Abstract
Henoch–Schönlein purpura (HSP) could involve multiple organs, including gastrointestinal tract. It is commonly observed in children, but occasionally also in the elderly. It is challenging to diagnose HSP with delayed purpura. Herein, we report an elderly with HSP, whose non-thrombocytopenic purpura erupted more than 1 month after the first onset of abdominal pain.
Introduction
Henoch–Schönlein purpura (HSP) is a small-vessel vasculitis associated with deposition of IgA immune complex, usually involving multiple organs, such as the gastrointestinal tract, skin, kidney, joint, and even central nervous system. While the cause of HSP remains elusive, the prevalence of HSP shows a dramatic difference between youngsters and seniors. More than 90% HSP patients are under 10 years old with the average age at onset of 6.4 years, 1 whereas HSP is relatively uncommon in adults.2–4 The annual incidence of HSP in adults is reported to be about a tenth of that in children. 5 Since HSP is infrequent in the middle aged and the elderly, when its initial manifestation is atypical, it might be challenging to recognize. Atypical clinical presentations of HSP, including bullous lesions, anuria, and intussusception, increase the difficulty in prompt diagnosis and management.6–8 Although gastrointestinal involvement is relatively common in adult HSP, 9 a long time gap between preceding gastrointestinal presentations and delayed erupted cutaneous lesions is uncommon.2,3,7 Considering the typical cutaneous lesions such as palpable purpura or petechiae is necessary to diagnose HSP under the current criteria, 10 it is a challenging task to diagnose HSP in a senior without cutaneous lesions. Herein, we report an HSP case, whose purpura erupted at more than 1 month after the onset of abdominal pain and lower gastrointestinal erosions.
Case report
A 51-year-old woman was admitted to our department with a complaint of intermittent abdominal pain for 20 days. Unbearable colicky pain recurred every 3–4 days. She did not have hematemesis, melena, hematochezia, diarrhea, or fever. The patient denied intake of non-steroidal anti-inflammatory drugs. Two weeks before the admission, erosions in ileocecum and terminal ileum were found by colonoscopy (Figure 1(a)). Biopsy suggested significant neutrophils infiltration in mucosa and submucosa, small vessels being encompassed by neutrophils, and microthrombus formation in small vessels (Figure 1(b)). The patient was prescribed with 5-amino salicylic acid, but abdominal pain still recurred intermittently. A series of laboratory tests were performed, and the results were as follows: normal hemoglobin (132 g/L), elevated leukocyte ((9.88 × 109/L, normal value (nv): (3.50–9.50) × 109/L)) and platelet counts ((388 × 109/L, nv: (100–300)×109/L)), normal liver and renal function, normal urine test, normal serum tumor marker levels (carcinoembryonic antigen, alpha-fetoprotein, carbohydrate antigen 125, carbohydrate antigen 199, and carbohydrate antigen 724), positive fecal occult blood test, increased serum level of IgA (4760 mg/L, nv: 836–2900 mg/L) and IgE (163.0 IU/mL, nv: 0.1–150.0 IU/mL), negative autoantibody (including rheumatoid factor, antinuclear antibody, anti-double-stranded DNA, and antineutrophil cytoplasmic antibody), and negative tuberculin test. Gastroduodenoscopy was also performed and only verrucosa gastritis was found in the antrum of stomach. In addition, chest and abdominal computed tomography suggested slight edema and thickening of distal ileum (Figure 1(c)). The diagnosis remained obscure and the symptoms persisted.
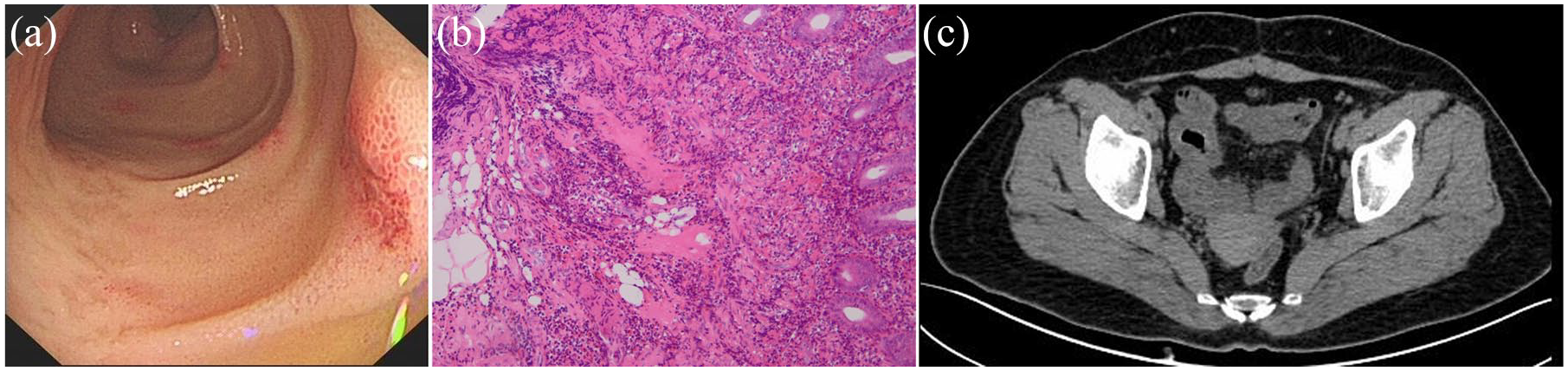
Imaging of colonoscopy, hematoxylin–eosin staining, and computed tomography. (a) Erosions at terminal ileum, (b) hematoxylin–eosin staining of terminal ileum, magnifications 200×, and (c) slight edema and thickening of terminal ileum.
However, on day 32 after the first onset of abdominal pain, purpura appeared on the extensor surfaces of lower extremities (Figure 2(a)). No other cutaneous lesions, such as linear nonblanching lesion or bullous lesion,11,12 were observed. Thus, non-thrombocytopenic purpura in lower extremities, together with recurrent abdominal pain, indicated the diagnosis of HSP. 10 Further immunohistochemistry stain of intestinal samples obtained from colonoscopy found that IgA deposited in small vessels (Figure 2(b)), in keeping with HSP. After intravenous methylprednisolone (40 mg daily for 5 days) was given, abdominal pain rapidly relieved and purpura disappeared gradually. Then, oral administration of prednisone was prescribed and started at the dosage of 40 mg/day. Then, prednisone was titrated down by 5 mg every 3 days when dosage was higher than 20 mg/day, followed by 5 mg decrement weekly until the withdraw. The whole duration of steroid treatment was about 7 weeks. The second-look colonoscopy was performed at 2 months later, showing that the erosions in ileocecum and terminal ileum completely resolved (Figure 2(c)).

Imaging of cutaneous lesions, immunochemistry staining, and second-look colonoscopy. (a) Purpura on lower extremities, (b) positive immunochemistry staining of small vessels in terminal ileum with IgA, 400×, and (c) normal appearance of terminal ileum without erosions.
Discussion
The diagnosis of HSP is made with the presence of palpable purpura or petechiae with lower limb predominance plus any one or more items listed as follows: (1) diffuse abdominal pain; (2) leukocytoclastic vasculitis (small vessel) or proliferative glomerulonephritis with predominant deposition of IgA; (3) arthritis or arthralgia; and (4) renal involvement (hematuria with or without proteinuria). 10 However, when the cutaneous lesions are absent, HSP might be difficult to diagnose with the non-specific symptom of abdominal pain. In 16.7% pediatric patients, abdominal pain precedes the onset of purpura by 5.1 days averagely, making the prompt diagnosing difficult. 13 Moreover, gastrointestinal involvement as the initial manifestation of HSP is even less in adults (around 10%) than that in children.2,3 Thus, it is challenging to uncover the mask of HSP in this 51-year-old woman with gastrointestinal involvement as the initial manifestation and delayed eruption (after 1 month) of purpura, which is very atypical. It is also worthy of noticing that other atypical symptoms of HSP include articular and renal manifestations before the presence of skin lesions. 14
Leukocytoclastic vasculitis and proliferative glomerulonephritis are the typical histological features for HSP. As 15% and 21% biopsies for skin and kidney are negative, respectively, 4 other susceptible sites, such as gastrointestinal tract, should be scrutinized for biopsy in suspected HSP individuals. Similarly, for gastrointestinal pathology of HSP, the typical findings were leukocytoclastic vasculitis, inflammation, and extravasated red blood cells (RBC).15,16
In addition to the routine pathological observation, it is reported that IgA deposition could be found in dermal capillaries in HSP patients without typical cutaneous lesions.5,17 Moreover, IgA deposition in gut is identified in more than 60% of HSP patients, and the obstruction of intestinal capillary could be elicited by IgA complex, giving rise to intestinal ischemia. 18 Based on this pathogenesis, the gastrointestinal symptoms and signs of HSP vary from abdominal pain, gastrointestinal bleeding to intussusception, and even perforation. 18 In the intestinal specimen of our patient, neutrophil infiltration, microthrombus formation in small vessels, as well as diffused IgA deposition were highly suggestive of HSP, even at the time of biopsy when cutaneous lesions were not developed. Therefore, IgA stain might be a promising way to facilitate HSP diagnosing.
Conclusion
An elderly woman with HSP is reported in this case, which highlights that in HSP patients, the onset of gastrointestinal manifestations could precede non-thrombocytopenic purpura for more than 1 month, and the pathology of gastrointestinal lesions would be helpful to the diagnosis of HSP.
Footnotes
Acknowledgements
The authors thank Ms. Yu (Sichuan University) for the inspiring effect of her special personality.
Author’s note
The authors wish it to be known that, in their opinion, the first two authors should be regarded as joint First Authors.
Author contributions
L.-H.Z. and H.T. conceived and designed the project. T.L., D.H., and S.-L.W. collected the data. L.-H.Z., T.L., and H.T. analyzed and interpreted the data. L.-H.Z. and S.-L.W. searched the literature. L.-H.Z. and T.L. drafted the manuscript. H.T. supervised the project and revised the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Natural Science Foundation of China (Grant No. 81700539); Foundation of Health and Family Planning Commission of Sichuan Province (Grant No. 16PJ276); Fundamental Research Funds for the Central Universities (Grant No. 2017SCU11047); China Postdoctoral Science Foundation Grant (Grant No. 2019M653436); and Post-Doctor Research Project, West China Hospital, Sichuan University (Grant No. 2019HXBH013).
Informed consent
Written informed consent was obtained from the patient for her anonymized information to be published in this article prior to study initiation.
