Abstract
Henoch-Schönlein purpura (HSP) is a kind of allergic systemic necrotizing vasculitis. The incidence of renal involvement is 30%–50% in HSP patients. A recent national survey demonstrated that the treatment results of several randomized controlled trial (RCT) studies on Henoch-Schönlein purpura nephritis (HSPN) using Western medicine, such as glucocorticoids, immunosuppressive agents, and supportive therapy, were not satisfactory. Hence, there is a need to explore traditional Chinese medicine (TCM) syndrome elements and its related factors of HSPN in the real world to observe the clinical curative effect of TCM treatment. The Chinese Cohort Study of HSPN is the first national HSPN cohort in China. This study will enroll approximately 350 HSPN patients who are within 14–74 years old and will be followed up for 24 months. Questionnaires, body measurement, laboratory tests, and biomaterials will be collected at baseline, quarterly, and annually. The principal clinical outcomes of the study will include the summing up of TCM syndrome elements and its related factors of HSPN. At the same time, the clinical effect of TCM in alleviating the clinical uncomfortable symptoms of HSPN, proteinuria, and renal function will be observed and confirmed. This study will provide important evidences for the constitution and clinical effect of TCM syndrome elements and lay a foundation for further conducting RCT clinical research on the treatment of HSPN by TCM. At the same time, this will provide valuable information to establish clinical guidelines and healthcare policies for TCM treatment of HSPN in China.
Introduction
Henoch-Schönlein purpura (HSP) is a kind of allergic systemic necrotizing vasculitis that affects the kidneys in approximately 30%–50% of patients.1,2 It usually manifests as proteinuria, hematuria, and renal impairment, which is known as Henoch-Schönlein purpura nephritis (HSPN). Previous data show that about 1%–3% of patients progressed to the end-stage kidney disease gradually. 1 The above findings prompt us that HSPN is harmful to the health and is highly concerned. Both of guidelines on the clinical practice of glomerular disease released by the China HSPN expert consensus in 2008 and the Kidney Disease: Improving Global Outcomes (KDIGO) in 2012 recommend angiotensin-converting enzyme inhibitors (ACEIs) or angiotensin receptor blockers (ARB) as therapeutic drugs to reduce proteinuria. Meanwhile, glucocorticoids and immunosuppressive agents can be selected based on the differences of proteinuria level and the degree of the pathological changes.3,4 However, some randomized controlled trial (RCT) findings showed that the curative effects of glucocorticoids, immunosuppressive agents, and supportive treatment were not satisfactory, or no significant difference in efficacy between the experimental group and the control group (placebo or supportive treatment).5–10
Nevertheless, three non-strict random RCT studies11–13 indicated that guidelines for traditional Chinese medicine (TCM) diagnosis and treatment played an important role in standardizing TCM treatment of HSPN, which was obviously better than glucocorticoid and renin–angiotensin system (RAS) blockers in relieving clinical symptoms. But these researches included no long-term follow-up studies. The existing clinical research results have no high-level evidence-based medical evidence to support curative effect of TCM treatment of HSPN. So it urgently needs to have a strictly designed prospective clinical study to define curative effect.
We have, therefore, established the first prospective, multicenter and large samples Cohort Study in China, aiming to explore HSPN syndrome elements of TCM in the real world, to observe the clinical curative effects of TCM treatment, further to lay the foundation for RCT studies of HSPN by TCM treatment.
Methods
Study design
This Chinese HSPN population-based multicenter prospective cohort study comprises 10 clinical centers located in seven cities of seven provinces in China (Figure 1). All of these clinical centers are Renal Departments from different hospitals. The study will establish a baseline cohort of approximately 350 Chinese HSPN patients, and each study center will collect 35 patients. The TCM syndrome scale will be used as the basic evidence of syndrome judgment, and the efficacy indexes will be measured in 24-h urinary total protein (24 h UTP), Scr, and estimated glomerular filtration rate (eGFR). Participants in the study will be followed up until the 24-h UTP reaches more than 3.0 g, the serum creatinine level doubles, or the end-stage renal disease is reached. Their clinical information and biomaterials will be collected at baseline, quarterly, and annually during the follow-up period.

Geographical distribution of clinical centers (red stars) in HSPN study. All clinical centers are located at different regions of seven provinces that are indicated by a star.
Study organization
This study is organized by Hubei Hospital of TCM, which hosts the central database, data coordinating center, and central laboratories. In addition, the study has a steering committee for developing policies for ancillary studies, performance standards, publications and presentations, and a scientific advisory committee for providing professional advices for the study protocol and practice.
The study was approved by the Ethics Committee of Hubei Hospital of TCM and in adherence with the Declaration of Helsinki. All participants provided a written informed consent before data collection.
Participants
This study enrolled HSPN patients who had an eGFR of ≥60 (calculated by the modification of diet in renal disease (MDRD) formula; mL min−1 1.73 m−2) and a mild-to-moderate spectrum of proteinuria (Table 1). This setting ensured achieving the desired composition of sub-cohorts for further analysis. There were target ranges for recruitment based on the different degree of proteinuria. For each research center, the patient component ratio is not required to be consistent with Table 1, which will be monitored and adjusted by Hubei Hospital of TCM from the general perspective of the cohort study. Therefore, each clinical center is required to input the information into the database in time. The recruitment goals will be monitored every 3 months.
Expected component ratio of patients enrolled in the HSPN cohort study.

HSPN: Henoch-Schönlein purpura nephritis; UTP: urinary total protein.
Components of TCM syndrome elements of HSPN
The common syndrome types of HSPN are as follows:
Blood collateral injured by heat pathogen syndrome;
Deficiency of Qi of the spleen and kidney syndrome;
Deficiency of Qi and Yin syndrome;
Deficiency of Yin of the liver and kidney syndrome;
Deficiency of Yang of the spleen and kidney syndrome;
Damp-heat syndrome;
Blood stasis syndrome.
Relative to syndrome, Chinese medicine prescription treatments:
Clearing the nutritive stage decoction or Rhinoceros horn and Rehmannia decoction;
Four-noble drug decoction or restoring the spleen decoction;
Ginseng–Astragalus and Rehmannia decoction;
Anemarrhena–Phellodendron and Rehmannia decoction;
Strengthening Yang, tonifying the middle and replenishing Qi decoction;
Three-nut decoction or
Screening and enrollment
Screening visit was conducted by nephrologists at each clinical center. This visit was conducted to assess eligibility determination, including inclusion criteria, exclusion criteria, and compliance. After providing the informed consent, the eligible participant was included.
Inclusion and exclusion criteria
Participants must meet the following five criteria to be eligible for the enrollment. The details of the inclusion criteria are listed in Table 2. Participants who meet any of the exclusion criteria should be excluded. The details of the exclusion criteria are listed in Table 3.
Inclusion criteria in the HSPN cohort study.

HSPN: Henoch-Schönlein purpura nephritis; UTP: urinary total protein; eGFR: estimated glomerular filtration rate; MDRD: modification of diet in renal disease; TCM: traditional Chinese medicine.
Exclusion criteria in the HSPN cohort study.

HSPN: Henoch-Schönlein purpura nephritis; NYHA: New York Heart Association.
Baseline and follow-up visit
The baseline visit includes the following items: detailed contact information; general information and healthcare services; medical and family history; personal renal history; behavioral habits, chronic disease and risk factors, and questionnaires that concern quality of life, depressive and anxiety symptoms, mental state, physical activity, and the TCM syndrome scale (scored by four grades: “none, mild, moderate, and severe”). Anthropometric measurements (height, weight, waist circumference, hip circumference, resting blood pressure, and pulse) will be made. Laboratory parameters (routine blood test, routine urine test and urine dregs, 24-h UTP, renal function, and electrocardiogram) will be evaluated for each participant. Among these items, 24-h UTP and renal function will be measured in the central laboratory to avoid the variation of testing values between laboratories.
There will be two types of patient follow-ups: clinical follow-up, and quarterly and annual visit. After the baseline visit, participants will receive follow-up visits and be evaluated quarterly and annually. The evaluation items for these follow-up visits will be similar to that for the baseline visit, sample collection will be performed, and the detailed content of each item will be adjusted quarterly and annually according to the research protocol.
Enrollment and baseline data collection was started in August 2016. At present (7 July 2017), 60 participants have completed the screening visits, among which 40 participants were found to be eligible and 38 participants have completed their baseline visit (Figure 2).

Participant recruitment flow diagram.
Prospective cohort study roadmap
Expected study outcomes
The principal clinical outcomes of the study include the summing up of TCM syndrome elements and its related factors of HSPN, and the observation and confirmation of its clinical effect in alleviating the clinical uncomfortable symptoms of HSPN patients, reducing proteinuria and improving renal function (Figure 3).

Prospective cohort study roadmap.
These outcomes will be assessed at 3-month intervals either by phone call or by route clinical visits, depending on the arrangement of each clinical center. Finally, suspected end-point events would be ascertained by an independent end-point assessment committee.
Sample size estimation
The 7th edition cohort study sample size estimation formula for epidemiological studies will be used as reference
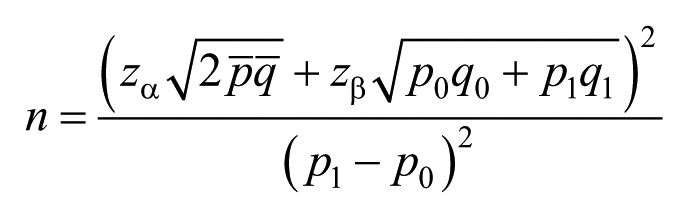
As a result,
Statistics and analysis of data
The baseline data and follow-up data of all participants will be objectively recorded and uploaded to the research commitment unit (Hubei Hospital of TCM) by each research center in time. Finally, the whole data will be statistically analyzed uniformly by the research commitment unit (Hubei Hospital of TCM).
Discussion
In 2011, led by the Nephrology Department of Hubei Hospital of TCM, its members and cooperative units have verified 140 clinical cases of HSPN and developed the TCM diagnosis and treatment scheme and clinical pathway of HSPN, which were more in line with clinical practice and recognized by most experts. 14 This study adopted the latest TCM diagnosis and treatment plan in China. During this study, we will reevaluate and optimize the scheme and further review the composition of TCM syndrome elements of HSPN. The main clinical symptoms of HSPN patients include cutaneous purpura, hematuria, and proteinuria. 15 According to the relevant literature, the treatment of RAS blockers exhibited a limited effect, and the clinical efficacy of the glucocorticoids and immunosuppressants remains controversial. However, involving TCM in the comprehensive prevention and treatment of HSPN has a unique advantage and good application prospect. These expected results will be significant in helping to reduce urinary protein, stabilizing kidney function, and alleviating uncomfortable symptoms. However, the present TCM research outcomes lack high-level evidences to support this. Hence, we conducted the first Chinese population-based national HSPN cohort. This study will provide important evidences for the constitution of TCM syndrome elements and clinical effect of TCM, and valuable information for the clinical guidelines and policies in China, serving as a foundation for future TCM intervention studies in China.
Footnotes
Acknowledgements
We thank the staff of traditional Chinese medicine Clinical Research Bases of Guangdong affiliated to the state administration of TCM; thank expert guidance team and cooperative group members who participate in the HSPN study. The detailed list of the HSPN study group is as follows:
Xiao-qin Wang, Department of Nephrology, Hubei Hospital of Traditional Chinese Medicine, Wuhan, Hubei 430061, China; Kun Bao, Department of Nephrology, Guangdong Hospital of Traditional Chinese Medicine, Guangzhou, Guangdong 440100, China; Li-qun He, Department of Nephrology, Shuguang Hospital affiliated to Shanghai University of Traditional Chinese Medicine, Shanghai, Shanghai 200021, China; Pei-qing Zhang, Department of Nephrology, Heilongjiang Academy Hospital of Traditional Chinese Medicine, Harbin, Heilongjiang 150321, China; Ya-yu Li, Department of Nephrology, Hangzhou Hospital of Traditional Chinese Medicine, Hangzhou, Zhejiang 310007, China; Ying Lu, Department of Nephrology, Tongde Hospital of Zhejiang Province, Hangzhou, Zhejiang 310012, China; Wei Shi, Department of Nephrology, The First Hospital affiliated to Guangxi University of Traditional Chinese Medicine, Nanning, Guangxi 530023, China; Ji-hong Zhan, Department of Nephrology, The First Hospital affiliated to Guiyang College of Traditional Chinese Medicine, Guiyang, Guizhou 550001, China; Fei Xiong, Department of Nephrology, Wuhan Hospital of Traditional Chinese and Western Medicine, Wuhan, Hubei 430022, China; Zhi Gao, Department of Nephrology, Wuhan Hospital of Traditional Chinese Medicine, Wuhan, Hubei 430014, China.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by Research Project for Practice Development of National TCM Clinical Research Bases (Grant/Award Number: JDZX2015194).
