Abstract
The pathogenesis of the pruritus associated with chronic kidney disease-(CKD-aP) is not completely understood. Endocrine, metabolic, neuropathic, and inflammatory disorders were suspected to be the origin of CKD-aP. Based on the hypothesis which suggests that deregulated systemic inflammation may play a crucial role in CKD-a, we investigated the potential relation of an inflammatory monocyte subset (slanDCs) with CKD-aP. Itch questionnaire, visual analogue scale (VAS)-scoring, and Dermatology Life Quality Index (DQLI) were applied for the characterization of itch sensation. VAS-scoring was re-evaluated after 6 months. Monocytes were flow-cytometrically categorized into classical, intermediate, and non-classical subsets. slanDCs are part of the non-classical monocyte subpopulation. Sixty-six hemodialysis patients (CKD5-D) were screened of whom 43 met the study inclusion criteria. In all, 46.5% of patients were scored pruritus-positive (CKD-aP+). CKD-aP severity level of patients was moderate at the start of the study (VAS 5.3 ± 2.5) and remained unchanged after 6 months (VAS: 5.2 ± 1.9, P < 0.757). Thirty percent of patients were affected with mild, 30.0% with moderate, and 35.0% with severe itchiness. In contrast to all other factors tested solely slanDC showed a weak correlation to VAS-score (r = 0.41, P = 0.07). slanDC frequencies between CKD5-D patients with and without itch sensation, however, were not significantly different. Endocrine problems appeared to influence CKD-aP. CKD-aP + patients had significantly higher L-thyroxin supplementation than CKD-aP- (50.0% vs 8.7%, P < 0.005). A binary logistic regression model confirmed the significance of L-thyroxin medication on chronic itch problems of our CKD5-D patients (P < 0.007). There is no clear evidence that slanDCs are related to uremic pruritus. Therefore, other factors underlie the pathophysiology of CKD-aP.
Introduction
The prevalence of chronic kidney disease–associated pruritus (CKD-aP) widely varies ranging from 20% to 87%. The recently performed German Epidemiological Haemodialysis Itch Study (GEHIS) reported a point prevalence of 25.2% and a lifetime prevalence of chronic itch of 35.2%. Although the CKD-aP prevalence seems to be decreasing in the recent years the symptom nevertheless has a considerable impact on quality of life. 1 The causes leading to CKD-aP remain obscure. 2 Neuropathic, endocrine, metabolic or inflammatory disorders were suspected to be causative for the disease.3–5 While some authors find high creatinine, calcium, phosphorous or thyroid hormone levels being associated with pruritus,6,7 others consider CKD-aP a systemic immune disorder (‘immuno-hypothesis’), characterized by activated T-cells that produce an inflammatory Th1-cytokine milieu. 8 Recently, Hänsel et al. 9 raised the concept that 6-sulpho LacNAc dendritic cells (slanDCs) are proinflammatory cells which are pathophysiologically involved in psoriasis by driving strong Th1 and Th17 T-cell responses. slanDCs are a dendritic cell population in human blood, belonging to the CD14 + CD16 ++ monocyte subset which is significantly elevated in chronic kidney disease. The high variability of these cells (20%–80% within CD14 + CD16 ++) and the involvement in skin diseases prompted us to speculate that CKD-aP patients may relate to high slanDC levels.
Methods
Subjects
Sixty-six patients were screened for uremic pruritus at the nephrology outpatient dialysis of the Department of Internal Medicine II of the University Halle-Wittenberg. Inclusion criteria implied age > 18 years and a history of hemodialysis treatment > 12 weeks. Five patients starting dialysis with PD, switched to HD in the course of their dialysis history. Hemodialysis was performed thrice weekly using bicarbonate dialysate and polyamide or polysulfone dialyzers. Patients with a history of other dermatologic diseases (n = 3), acute infection (C-reactive protein (CRP) > 50 mg/l; n = 1), immunosuppressive, and/or glucocorticoid therapy (n = 17) or dialysis vintage < 3 months (n = 2) were excluded. The study was conducted according to the Declaration of Helsinki. Written informed consent was obtained from all study subjects and the study protocol was approved by the local ethics committee.
Assessing chronic kidney disease-associated pruritus
For acquisition of data on uremic pruritus a questionnaire adapted from the ‘Eppendorf Itch Questionnaire’ was applied. 10 The self-reported assessment included questions about the frequency (Is the itching ‘sporadical’, or ‘periodical’, or ‘permanent’?), the duration ( ‘how long does the itching generally last’: ‘seconds’, ‘minutes’, or ‘is there a constant itching?’), and the localization of the itch ( ‘extremities’, ‘torso’, or ‘both’).
The intensity of uremic itching was determined by using the visual analogue scale (VAS) 7 which allowed categorization of itching qualities on a scale reaching from 0 to 10 (0 = no itching; 0 < 4: mild; ⩾ 4–< 7: moderate; ⩾ 7–< 9: severe; ⩾ 9–10 = very severe itching). 11 VAS scoring was applied at study start and repeated after 6 months. The quality of life was assessed using the Dermatology Life Quality Index (DLQI). 12
Quantification of monocyte subpopulations using flow-cytometry
Monocytes were flow-cytometrically (MACSQuant analyzer, Miltenyi Biotec, Bergisch-Gladbach, Germany) analysed from freshly drawn samples anti-coagulated with ethylenediaminetetraacetic acid (EDTA). Samples were processed due to a ‘lyse no wash’ procedure at study start. In short, 50 µl blood was stained with antibodies for 15 min in the dark. Erythrocytes were lysed by addition of 600 µl BD lysing solution (BD Biosciences). Samples were incubated for 15 min, followed by dilution to 1 ml with buffer (0.2% BSA, 2 mM EDTA, 0.07% NaN3). The following antibodies were used: anti-CD45 (clone 5B1), -CD86 (clone IT2.2), -CD16 (clone 3G8, all BD Biosciences, Heidelberg, Germany), -CD14 (clone 61D3, eBioscience, Frankfurt, Germany), -slanDC (clone DD-1) and FcR-Block (both Miltenyi Biotec). Anti-IgM antibody (Miltenyi Biotec) was used as isotype-control. Monocytes defined as CD45 and CD86 positive were divided into subpopulations according to their expression of CD14 and CD16 (classical, CD14++CD16-, intermediate, CD14++CD16+ and non-classical, CD14+CD16+). Data were analysed using MACSQuant software (Miltenyi Biotec) (Supplementary Data, Figure S1).
Laboratory parameters
High sensitive CRP (hsCRP), albumin, urea, creatinine, HbA1c, calcium, phosphorous, parathyroid hormone (PTH), thyroid stimulating hormone (TSH), free tri-iodthyronine (fT3), and free thyroxine (fT4) were analysed by the central laboratory of the University Hospital Halle (Saale) using certified, standardized methods.
Data analysis
Data were presented as mean ± SD if not otherwise indicated. Data were compared by chi-square test, Student’s t-test, Mann–Whitney-U Test or one way analysis of variance (ANOVA) as appropriate. Relationships between data were examined by Spearman rank correlation. A binary logistic regression model was assessed (backward logistic regression model) which included the prevalence of pruritus as independent variable and frequency of slanDC as well as the calcium phosphorous product (Ca x P), the PTH level and L-thyroxin medication as independent variables. The good fit of the model was assessed by the Hosmer-Lemeshow test.
All calculations were carried out using the SPSS 21.0 (SPSS Inc., Chicago, USA) or GraphPad Prism 6.0 statistics software (GraphPad Software Inc., La Jolla, USA). The level of significance was set at P < 0.05. The initial statistical power calculation was done by G*Power (http://psycho.Uni-duesseldorf.de/abteilungen/aap/gpower3). As analysed by G*Power calculations of slanDC frequencies (t-test, difference between two independent means) an actual power of 0.95 (the α-error probability 0.05, effect size d = 1.26) will be reached if 34 patients (17 CKD-aP+ and17 CKD-aP-) are included in the study.
Results
The study included 43 haemodialysis patients with a mean age of 63.4 ± 15.2 years. In all, 37.2% were female and 46.5% were diabetics. Twenty patients (46.5%) were scored pruritus-positive according to the oral interview. The clinical baseline characteristics between both groups were not different (Table 1). However, we found notably CKD-aP+ patients to be afflicted with hypothyroidism and L-thyroxin medication was accordingly significantly higher in these patients (Table 2), albeit thyroid parameters (CKD-aP+ vs CKD-aP-: TSH (mU/l): 2.3 ± 2.3 vs 2.4 ± 4.1, P = 0.930; fT3 (pmol/l): 3.7 ± 0.6 vs 3.7 ± 0.7; P = 0.874; fT4 (pmol/l): 15.0 ± 2.8 vs 16.0 ± 3.5; P = 0.274) nor parathomone levels (PTH (pmol/l): 21.1 ± 19.6 vs 18.0 ± 12.6; P = 0.523) were not significantly different between both study cohorts.
Relationship between pruritus and clinical parameters.

BMI: body mass index; CRP: C-reactive protein; eGFR: estimated glomerular filtration rate; PTH: parathyroid hormone; TSH: thyroid stimulating hormone.
Medication.

Regarding the frequency of itching periods, 5 patients (25.0%) reported to suffer from sporadic itching pain, 9 patients (45.0%) from periodical, and 6 (30.0%) from permanent uremic associated pruritus. Regarding the duration of itching symptoms most uremic patients complained about itching stimuli lasting a few minutes (n = 11, 55.0%). Five patients (25.0%) had itching problems for a few hours and 4 (22.0%) felt exposed to constant itching. The itching symptoms were located at the extremities or torso in 4 patients each, the majority (60%) felt generalized itching. Seven patients reported that the itch was moving around the body while the rest had the itch sensation at a defined site of the body. The VAS scoring demonstrated a moderate itching intensity of CKD5-D patients (VAS1: 5.3 ± 2.5).
Although there is currently no proven treatment therapy, most patients were offered treatment with emollients. As described in other studies, the skin of our patients was primarily unchanged and primary skin lesions were not observed. The DLQI demonstrated that for about 45.0% of the patients itch means no restriction in life quality; 35% complained about a small, 15.0% about a moderate and 5.0% about a very large restriction in life quality. VAS and DLQI significantly correlated (r = 0.506, P = 0.023) implying that VAS score translates into gradual quality of life restriction. Validation of CKD-aP data was done by VAS scoring after 6 months without demonstration of significant disease improvement (VAS2: 5.2 ± 1.9 vs VAS1, P < 0.757), although a slight switch from people feeling bothered by severe towards moderate CKD-aP (35% vs 10%) could be observed (Table 3).
VAS scoring at study entry (VAS1) and after 6 month (VAS2).

VAS: visual analogue scale.
Regarding the pathophysiology of CKD-aP we tried to extend the ‘immuno-hypothesis’ by assigning monocyte derived DC precursor cells an important role in the setting of CKD-aP. As demonstrated in a pilot experiment (Supplementary Data, Figure S2) slanDCs were two- to threefold elevated in CKD5-D versus healthy control subjects, showing a kind of specificity of this inflammatory cell type for CKD. In contrast to other disputed factors playing a putative role in CKD-aP, that is, Ca x P product or parathormone, slanDC showed a weak positive correlation to VAS (Table 4). Stratifying CKD-aP patients (n = 10, each) according to VAS < 5 and VAS ⩾ 5, we dichotomized patients with milder versus stronger itch sensation. The ‘high VAS-group’ showed the greatest slanDC level (VAS < 5: 44.6 ± 15.8% vs VAS ⩾ 5: 58.6 ± 13.2%, \ < 0.06); but taking slanDC-frequencies of CKD5-D without pruritus (54.6 ± 15.8%) into account, our hypothesis seems to be questionable (Figure 1). Furthermore, in contrast to slanDC or any other laboratory data binary logistic regression analysis revealed that drug intake, that is, L-thyroxin supplementation, had a significant impact on the prevalence of pruritus (Table 5). Therefore, adverse drug effects may superimpose the pathophysiological mechanism(s) of CKD-aP.
Relationship between VAS and clinical and immunological data.
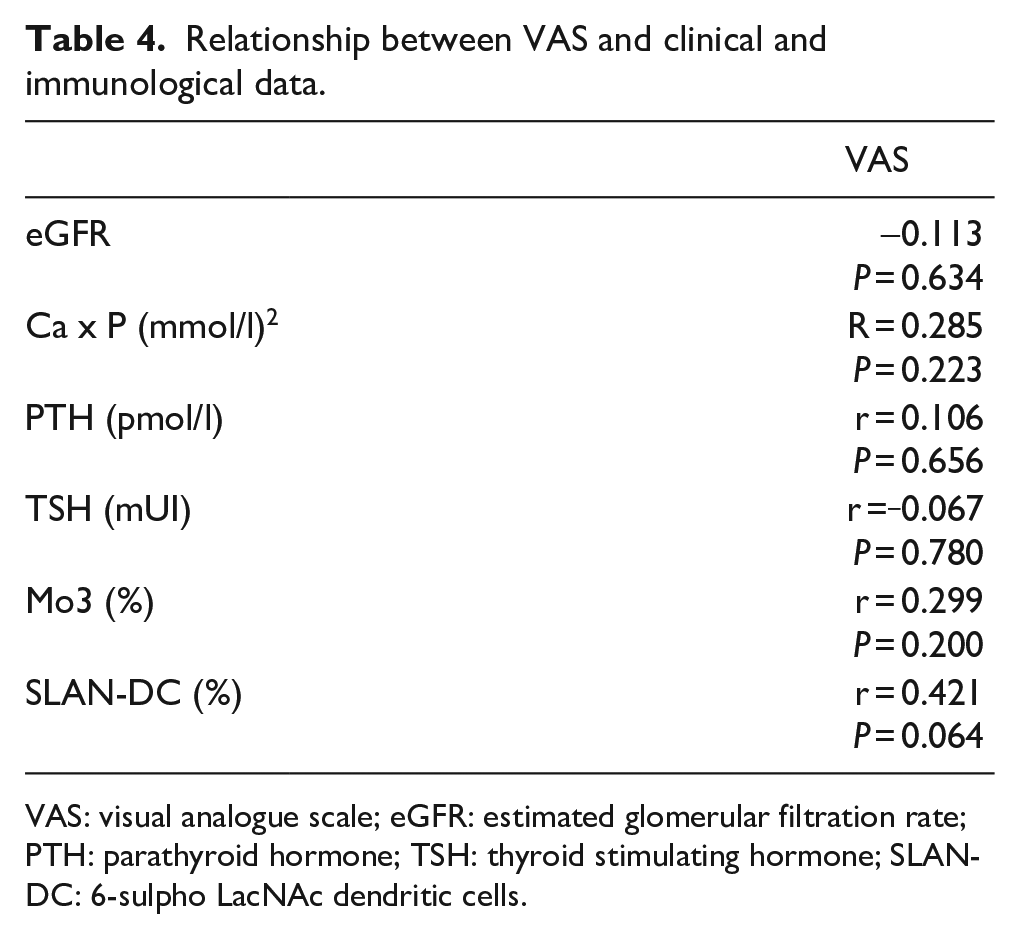
VAS: visual analogue scale; eGFR: estimated glomerular filtration rate; PTH: parathyroid hormone; TSH: thyroid stimulating hormone; SLAN-DC: 6-sulpho LacNAc dendritic cells.
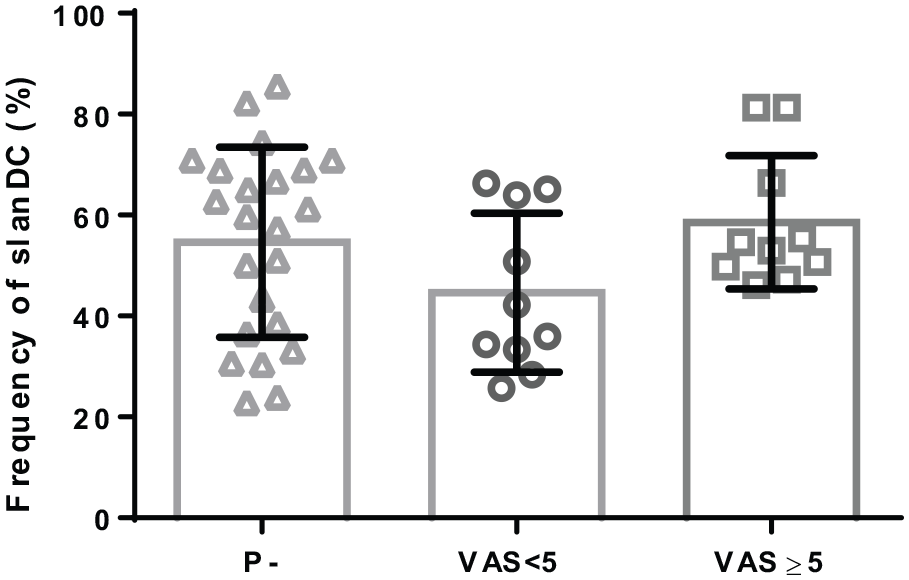
Frequency of slanDC + cells in patients without CKD-aP (P-) and CKKD-aP categorized patients according the visual analogue scale < 5 (VAS < 5) and ⩾ 5 (VAS ⩾ 5).
Binary logistic regression model.

The association between the incidence of CKD-aP and slanDC was investigated by using binary logistic regression analysis (backward stepwise logistic regression model). Variables included in step 1 were % slanDC, L-thyroxin-supplementation, calcium-phosphorous product (Ca x P) and parathyroid hormone level (PTH). PTH was eliminated at step 1, Ca x P at step 2 and slanDC at step 3 of the analysis.
Discussion
Pruritus is common among people on HD, 13 and it is a distressing problem which is difficult to diagnose. Although the incidence of CKD-AP declined in the last years, about 40% to 50% of CKD5-D patient remain afflicted with. 14 This is in the line with our study where we find about 47% of the patients being bothered with CKD-aP and categorized by moderate itch sensation. The pathophysiological mechanism of uremia-related pruritus is poorly understood. Following the hypothesis that derangement of the immune system plays an important role at the appearance of CKD-AP several groups postulated T-cell involvement in CKD-AP8,15 resulting in Th1 differentiation and a proinflammatory milieu, including CRP and interleukin (IL)-6 elevation. 8 Indeed, tacrolimus 16 and thalidomide treatment 17 –both therapies are known to decrease the production of proinflammatory cytokines–improved CKD-aP to some degree. We focused on monocyte derived cells, namely slanDC cells which are elevated in CKD-5D patients. Recently, slanDC were found to be a part of dermal dendritic cells, 18 playing a role in psoriatic skin disease 19 we therefore speculated that these cells could be involved in CKD-aP. Pruritus stratification of patients according to chart review and/or self-report did not result in different slanDC frequencies between P- and P+ patients. In contrast, scoring of CKD-aP by the visual analogue scale resulted in a weak positive relationship between VAS and slanDC frequency. That means that patients with VAS score >= 5 have significantly higher slanDC than those patients with VAS < 5. However, as the patients with VAS >= 5 do not have significant higher slanDC cells in comparison to patients diagnosed pruritus negative, there is no evidence for an association between these parameters.
Recently gabapentin, a structural analogue of the neurotransmitter gamma-aminobutyric acid (GABA) was reported to have anti-pruritic effects. 20 This observation links CKD-aP to neuropathology. Taking in mind that GABA may also be involved in the regulation of inflammatory cytokines from mononuclear cells, 21 it is quite reasonable that inflammation and CKD-aP are at least partially influenced by neurotransmitters.
End stage renal failure also affects parathyroid and thyroid functions. While parathyroid hormone level is elevated, both plasma tri-iodothyronine (fT3) and thyroxine (fT4) are often reduced in hemodialysis patients. 22 On the one hand parathyroid hormone is suspected to be in involved in uremic pruritus–but as parathormone did not elicit any cutaneous reaction upon intradermal application, the crucial proof for a causal relationship between itching sensation and parathormone levels is lacking. On the other hand, it is interesting to note that a high number of chronic pruritus patients (50%) had to be treated with L-thyroxin for normalization of thyroid function compared to CKD-aP- patients. But as far as we know, pruritus related to hyperthyroidism is a quite rare feature. Therefore, it is not clear at the moment to what extent the endocrine disorder itself contributes to uremic pruritus. However, the binary regression analysis clearly proves that L-thyroxin medication has a significant impact on CKD-aP. Therefore, adverse drug effects add to the uremic itch problem in end stage renal failure, making it difficult to identify the main pathophysiological mechanism of CKD-aP. Most probably the pathogenesis of CKD-aP is multi causal fostered by the inflammatory setting in end-stage renal failure.
Study limitations
The small sample size may be regarded as drawback of our study, but compared to most other CKD-aP relevant topics, we provide data about two VAS analyses, demonstrating no significant change between both measurements. Due to the undulating pattern of CKD-aP,23,24 however, a two point measurement may still be not enough to characterize chronic pruritus. The positive correlation between VAS and frequency of slanDC may pretend some association between CKD-aP and monocytes which can be questioned when the level of slanDCs between CKD5-D with and without pruritus is compared. Besides, the final proof, detection of slanDC in skin specimen of CKD-aP patients, is pending. This effort, however, is difficult to implement as the skin of our patient was predominantly unchanged and primary skin lesions were not observed.
Supplemental Material
Ulrich_et_al_SLAN_Pruritus_20181119_European_Journal_of_Inflammation_supplementary_data – Supplemental material for No significant relation of proinflammatory slanDCs with uremic pruritus
Supplemental material, Ulrich_et_al_SLAN_Pruritus_20181119_European_Journal_of_Inflammation_supplementary_data for No significant relation of proinflammatory slanDCs with uremic pruritus by Christof Ulrich, Anja Leonhardt, Bogusz Trojanowicz, Eric Seibert, Roman Fiedler and Matthias Girndt in European Journal of Inflammation
Footnotes
Acknowledgements
We appreciate the support of Mrs. M. Hajri and the staff of the KfH Nierenzentrum, Halle. C.U., M.G. conducted the study, reviewed the literature, drafted the manuscript, and performed the statistical analysis. A.L. and R.F. organized the sample collection. A.L. measured slanDCs by flow cytometry. B.T. and E.S. reviewed the literature and helped to draft the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported solely by intramural research grants.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
