Abstract
To compare growth and glycemic control in children with type 1 diabetes and silent celiac disease treated with a gluten-free diet for 1 year with those of similar age and gender with type 1 diabetes but without celiac disease, 16 type 1 diabetes patients with silent celiac disease were enrolled and each celiac disease-positive case was matched for age, sex, and duration of diabetes with two type 1 diabetes controls with negative serologic markers of celiac disease. All 16 children with positive celiac disease serology had histologic features consistent with celiac disease despite the absence of symptoms. The mean growth and metabolic control values were similar between children with type 1 diabetes and celiac disease and those with type 1 diabetes but without celiac disease. This study seems to suggest that the early diagnosis of celiac disease and initiation of a gluten-free diet may prevent further deterioration in the nutritional status of children with type 1 diabetes and celiac disease and may reduce the prospect of celiac disease complications without any impact on type 1 diabetes control.
Background
In children with type 1 diabetes (T1D), celiac disease (CD) is diagnosed more frequently than in the general population. 1 As the development of T1D usually precedes symptomatic CD, some guidelines recommend that systematic screening for CD should be periodically performed in all children with T1D, and, in positive cases, a gluten-free diet (GFD) should be initiated. 2 However, both the systematic screening and the GFD have been debated by several experts. 3 Most of the CD cases in T1D patients are silent has recently been reported that a portion of these patients tends to have CD serology that normalizes and does not develop the overt disease. 4 Moreover, children with T1D frequently have relevant eating problems, 5 which predispose them to poor adherence to the GFD. 6 Finally, the influence of a GFD on growth and metabolic control in children with T1D is uncertain. In some previous studies, the effect of a GFD on symptoms, growth, and metabolic control has been investigated, but with inconsistent results.6–10 The main aim of this study was to compare growth and glycemic control in children with T1D and asymptomatic CD treated with a GFD with those in controls of a similar age and gender with T1D but without CD in order to assess the importance of a systematic screening for CD in all the patients with T1D and the impact of GFD on patients with both diseases.
Methods
Study design
This was a case-control study in which a total of 48 T1D patients were enrolled, 32 without any evidence of CD, and 16 with silent CD, defined as a condition in which patients do not complain of any symptoms, but still experience villous atrophy damage to their small intestine. In the Regional Center for Children with T1D at the Pediatric Clinic of the Università degli Studi di Perugia, Perugia, Italy, all of the children with T1D underwent antibody screening for CD beginning at the first year of diagnosis to examine the dosage of IgA-anti-tissue transglutaminase (anti-tTG) antibodies and anti-endomysial antibodies (EMA) by immunoassay. Those positive for anti-tTG (tTG ⩾ 2.0 U/mL) and/or EMA test underwent a duodenal–jejunal biopsy, even if asymptomatic. Patients with histologic features of CD (i.e. evidence of villous atrophy with crypt hyperplasia and an increase in intraepithelial lymphocytes) started a GFD. All T1D patients were monitored with clinic visits and glucose metabolism evaluation once every 3 months, whereas the anti-tTG and EMA serum concentrations were tested annually. Dietary adherence was assessed at each visit according to the method described by Assor et al. 11 that includes a comprehensive dietary interview with alternating 3-day food records and typical day recalls and the application of the Celiac Dietary Adherence test. In addition, it was indirectly evaluated through the trend of CD serologic markers.
All of the children with T1D and asymptomatic CD were included in the study after CD confirmation by biopsy, and each case was matched for age, sex, and T1D duration with two subjects followed in the same Center who were CD negative. Medical records of all patients were examined, and data for demographic characteristics, body mass index (BMI), BMI standard deviation score (SDS), glycated hemoglobin (HbA1c), and daily insulin dose were extracted during the year before (T0) and the year after (T1) the diagnosis of CD and the beginning of GFD.
Study population
Study population involved 16 case subjects (7 males and 9 females) and 32 control subjects (14 males and 18 females). Mean age at T1D diagnosis was 7.97 years for cases (range: 1–16 years) and 7.91 years for controls (range: 1–15 years). At diagnosis of CD, case subjects had a mean age of 11.13 years (range 5–17 years) and an average duration of T1D of 3.16 years (range 2–6 years).
Assessment of outcome and exposure variables
A comparison was made between the data collected at the time of diagnosis of CD (T0) and the data collected at 1 year after the beginning of the GFD in CD-positive patients or at 1 year from enrollment in the CD-negative ones (T1). The BMI (kg/m2) was calculated from the participant’s weight and height and was then converted into BMI SDS using reference data based on the growth charts of the Italian Society of Pediatric Endocrinology and Diabetology. 12
Ethics
The study was approved by the Ethics Committee of Ospedale Santa Maria della Misericordia, Perugia, Italy. Written informed consent was obtained from either the parent(s) or legal guardian(s) of each study participant, and the children who were aged >8 years signed to confirm their consent.
Statistical analysis
Time-related analysis was performed for each parameter to verify intragroup and intergroup variations. Longitudinal comparison of variables (within-subjects factor) and evaluation of group interactions (between-subjects factor) were performed using analysis of variance (ANOVA) for multiple measures. The chi-square test with Yates correction was used for unrelated categorical data. McNemar’s test was used for related categorical data. Data were expressed as the mean values, and a P value of <0.05 was considered statistically significant.
Results
Among the 16 children with asymptomatic CD, 14 (87.5%) were anti-tTG and EMA-positive, 1 child (6.25%) was anti-tTG-positive only, and 1 (6.25%) was EMA-positive only. All 16 children with positive CD serology showed histologic features consistent with CD (type 2 and type 3 of Marsh classification) despite the absence of clinical symptoms. Among the 16 CD patients, 13 (81.2%) were positive for HLA-DQ2 (all positive for DQA1*05 and DQB1*02), whereas three patients were positive for HLA-DQ8 (all DQB1*02/*02). T1D was diagnosed at a mean age of 7.97 years (range: 1–16 years) in CD patients and at 7.91 years (range: 1–15 years) in controls without CD.
Table 1 shows data regarding BMI, BMI SDS, HbA1c, and total daily insulin dosage measured at enrollment (T0) and after 1 year (T1). The mean values of all of the studied parameters did not significantly change from T0 to T1 in either of the study groups. Moreover, evaluation of glucose metabolism during the study year did not show relevant fluctuations of studied parameters. Finally, neither severe hypoglycemia nor diabetic ketoacidosis occurred in any of the enrolled children.
Body mass index (BMI), BMI standard deviation score (BMI SDS), glycated hemoglobin (HbA1c), and total daily insulin in children with type 1 diabetes (T1D) with or without celiac disease (CD).

BMI, body mass index; BMI SDS, body mass index standard deviation score; CD, celiac disease; HbA1c, glycated hemoglobin; SD, standard deviation; T1D, type 1 diabetes.
Values were obtained at T0 (i.e. CD confirmation with endoscopy in CD-positive patients and enrollment of the matched patients without CD) and after 1 year (i.e. 12 months from the beginning of a gluten-free diet in CD-positive patients or from enrollment in the CD-negative ones).
No other significant differences among CD-negative patients or CD-positive patients from T0 to T1; no significant difference between CD-negative or CD-positive patients.
P < 0.0001 versus BMI in CD-negative patients at T1.
P < 0.0001 versus BMI in CD-positive patients at T1.
Table 2 reports the trend of the anti-tTG antibodies and EMA in children with silent T1D 1 year after beginning the GFD. The data were obtained for only 15 out of the 16 (93.7%) initially enrolled patients because one refused to donate the blood for laboratory tests. The anti-tTG status became negative in 10 out of 13 anti-tTG-positive subjects (66.7%), and the EMA status became negative in 9 out of 14 EMA-positive subjects (60.0%). In children with persistent anti-tTG antibody levels, titers were slightly lower at T1 than at T0. In CD-negative patients, markers of CD were negative both at T0 and at T1.
Trend of IgA-anti-tissue transglutaminase (anti-tTG) antibodies and anti-endomysial antibodies (EMA) in children with type 1 diabetes (T1D) and celiac disease (CD)-positive serology at CD diagnosis by biopsy (T0) and 1 year after beginning a gluten-free diet (T1).
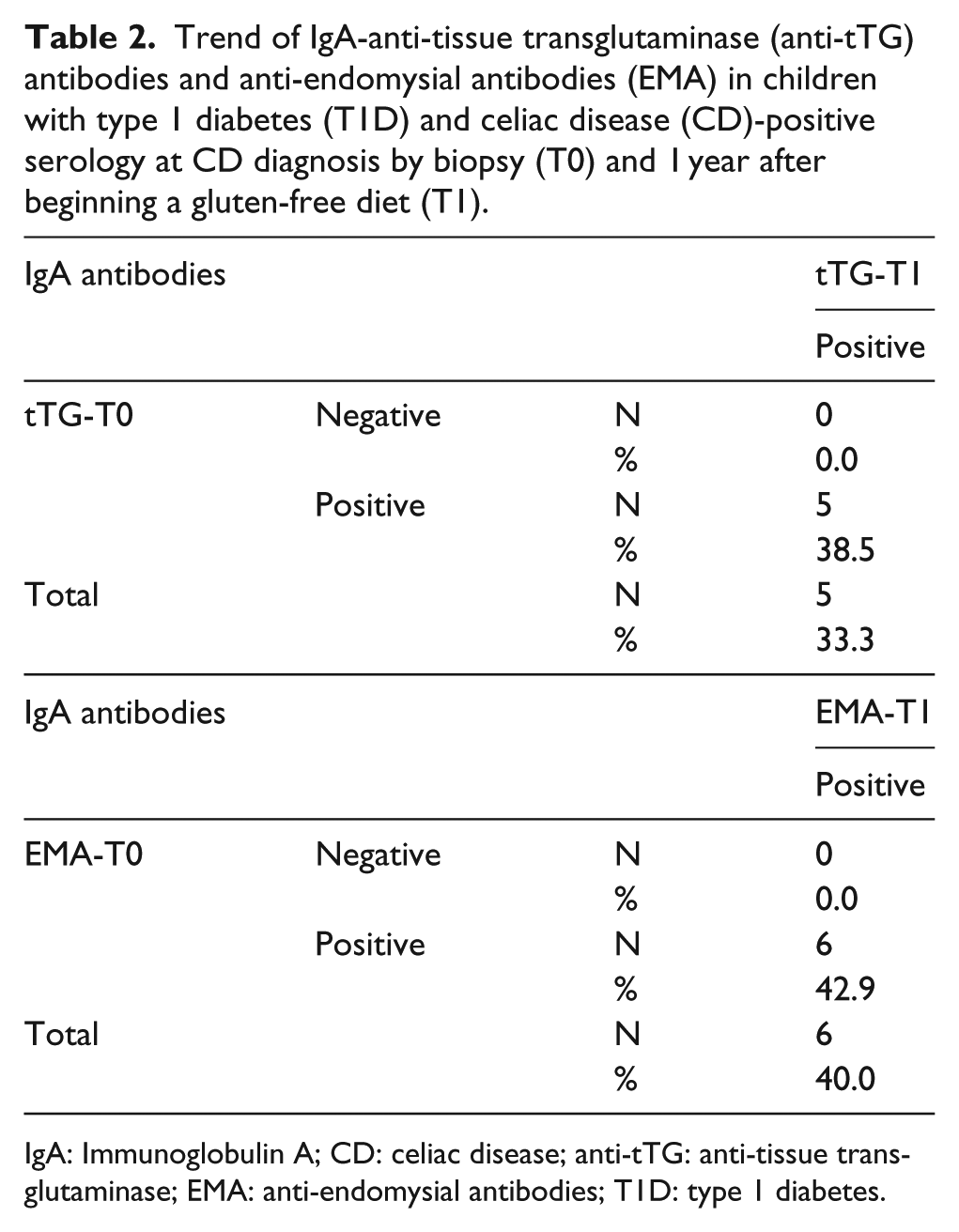
IgA: Immunoglobulin A; CD: celiac disease; anti-tTG: anti-tissue transglutaminase; EMA: anti-endomysial antibodies; T1D: type 1 diabetes.
Discussion
This study seems to suggest that in children with T1D who develop CD, association of both diseases does not affect the nutritional status and growth, at least until CD remains silent. Moreover, it seems to indicate that children with T1D and silent CD who receive GFD have growth quite like patient with T1D but without CD. Finally, GFD was associated with good metabolic control as HbA1c levels and insulin daily dose required to maintain acceptable blood glucose levels did not change during the study period and remained similar to those of diabetic children without CD.
This study has some limitations. The number of enrolled children is relatively small. Duration of follow-up was limited only to a year. Moreover, it is not a randomized controlled trial in which children with T1D and silent CD receiving GFD or normal diet were compared. However, children were carefully monitored and compliance with GFD has been assessed with a very efficient methodology. In agreement with some previous studies,9,10 this study seems to suggest that laboratory screening of CD should be performed in all the children with T1D and, in confirmed cases, GFD should be prescribed as soon as possible. The early diagnosis of CD and initiation of a GFD may prevent further deterioration in the nutritional status of children with T1D and CD and may reduce the prospect of CD complications without having any evident impact on T1D control. These apparent clinical benefits need to be confirmed by larger studies with a longer follow-up.
Footnotes
Acknowledgements
The authors would like to thank WAidid Secretariat Elisabetta Di Felice for her support.
Author contributions
G.M. wrote the first draft of the manuscript; N.P. co-wrote the manuscript; E.S., M.A., F.R., and G.C. participated in patient management; U.G. and M.G.B. critically revised the text; S.E. supervised patient management and made substantial scientific contributions. All authors approved the final version of the manuscript.
Availability of data and material
All included.
Consent for publication
The publication of this study was approved by the Ethics Committee of Ospedale Santa Maria della Misericordia, Perugia, Italy. Written informed consent was obtained from either the parent(s) or legal guardian(s) of each study participant, and the children who were aged >8 years signed to confirm their consent.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
The study was approved by the Ethics Committee of Ospedale Santa Maria della Misericordia, Perugia, Italy. Written informed consent was obtained from either the parent(s) or legal guardian(s) of each study participant, and the children who were aged >8 years signed to confirm their consent.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
