 and No.818770499), the Natural Science Foundation of Hunan Province (No. 2019JJ40463), and the Fundamental Research Funds for the Central Universities of Central South University (2019zzts909). We thank all patients for their participation in this study.
Informed consent
Informed consent was obtained from all individual participants included in the study. Written informed consent was obtained from all subjects before the study.
