Abstract
Green tea is one of the most popular non-alcoholic beverage crops worldwide. Leaf extracts of the green tea, Camellia sinensis, are known to have natural anti-oxidant and anti-inflammatory effects. However, relatively few studies have focused on the biological activities of green tea petiole (GTP) (the stalk that attaches the leaf blade to the stem, currently considered waste material) compared with other parts of the tree, such as the leaf, root, and flower. Here, we report the first investigation of the anti-inflammatory effect of GTP extract against poly(I:C)–stimulated human keratinocytes (a model of skin inflammatory conditions) and EpiDerm (a three-dimensional skin equivalent). Our results reveal that GTP extract suppresses the induction of the messenger ribonucleic acids (mRNAs) encoding pro-inflammatory cytokines (tumor necrosis factor alpha (TNF-α), Interleukin 1 beta (IL-1β), and Interleukin-8 (IL-8)) and matrix metalloproteinase-1 (MMP-1). These findings indicate that GTP extract could prove useful in protecting skin cells from inflammatory conditions and skin aging.
Introduction
Green tea beverage, which is produced from the leaves of Camellia sinensis, is one of the most widely consumed drinks worldwide owing to its broad range of biological functions, which include anti-inflammatory effects. Green tea contains polyphenolic ingredients, such as epicatechin (EC), epicatechin gallate (ECG), epigallocatechin (EGC), and epigallocatechin-3-gallate (EGCG). 1 EGCG is the major ingredient that exerts an anti-inflammatory effect; it acts by inhibiting NF-κB activation. 2
The members of the toll-like receptor (TLR) glycoprotein family function as surface transmembrane receptors and play indispensable roles in defending the host against infection by recognizing pathogen-associated molecular patterns (PAMPs) in the innate immune system. 3 Among the TLRs, TLR3 recognizes double-stranded ribonucleic acid (RNA), such as poly(I:C), and induces the MyD88-independent activation of NF-κB 4 to induce inflammatory cytokines and chemokines. 5
In both intrinsically and photo-aged skin, dermal collagen damage is mediated by increased expression of matrix metalloproteinase-1 (MMP-1), which is a matrix-degrading enzyme known to be important for inflammation.6,7 Recently, Lee et al. 6 reported that activation of TLR3 by the poly(I:C) ligand induces MMP-1 via NF-κB signaling in human epidermal keratinocytes.
The potential beneficial effects of green tea petiole (GTP) (the stalk part of the green tea tree) have not been explored, and this portion of the plant is currently considered waste. Here, we demonstrate for the first time that GTP extracts suppress the poly(I:C)–induced up-regulations of pro-inflammatory cytokines and MMP-1 in human epidermal keratinocytes and a three-dimensional (3D) human skin equivalent. Our findings provide the first indication that GTP extracts could have potential therapeutic anti-inflammatory and anti-aging effects on skin.
Results
Prior to evaluating the anti-inflammatory efficacy of GTP extract, we used high-performance liquid chromatography (HPLC) to analyze their major components (Figure 1). HPLC analysis of catechins revealed that the chromatogram pattern of GTP extract differed from that of green tea leaf extract, suggesting that other ingredient components rather than catechins constitute the extract compared with regular green tea leaves extract (Supplemental Figure S1). Notably, the level of EGCG was found to be much lower in GTP extract than in green tea leaves extract. Next, we examined the cytotoxicity of different doses of GTP extract in human epidermal keratinocytes, but found no evidence of GTP-related cytotoxicity at the tested doses (Figure 2).
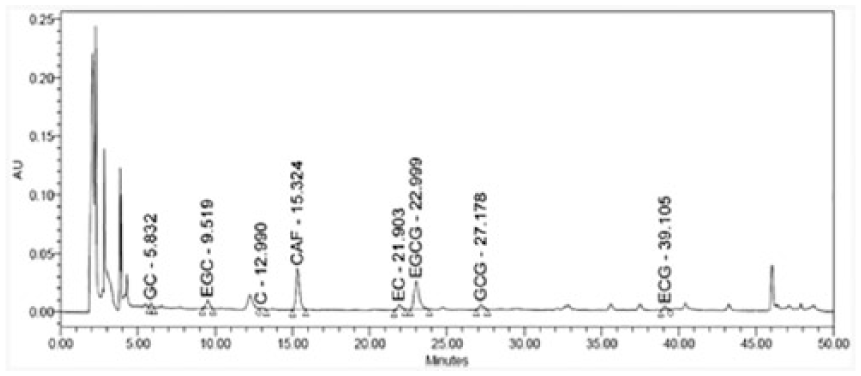
HPLC catechin analysis of GTP extracts.

Cell viability was measured with the MTT assay. Cells were incubated with GTP at 10 µg/mL for 24 h. MTT assay was performed as described in Materials and methods (Supplemental Material).
As age-related alterations in TLR and/or TLR ligand expression levels have recently been highlighted as a probable cause of inflammatory imbalance, 6 we examined the effect of GTP extract on TLR signaling in the context of skin inflammation. We induced the expression of ligands for TLR2, TLR3, and TLR5 (Pam3CSK4, poly(I:C), and flagellin, respectively) to mimic the conditions of inflammation induced by TLR signaling, and examined the up-regulation of the messenger ribonucleic acid (mRNA) encoding matrix metalloproteinase-9 (MMP-9). We found that poly(I:C) (10 µg/mL) more effectively induced MMP-9 mRNA expression in human epidermal keratinocytes, compared with the other TLR ligands tested (Supplemental Figure S2(a)); we thus used this ligand for subsequent experiments. To test the effect of GTP extract in our model system of inflammation, we pretreated human epidermal keratinocytes with 10 µg/mL GTP extract for 30 min, stimulated them with 10 µg/mL poly(I:C) for 24 h, and then used reverse transcription polymerase chain reaction (RT-PCR) to assess the cellular mRNA level of MMP-9. The induction of MMP-9 by poly(I:C) was reduced to ~50% when human epidermal keratinocytes were pretreated with GTP extracts (Supplemental Figure S2(b)). This suggests that GTP extract are likely to inhibit TLR signaling in human epidermal keratinocytes.
To further investigate whether GTP extract affects TLR-mediated inflammation, we analyzed the mRNA levels of the pro-inflammatory cytokines, tumor necrosis factor alpha (TNF-α), Interleukin 1 beta (IL-1β), and Interleukin-8 (IL-8). The activation of TLR3 by poly(I:C) has been reported to induce pro-inflammatory responses in human epidermal keratinocytes and an airway epithelial model.8–10 Here, we treated human epidermal keratinocytes with 10 µg/mL poly(I:C) for 24 h in culture, extracted total RNA, and used quantitative RT-PCR to measure the mRNA levels of TNF-α, IL-1β, and IL-8. Our results revealed that poly(I:C) dramatically increased the mRNA expression levels of TNF-α, IL-1β, and IL-8, but this effect was significantly and dose-dependently suppressed by GTP extract (Figure 3(a)–(c)). This suggests that the GTP extract-mediated inhibition of TLR could suppress inflammatory responses in human epidermal keratinocytes.

GTP extracts suppress poly(I:C)–induced inflammatory cytokines in human epidermal keratinocytes. Cells were stimulated with 10 µg/mL poly(I:C) and the indicated doses of GTP extracts for 24 h. The relative transcript levels of (a) TNF-α, (b) IL-1β, and (c) IL-8 were determined via real-time polymerase chain reaction (RT-PCR) analysis and plotted in relation to GAPDH endogenous levels.
Previous studies have demonstrated that poly(I:C) up-regulates MMP-1 and MMP-9 via TLR3 activation in many cell types, including human epidermal keratinocytes.6,11 To explore the effects of GTP extract on the induction of MMP-1 by poly(I:C), we pretreated human keratinocytes with GTP extract for 30 min, co-incubated the cultures with poly(I:C) and GTP extract for 24 h, and used RT-PCR to examine the mRNA expression of MMP-1. We found that the poly(I:C)–induced up-regulation of the MMP-1 mRNA was significantly and dose-dependently inhibited by GTP extract (Figure 4(a)). We used enzyme-linked immunosorbent assay (ELISA) to examine the secretion of MMP-1 in human epidermal keratinocytes co-incubated for 2 days with poly(I:C) after the pretreatment of GTP extract for 30 min, and found that GTP extract dramatically decreased the poly(I:C)–induced secretion of MMP-1 (Figure 4(b)). Notably, the mRNA and protein levels of MMP-1 dropped below their basal expression levels following treatment with GTP extract. To further clarify the inhibitory effect of GTP extract on MMP-1 secretion, we applied the extract to EpiDerm (a reconstituted 3D human skin equivalent) in the presence or absence of poly(I:C) for 9 days, and then performed hematoxylin and eosin (H&E) staining and immunohistochemistry with an antibody against MMP-1. H&E staining did not reveal any poly(I:C)– and/or GTP extract–related structural changes in the epidermis (Figure 4(c)). MMP-1 expression (Figure 4(c), brown stain) was induced by poly(I:C); it was detected in the epidermis as cytosolic staining of keratinocytes and in the dermis as a secreted protein. Treatment with GTP extract markedly reduced the poly(I:C)–induced MMP-1 expression level to the point that it was comparable to the level seen in the negative-control 3D human skin equivalent.

GTP extracts inhibit poly(I:C)–induced MMP-1 expression in human epidermal keratinocytes and 3D human skin equivalent. (a) Cells were stimulated with 10 µg/mL poly(I:C) and the indicated doses of GTP extracts for 24 h. The bars represent MMP-1 mRNA in human epidermal keratinocytes. (b) Cells were stimulated with 10 µg/mL poly(I:C) and the indicated doses of GTP extracts for 48 h. MMP-1 secretion was detected using the ELISA kit as described in Materials and methods (Supplemental Material). (c) The inflammatory response using poly(I:C)–treated 3D human skin equivalent was developed as described in Materials and methods (Supplemental Material). H&E staining shows the morphological status of human skin equivalent. In addition, MMP-1 in the dermis was stained as described in Materials and methods (Supplemental Material) (brown color) (bar length = 50 µm).
Discussion
We herein provide the first evidence showing that GTP extract suppresses the inflammatory responses induced by poly(I:C)–activated TLRs, and may have anti-inflammatory and anti-aging effects in skin. Future work is needed to identify the active constituents that drive these effects of GTP extract, but the results of our HPLC analysis suggest that catechins do not constitute the major active molecules. Instead, other active components (e.g. glycoproteins) are likely to be involved.
Previous studies showed that the expression and function of TLRs are impaired in aged tissue.6,12 Thus, it may be beneficial to maintain an adequate balance of TLR signaling as the skin experiences aging. Based on our present results, we propose that GTP extract can effectively inhibit abnormally elevated TLR activities under specific conditions. Various pathways may be involved. For example, GTP extract may suppress the activation of NF-κB (a major inflammatory regulator) in poly(I:C)–stimulated keratinocytes. 6 Further studies are needed to establish the action mechanism of GTP.
Skin aging is a complicated process that involves functional and structural alterations. Both extrinsic and intrinsic skin-aging processes lead to decreases in the collagen content and skin thickness; this is largely related to the activation of MMPs, such as MMP-1, which triggers collagen degradation. 7 In our experimental setting, poly(I:C) stimulation increased MMP-1 production and secretion in keratinocytes and a human 3D skin equivalent, but these increases were markedly inhibited by GTP extract. These findings support the idea that GTP extract could represent promising agents for the prevention of skin aging.
In sum, our results collectively show that GTP extract inhibits inflammatory responses and MMP-1 expression in poly(I:C)–stimulated keratinocytes, supporting the potential utility of this material as a unique anti-inflammatory and anti-aging agent for skin.
Supplemental Material
Supplementary_Figures_(Park_et_al_20190507) – Supplemental material for Anti-inflammatory effect of green tea petiole extracts in poly(I:C)–stimulated human epidermal keratinocytes and a human 3D skin equivalent
Supplemental material, Supplementary_Figures_(Park_et_al_20190507) for Anti-inflammatory effect of green tea petiole extracts in poly(I:C)–stimulated human epidermal keratinocytes and a human 3D skin equivalent by Nok Hyun Park, Il-Hong Bae, Seonsoo Han, Minkee Kim, Sung Hoon Lee, Won Seok Park, Chang Seok Lee and Jae Sung Hwang in European Journal of Inflammation
Supplemental Material
Supplementary_Material_and_Methods_(Park_et_al_20190507) – Supplemental material for Anti-inflammatory effect of green tea petiole extracts in poly(I:C)–stimulated human epidermal keratinocytes and a human 3D skin equivalent
Supplemental material, Supplementary_Material_and_Methods_(Park_et_al_20190507) for Anti-inflammatory effect of green tea petiole extracts in poly(I:C)–stimulated human epidermal keratinocytes and a human 3D skin equivalent by Nok Hyun Park, Il-Hong Bae, Seonsoo Han, Minkee Kim, Sung Hoon Lee, Won Seok Park, Chang Seok Lee and Jae Sung Hwang in European Journal of Inflammation
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
