Abstract
Accumulating epidemiological and clinical evidence shows that inflammation is an important risk factor for gastrointestinal diseases. Glycyrrhiza glabra, a traditional Chinese medicine, has been shown to safely suppress gastric cancer; however, the anti-inflammatory mechanisms in gastric tumorigenesis have been poorly investigated. Therefore, this study is committed to demonstrate the in vivo anti-inflammatory effect of 18β-glycyrrhetinic acid (GRA), the main active component of G. glabra. The lymphocytes and macrophages were heavily infiltrated in the transgenic mice that highly expressed cyclooxygenase (COX)-2 and microsomal prostaglandin E synthase (mPGES)-1; however, a significant reduction was observed after treatment with GRA. In addition, GRA downregulated the protein levels of COX-2, GαS, EP2, and β-catenin, which were involved in the arachidonic acid pathway. In conclusion, our study showed the potential protective effects of GRA against inflammatory environment that might be involved in gastric tumorigenesis in vivo through the PGE2-EP2 receptor-mediated arachidonic acid pathway.
Introduction
Accumulating epidemiological and clinical evidences show that inflammation is an important contributor to the development and progression of various human diseases including gastrointestinal cancer. 1 Thus, blocking of inflammatory signaling is usually recognized as a potential therapeutic modality for gastric cancer chemoprevention.
The arachidonic acid (AA) pathway was identified firstly as a key player among various inflammatory networks. Prostaglandin (PG)H2 is catalyzed by cyclooxygenases (COX-1 and COX-2) and subsequently converted to PGE2 by microsomal prostaglandin E synthase (mPGES)-1. K19-C2mE transgenic mice highly expressed transgene-encoded COX-2 and mPGES-1 in forestomach and glandular stomach, whereas expression of endogenous COX-2 was undetectable and mPGES-1 was weakly expressed in the whole digestive tract. The increased level of PGE2 enhances macrophage infiltration, and that they are activated through epithelial cells by the gastric flora, resulting in gastric metaplasia, hyperplasia, and tumorous growth in the glandular stomach, with heavy macrophage infiltrations. 2 The inhibitors of prostaglandin formation or nonsteroidal anti-inflammatory drugs (NSAIDs) are well documented as chemopreventive agents that may decrease cancer risk. 3
Natural products have been a long-standing source of innovative treatments that have revolutionized the management of diverse diseases and shaped contemporary medical practice. In general, many traditional Chinese medicinal herbs (TCMHs) have been shown as effective chemopreventive and therapeutic agents. Glycyrrhiza glabra, an important TCMH, has been shown to protect Helicobacter pylori-infected gastric mucosa and safely suppress gastric cancer in vivo and in vitro.4,5 This study aimed to assess the in vivo chemotherapeutic potential of GRA, the main active component of G. glabra, on PGE2-EP2 receptor-mediated inflammatory response.
Materials and methods
Animal study
K19-C2mE transgenic (Tg) mice were maintained by breeding male Tg mice with female C57BL/6N mice at the Animal Facility of Jilin University. Six mice were housed in per plastic cage in a light-dark (12 h–12 h), temperature (23°C ± 2°C), and humidity (50% ± 8%) controlled laboratory environment according to the Institutional Animal Care Guidelines in Jilin University. Sterilizing water and standard diet with free access were provided for the animals. For genotyping, DNA samples were extracted from the tails of 4-week-old Tg mice using Genomic DNA Purification Kits (GeneMark, Taiwan, China) and subjected to polymerase chain reaction (PCR) as reported earlier. 2
Chemical reagents and solutions
18β-glycyrrhetinic acid (GRA) and dimethyl sulfoxide (DMSO) were purchased from Sigma-Aldrich (St. Louis, MO, USA). GRA dissolved in DMSO (vehicle) with the concentration of 0.05% was administered once daily to the mice for in vivo study. For 48 weeks of GRA treatment, all the mice were killed under deep anesthesia using diethyl ether to collect gastric tissues for further study. The study was approved by the Animal Care Committee of Jilin University and complied with national laws relating to the conduct of animal experiments.
Histological and immunohistochemical analyses
The stomach was resected and cut along the greater curvature. A part of tissues was fixed in 10% neutral buffered formalin, wax embedded, and sectioned at 4 μm thickness. Sections were stained with hematoxylin and eosin for histological analysis. Antibodies against Ki-67, F4/80 (1:200, Abcam, Cambridge, USA), EP2 (1:200, Cayman Chemical, MI, USA), and COX-2, Gαs, β-catenin (1:200, Santa Cruz, CA, USA) were used as the primary antibodies. Horseradish peroxidase (HRP) anti-rabbit/mouse IgG (Maixin, Fuzhou, China) was used as the secondary antibody. Immunostaining signals were visualized using the DAB (3, 3-diaminobenzidine) kit (Maixin). The stained slides were independently evaluated by two pathologists. For the grade of COX-2, EP2, Gαs, and β-catenin expression, H score calculation was performed as previously described. 6
Extraction of total RNA and real-time PCR quantification
The stomach was resected and cut along the greater curvature. The other part of tissues was frozen in liquid nitrogen and stored at −80°C for total RNA extraction. Total RNA was extracted using Eastep Total RNA Extraction Kit (Promega, Shanghai, China) following the manufacturer’s guidelines. The concentration and purity of the RNA were calculated by measuring its absorbance at 260 and 280 nm using NanoDrop ND-1000 spectrophotometer (NanoDrop Technologies, DE, USA) and agarose gel analysis. Thereafter, RNA was reversed-transcribed using cDNA synthesis kit (Roche, Basal, Switzerland). Quantitative PCR was performed for COX-2, interleukin (IL)-1β, IL-6, IL-12b, IL-1b, and tumor necrosis factor (TNF)-α using a SYBR Green PCR Mix (Roche) with the LightCycler 480 (Roche), using GAPDH gene as an internal control.
Statistical analysis
Data were expressed as mean ± standard deviation (SD). The statistical analysis was performed using GraphPad Prism Software (GraphPad Software Inc., San Diego, CA, USA).
Results
Effects of GRA on incidence, multiplicity, and size of gastric tumors
In K19-C2mE Tg group, the mice developed hyperplasia, dysplasia, and tumor growth in the glandular stomach. The mucosal thickness was increased, the cell was irregularly, the nuclear volume was increased with larger nucleus, and the glands were abnormally branched (Figure 1(a) and (c)). However, treatment with GRA suppressed the gastric hypertrophy, reducing the mucosal thickness (Figure 1(b) and (d)).

Effects of GRA on hyperplasia and dysplastic tumor in Tg mice. (a) and (c), HE staining showed irregular gastric mucosal and cell morphology in Tg mice (100× and 400×, respectively). (b) and (d), HE staining showed almost normal gastric mucosal after GRA treatment in Tg mice (100× and 400×, respectively).
In Tg group, the gastric tumor incidence was 77.8% (28/36), while GRA decreased the incidence to 33.4% (13/39) (P < 0.05). Larger and multiple tumors were presented in Tg mice (Figure 2). The mean tumor size was 12.1 ± 2.5 mm, and tumor multiplicity was 2.54 ± 1.50 in Tg mice. However, GRA treatment significantly decreased the tumor size (7.40 ± 2.80, P < 0.05) and tumor multiplicity (1.95 ± 0.66, P < 0.05; Table 1).

Effects of GRA on tumor incidence and multiplicity in Tg mice. (a), Wild-type mice, (b–d), Tg mice, and (e–h), GRA-treated Tg mice.
Effects of GRA on gastric tumor incidence and multiplicity in Tg mice.

GRA: 18β-glycyrrhetinic acid; SD: standard deviation.
Values are expressed as average ± SD.
*P < 0.05.
Effects of GRA on inflammation response in vivo
All gastric mucosal specimens from wild-type mice had normal histomorphology (Figure 3(a)). In Tg mice, numerous neutrophils and lymphoplasmocytic cells were found infiltrating in the submucosa (Figure 3(b)). The GRA treatment decreased the submucosal infiltration of mononuclear cells (Figure 3(c)). In addition, expression levels of several proinflammatory cytokines—COX-2, IL-1β, IL-6, IL-12b, IL-1b, and TNF-α—were analyzed by quantitative real-time polymerase chain reaction (qRT-PCR). And they were expressed at very high levels in Tg mice; however, the relative expression of IL-1β, IL-6, IL-1b, and TNF-α was decreased 0.36 ± 0.02 (P < 0.05), 0.42 ± 0.01 (P < 0.05), 0.51 ± 0.02 (P < 0.05), and 0.58 ± 0.06 (P < 0.05) times, respectively, after GRA administration (Figure 3(d)).
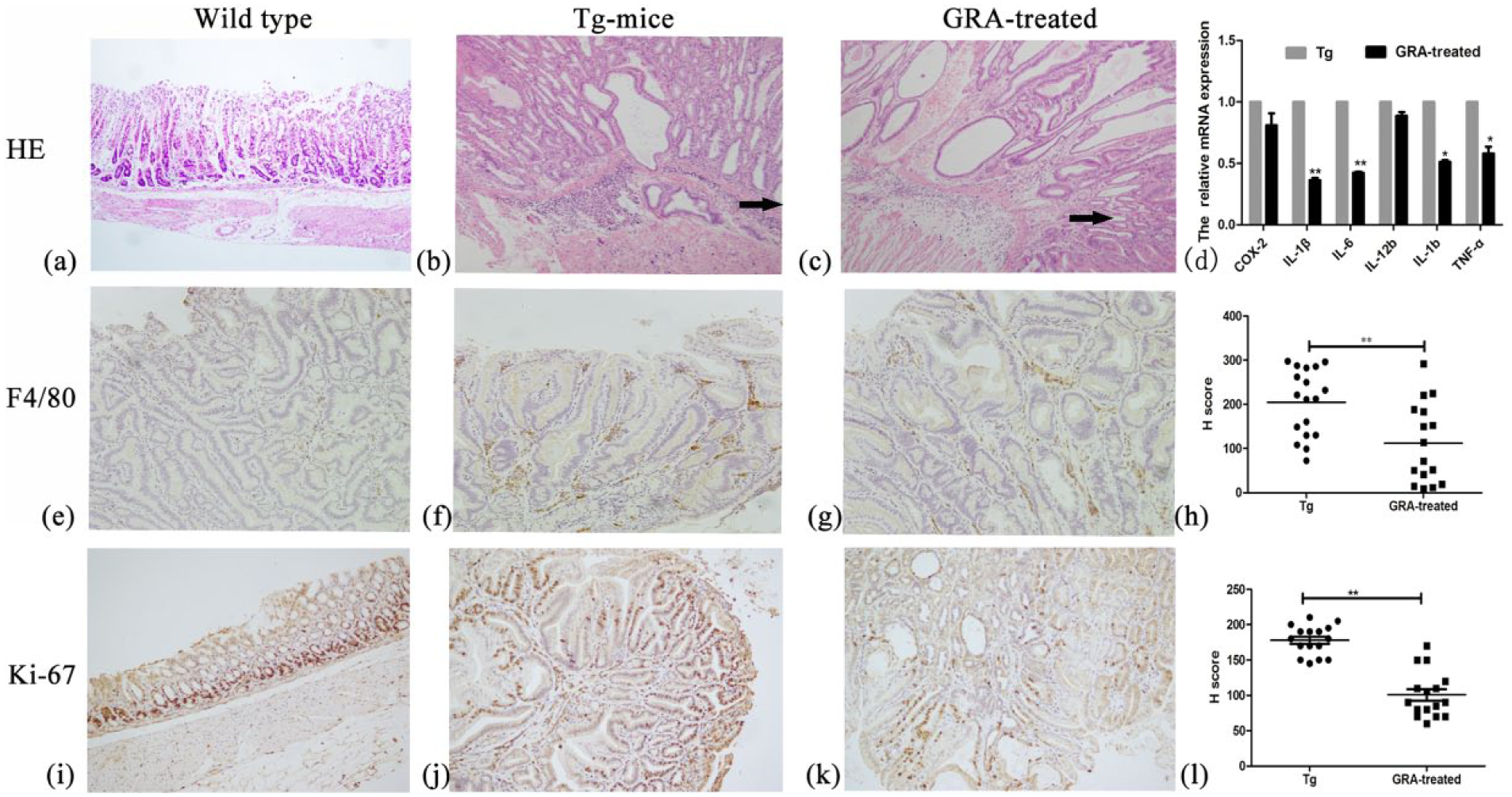
Effects of GRA on inflammation response occurred in Tg mice. HE staining showed (a) no neutrophils in wild-type mice, (b) heavy infiltration of neutrophils in Tg mice, and (c) decreased infiltration in GRA-treated Tg mice. (d), The expression levels of inflammatory cytokines. Immunostaining of F4/80 showed macrophages located in (e) wild-type mice, (f) Tg mice, and (g) GRA-treated Tg mice. (h), H score of F4/80 staining. Immunostaining of Ki-67 showed cell proliferation in gastric mucosa of (i) wild-type mice, (j) Tg mice, and (k) GRA-treated Tg mice. (l), H score of Ki-67 staining.
To investigate whether mucosal macrophages were involved in inflammatory responses, a macrophage marker F4/80 was next examined immunohistochemically. In the wild-type mice, tissue macrophages were sparsely scattered in the mucosal stroma (Figure 3(e)). In Tg mice, the mucosal stroma is heavily infiltrated with macrophages (Figure 3(f)). In contrast, treatment of GRA effectively suppressed the macrophage infiltration (Figure 3(g)). H score showed GRA downregulated F4/80 expression significantly (Figure 3(h)).
To detect whether the proliferation of epithelial cells was also influenced in the inflammation sites, Ki-67 expression was immunohistochemically analyzed. The Ki-67 labeling was focused on basal lamina in wild-type mice, while its staining was much stronger in Tg mice, and GRA treatment significantly decreased its expression (Figure 3(i)–(l)).
Effects of GRA on AA pathway
To elucidate the anti-inflammation and anti-cancer mechanisms of GRA, the expression of COX-2, Gαs, EP2, and β-catenin, several key components in AA pathway, were detected. The immunohistochemical staining results showed that COX-2 (Figure 4(a)), EP2 (Figure 4(b)), and Gαs (Figure 4(e)) mainly showed “nuclear/cytoplasmic” and “membranous” positive expression in almost all tumors in Tg mice. However, COX-2 only showed cytoplasmic and membranous expression (Figure 4(b)), EP2 (Figure 4(d)) only expressed in membrane, and Gαs mainly located in cell nuclear or cytoplasm (Figure 4(f)). β-catenin mainly expressed in cell nucleus in Tg mice (Figure 4(g)), while its nuclear accumulation was significantly alleviated (Figure 4(h)).
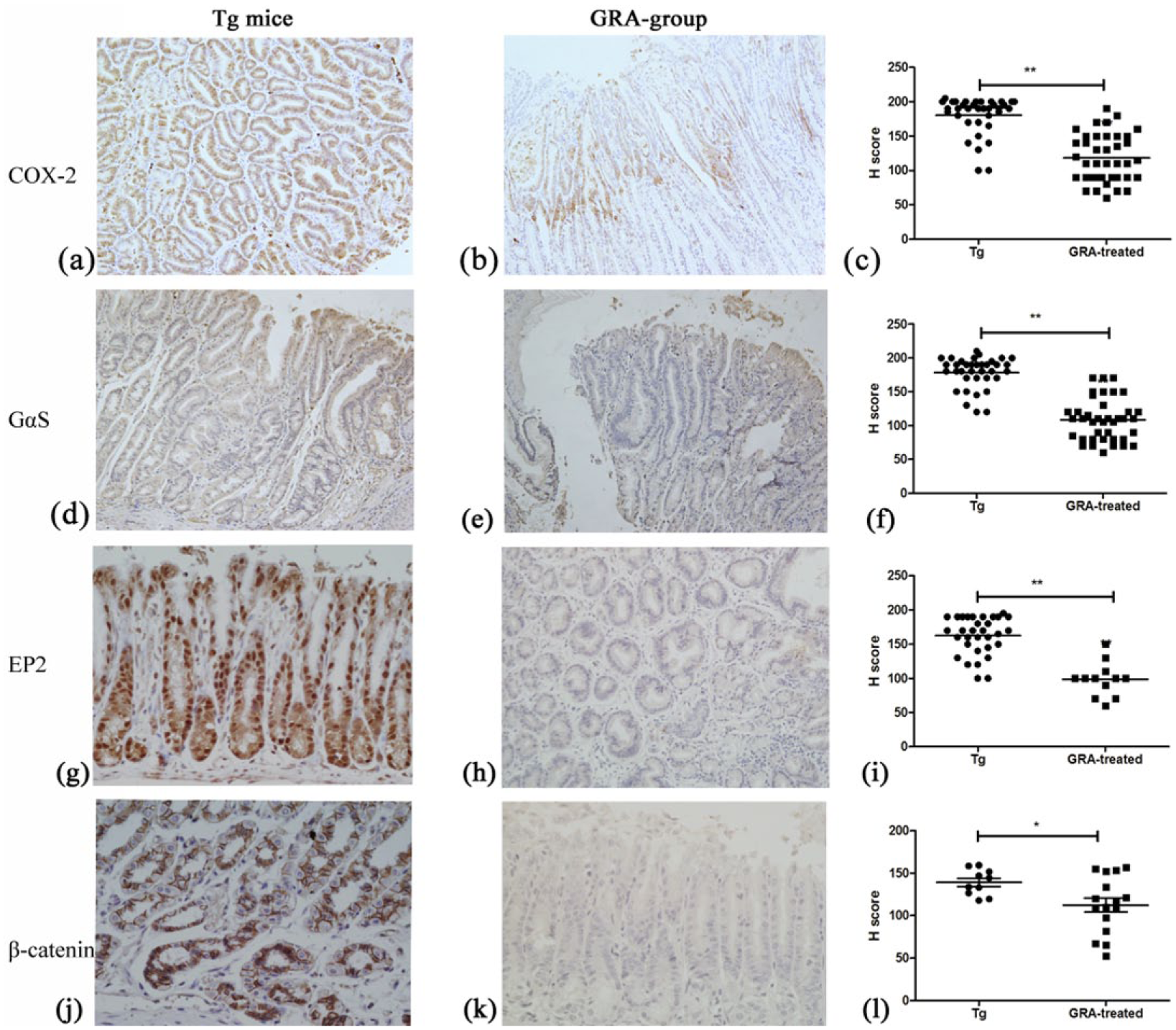
Effects of GRA on genes expression involved in arachidonic acid pathway. (a–c) COX-2, (d–f) Gαs, (g–i) EP2, and (j–l) β-catenin in Tg mice and GRA-treated Tg mice, respectively.
Discussion
GRA is a promising medicinal plant; it has been tested in H. pylori-infected Mongolian gerbils for its efficacy in anti-inflammatory activity, 4 and it also showed anti-oxidative and anti-carcinogenic effects in various models.5,7 However, the effect of GRA on PGE2-EP2 receptor-mediated inflammation microenvironment in gastric tumorigenesis remains poorly understood.
In this study, we assessed the effects of GRA on inflammation response and gastric tumor growth in Tg mice, because the Tg mice develop inflammation-associated hyperplastic tumors in the proximal glandular stomach, which was similar to those found in the Helicobacter-infected stomach. 2 The results indicated that GRA significantly decreased the submucosal infiltration of neutrophils and lymphoplasmocytic cells, downregulated the expression of various inflammation cytokines (Figure 3), inhibited the gastric tumorigenesis (Table 1), and reduced the expression of multiple key enzymes and mediators in AA pathway (Figure 4). Therefore, GRA had the potential to be developed as a new anti-inflammation agent with chemoprevention and chemotherapeutic potential for cancer.
Inflammatory mediators including cytokines and growth factors, as well as reactive oxygen species (ROS) and reactive nitrogen species (RNS), constituted the tumor microenvironment and were implicated in the etiology of inflammation-associated cancers. 8 The induction of the COX-2/PGE2 pathway activated the macrophages infiltration, and the activated macrophages might supply cytokines and growth factors to the epithelial cells. 9 COX-2 expression was increased in H. pylori-associated gastritis and was reported to be an independent factor for prognosis of gastric cancer. 10 Besides that, COX-2 expression is positively associated with cancer-related proteins APC and P53. PGE2 affects many mechanisms such as proliferation, invasion, and immunosuppression involved in tumorigenesis. PGE2-mediated immunosuppressive activity was partly associated with specific cytokines and chemokines, as well as their cognate receptors located on immune and epithelial cells. PGE2 directly inhibited the synthesis of IL-2 and the expression of the IL-2 receptor in T cells, thus reduced T cell proliferation and activation. 11 EP2 receptor was the major receptor mediating the PGE2 signal generated by COX-2 upregulation. In combination with stimulation of PGE2 formation, EP2 was aberrantly expressed in multiple gastrointestinal cancers. The homozygous deletion of EP2 decreased intestinal polyp formation in ApcΔ716 mice. 12
Emerging evidences place PGE2-EP2 receptor-mediated AA pathway directly with chronic inflammation and the pathogenesis of many human cancers. Inhibition of COX-2/PGE2/EP2 suppressed cell proliferation and the growth of tumors in vitro and in vivo.13,14 Several natural compounds have been evaluated for their ability to inhibit PGE2-EP2 signaling pathway. Curcumin (diferuloylmethane) suppresses IL-1β-induced PGE2 formation by inhibiting the expression of mPGES-1 in human lung epithelial cells. 15 in vivo This study provided evidences on the anti-inflammatory and anti-cancer effects of GRA and elucidated the mechanisms through modulating PGE2-EP2 receptor-mediated AA pathway, suggesting that GRA could be an ideal candidate as a new chemoprevention and chemotherapeutic agent for cancer.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by grants from the National Natural Science Foundation of China (No. 81673145) and the Scientific and Technological Development Program of Jilin Province (20180414055GH and 20160519016JH).
