Abstract
Angiogenesis is the process of new vascular formation, which is derived from various factors. For suppressing cancer cell growth, targeting angiogenesis is one of the therapeutic approaches. Vascular endothelial growth factor family receptors, including Flt-1, Flk-1 and Flt-4, have been found to play an essential role in regulating angiogenesis. Rapamycin is a macrolide compound with anti-proliferative properties, while platelet factor-4 (PF-4) is an antiangiogenic ELR-negative chemokine. Rapamycin inhibits mTOR ligands activation, thus suppressing cell proliferation, while PF-4 inhibits cell proliferation through several mechanisms. In the present study, we evaluated the effects of rapamycin and platelet factor-4 toward breast carcinoma at the proteomic and genomic levels. A total of 60 N-Methyl-N-Nitrosourea-induced rat breast carcinomas were treated with rapamycin, platelet factor-4 and rapamycin+platelet factor-4. The tumours were subsequently subjected to immunohistochemical protein analysis and polymerase chain reaction gene analysis. Protein analysis was performed using a semiquantitative scoring method, while the mRNA expression levels were analysed based on the relative expression ratio. There was a significant difference in the protein and mRNA expression levels for the selected markers. In the rapamycin+platelet factor-4-treated group, the Flt-4 marker was downregulated, whereas there were no differences in the expression levels of other markers, such as Flt-1 and Flk-1. On the other hand, platelet factor-4 did not exhibit a superior angiogenic inhibiting ability in this study. Rapamycin is a potent antiangiogenic drug; however, platelet factor-4 proved to be a less effective drug of anti-angiogenesis on rat breast carcinoma model.
Keywords
Introduction
Angiogenesis, a process by which new vasculature forms, occurs as a result of a variety of factors. Neo-angiogenesis is one of the main processes that allow tumours to grow larger than 2 mm in diameter. 1 Since the discovery of neo-angiogenesis, numerous intensive research studies have been conducted in order to determine the root factors that influence and enhance this process. Targeting angiogenic factors, such as vascular endothelial growth factor (VEGF), platelet-derived growth factor (PDGF), insulin-like growth factor (IGF), and fibroblast growth factor (FGF), and their receptors, have a potential impact on the development of new strategies to suppress tumour growth, especially at later stages. 2
Fms-like tyrosine 1 (Flt-1), Fms-like kinase 1 (Flk-1) and Fms-like tyrosine-4 (Flt-4) are members of the VEGF receptor family and are known as prognostic markers. 3 In addition, each of these VEGF receptors has been found to play a role in regulating neovascularization. 3 Moreover, these receptors co-regulate each other’s expression to trigger angiogenesis. Among these, Flk-1, known as a kinase-derived receptor (KDR), plays a significant role in the regulation of angiogenesis, whereas other molecules in the same family support this process. 4 Flt-1 has also been reported to play a significant role in branching new arteries, whereas Flk-1 influences vein formation, and Flt-4 plays a vital role in regulating lymphatic vessel formation.
Flt-1 is known to be the cognate receptor for VEGF-A. Thus, this receptor plays a crucial role in the regulation of neo-angiogenesis alongside Flk-1. Recent studies have revealed that Flt-1 induces angiogenesis along with Flk-1 through several intrinsic pathways, including the PLC-y, Grb2 and PI3K/Akt, which in turn activate downstream pathways, such as MAPK for proliferation, eNOS for cell permeability and Caspase 9 for survival. 5
Contrarily, Flt-4 plays a significant role in regulating lymphangiogenesis, which is a common route of cancer metastases. 6 Most cases of aggressive carcinoma exhibit significant Flt-4 expression, which reflects the degree to which several factors, especially Flt-4, play a role in the regulation of neo-lymphangiogenesis. 7 Based on our previous findings in rats with breast carcinoma, the treated groups exhibited a significant downregulation of the Flt-4 receptor; moreover, the cancer cells were significantly less aggressive, and most of the tumour cells appeared localized. In the present study, we examined and analysed the expression of VEGFRs in rat breast carcinoma following treatment with rapamycin and PF-4.
In this experiment, rapamycin or sirolimus is a macrolide compound which previously used as an antifungal agent. Rapamycin was then discovered to have potent immunosuppressive and anti-proliferative properties. 8 In the context of an antiangiogenic agent, rapamycin had been undergone extensive research on its effectiveness towards various cancer and several stages of a clinical trial. Rapamycin inhibits mTOR ligand, which suppresses the angiogenesis process occur. 3 In this experiment, the effectiveness of rapamycin was studied towards breast carcinoma in vivo. In addition, rapamycin was found to inhibit the progression of dermal Kaposi’s sarcoma in patients with renal transplants. On the other hand, the other mTOR inhibitors, temsirolimus and everolimus, were tested to treat glioblastoma multiforme and mantle cell lymphoma. 9 Thus, rapamycin was found promising for the future anticancer drug to act as an adjuvant or therapy drug in many research.
Platelet factor-4 (PF-4) is an antiangiogenic ELR-negative chemokine. 10 In a previous study, PF-4 was found to inhibit endothelial cell proliferation and migration as well as angiogenesis in vitro and in vivo. 11 According to the literature, three different mechanisms have been proposed to explain the inhibitory effects of PF-4 on the angiogenesis process. 12 The first mechanism, PF-4, may bind to proteoglycans and interfere with the proteoglycan-bystander impact on growth factor activity. The second mechanism is that PF-4 molecules can interact directly with angiogenesis growth factors such as fibroblast growth factors or vascular endothelial growth factors and inhibits the interaction of growth factors with cell surface receptors. Third, PF-4 may activate cell surface receptors on endothelial cells and induce inhibitory signals. Several PF-4 fragments such as PF-4-CTF have been made that exhibit anti-angiogenesis properties which is important for further therapeutic development. In this experiment, PF-4 was combined with rapamycin to study the synergistic effects of breast carcinoma in vivo.
Materials and methods
Animal procedures
Sixty female Sprague Dawley (SD) rats were obtained from the Animal Research and Services Centre, and ethical clearance was obtained from the Animal Ethics Committee (PPSG/07(A)/044/(2010) (56)). The rats were housed at the animal house unit. The caging and handling of the rats conformed to good laboratory practices, as outlined by the Animal Research and Services Centre (ARASC). To avoid discomfort, pain and stress, the rats were given an analgesic drug (ketamine) at a dose of 80 mg/kg intraperitoneally during the measurement of the tumour. The rats were maintained in groups of six and fed a standard laboratory diet. 13 They were divided into four groups, and each group was given different interventions/drug(s) as follows: the control (untreated) group 1 (n = 15), rapamycin-treated group 2 (n = 15), platelet factor-4-treated group 3 (PF-4) (n = 15), and rapamycin and PF-4-treated group 4 (rapamycin+PF-4) (n = 15). The rats were sacrificed 5 days after the drug intervention process.
N-nitroso-N-methylurea, rapamycin and platelet factor-4 preparation and tumour induction
N-nitroso-N-methylurea was dissolved in freshly prepared 0.9% normal saline before the induction process. The NMU (N-nitroso-N-methylurea) solution was injected intraperitoneally into 21-day-old rats at a dose of 70 mg/kg body weight, 14 after which each animal was monitored and palpated weekly for mammary tumour lesions. Daily inspection and palpation were also performed to monitor the onset of tumours in the mammary region, and the weight of the rats was measured daily. Rapamycin was mixed with absolute ethanol and diluted in mixtures of 10% polyethylene glycol (PEG)-400, 8% ethanol and 10% Tween-80 to a final concentration of 20 μg/0.2 mL, whereas PF-4 was dissolved in normal saline with a final concentration of 20 μg/0.2 mL
Study design
This study was conducted in vivo and involved an intraperitoneal injection of the NMU carcinogen into 60 female Sprague Dawley rats using a 21G needle to induce breast carcinoma. After developing breast tumours within 40 days of NMU carcinogen injection, the tumours were then suppressed using an angiogenic inhibitor as either a single treatment (PF-4 or rapamycin) or a combination of these two compounds; for combination treatment, both PF-4 and rapamycin were injected into the tumours. The rats were terminated after 5 days of intervention, whereas rats in the control group were terminated 45 days after tumour induction. Haematoxylin and eosin staining together with immunohistochemical stains were used to analyse the histology and expression of angiogenic protein markers. All results were then statistically analysed using the SPSS software version 21.
Immunohistochemistry
Tissue sections on slides were deparaffinized in xylene and then rehydrated in graded alcohol solutions. Antigen retrieval was conducted in a pressure cooker, and the selection of an appropriate buffer depended on the detected marker. Nonspecific background staining was blocked by a 3% hydrogen peroxide solution. Sections were incubated with the primary antibody for 1 h at room temperature, which was followed with several washes and incubation with the secondary antibody using an UltraVision ONE Large Volume Detection System HRP Polymer Kit. The protein reactivity was visualized using a 3,3′-diaminobenzidine (DAB) Plus substrate system (Cat. No. TL-125-PHJ, LabVision, United States). The tissue was counterstained with haematoxylin, which stained the nuclei.
Immunohistochemistry scoring system
Each section was scored in a blinded manner by two observers based on a previously reported semiquantitative scoring method. In addition, each sample was scored based on the percentage of tumour cells stained (0- the absence of staining; 1- <30% of tumour cells stained; 2 - 30–60% of tumour cells stained and 3- >60% of tumour cells stained).
Ribonucleic acid expression analysis
The tissue sample extraction was performed using TRIZOL (Invitrogen, United States) to obtain total ribonucleic acid (RNA). All RNA preparation and handling steps were carried out under RNAse-free conditions and were performed in a laminar flow hood. Total RNA from each fraction was dissolved in 20 μL of RNA storage buffer (Ambion, United States) and stored at −80°C until use. RNA concentration was determined by 1% gel electrophoresis and absorbance readings at 260 nm using the Nanodrop-1000 spectrophotometer (NanoDrop, Technologies, United States). The reading of RNA samples was within the range of 1.8–2.2, indicating the purity of the sample. This ratio was obtained from the 260/280 absorbance ratio. mRNA was isolated from total RNA using the Dynabeads mRNA purification kit (Invitrogen, United States) according to the manufacturer’s instructions. cDNA synthesis was performed using the SuperScript™ First-Strand Synthesis System (Invitrogen, United States) in a total volume of 20 μL according to the manufacturer’s instructions.
Quantitative RT- PCR analysis
List of gene markers by TaqMan® PCR assay used for angiogenesis analysis.

Quantification was accomplished with the PikoReal Real-Time qPCR System (Applied Biosystems, United States) using TaqMan® Universal PCR Master Mix and universal thermocycling parameters as recommended by Applied Biosystems. Reverse transcription polymerase chain reaction (RT-PCR) samples were run in duplicates using 1 μL cDNA, and the reactions were performed in 96-well plates (Applied Biosystems) in a reaction volume of 20 μL. The gene expression normalization factor for each sample was calculated according to the geometric mean of all three selected reference genes. 15 The Minimum Information for Publication of Quantitative Real-Time PCR Experiments (MIQE) guidelines was considered for the performance and interpretations of the qPCR reactions. 16 Gene expression was analysed according to the relative expression ratio (R-value) obtained from the cycle threshold (CT) value and amplification efficiency (E) value (E = −1+10 (−1/slope)). The expression of targeted genes for each sample provides evidence that these drugs act at the gene level.
Statistical analysis
A non-parametric one-way ANOVA test was used to determine the differences in the immunohistochemical expression of Flt-1, Flk-1 and Flt-4 among the experimental groups. Any results where p < 0.05 were considered statistically significant. All statistical analyses were conducted using the SPSS software version 21.
Results
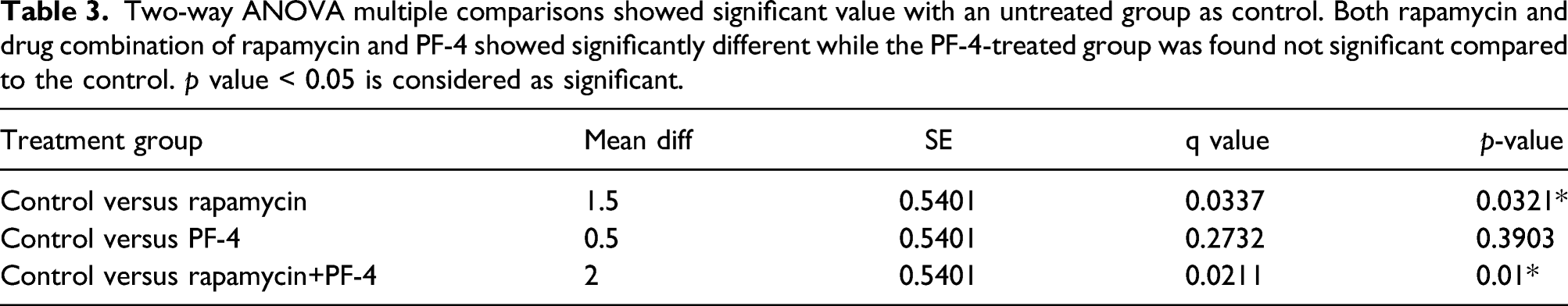
Two-way ANOVA multiple comparisons revealed a significant difference (p < 0.05) for rapamycin-treated groups in all angiogenic markers (Flt-1, Flk-1 and Flt-4), while rapamycin+PF-4-treated group was found significant for Flk-1 and Flt-4 protein; meanwhile, Flt-1 protein was found not significant compared to control. However, PF-4-treated group was not significant compared to the control (Figure 1). The reductions in proteins expression were highly influenced by the treatment used. The untreated group was used as control, and all protein expressions from rapamycin, PF-4 and rapamycin+PF-4 groups were compared with the untreated group. The expression of VEGFRs signalling protein receptor on rat’s mammary carcinoma. The VEGFRs expressions reflect the efficacy of treatment given to suppress angiogenesis via VEGFRs signalling blockage. All treatment groups showed reduced VEGFRs expressions compared to the untreated group. p-value <0.05 is considered significant.
Fms-like tyrosine-1/Vascular endothelial growth factor receptor1
Rapamycin (M = 90.1664, SD = 7.4487) exhibited better antiangiogenic effects than PF-4 (M = 93.7946, SD = 7.1303) and rapamycin+PF-4 (M = 93.6990, SD = 1.8432) with respect to Flt-1 marker. This demonstrated that PF-4 and rapamycin+PF-4 exerted fewer effects in suppressing this angiogenesis marker in tumour cells. Contrarily, rapamycin was effective in blocking angiogenic markers through the mTOR signalling pathway.
Fms-like kinase-1/Vascular endothelial growth factor receptor2
Rapamycin (M = 89.9043, SD = 7.2542) again revealed better antiangiogenic effects than PF-4 (M = 98.9175, SD = 2.0487) and rapamycin+PF-4 (M = 91.2330, SD = 4.0934) in the suppression of the angiogenic marker Flk-1. This marker has the potential to trigger angiogenesis along with other growth factors. These findings were slightly different from that of the Flt-1 marker, as the rapamycin+PF-4 group exhibited better suppression of this marker. The result still demonstrated a lack of synergistic activity between rapamycin and PF-4. Contrarily, PF-4 did not result in good suppression of this receptor, as mentioned in the literature. 17
Fms-like tyrosine-4/Vascular endothelial growth factor receptor3
Rapamycin+PF-4 (M = 87.8700, SD = 4.1620) was very efficacious in suppressing the Flt-4 marker compared with either rapamycin or PF-4 alone. Interestingly, the protein was downregulated more in the combination group than in the other groups, suggesting synergistic activity between these drugs. In contrast, the rapamycin group (M = 90.0236, SD = 7.2414) exhibited similar effects but to a lesser degree than those of the rapamycin+PF-4 group with respect to Flt-4. Nevertheless, both groups showed significant Flt-4 downregulation. However, PF-4 alone (M = 96.5385, SD = 1.8304) failed in that the protein was slightly downregulated, but the levels were not significantly different from those after no treatment (control).
Protein expression and scoring
Vascular endothelial growth factor receptors were highly expressed in the cytoplasm of tumour cells (breast carcinoma) (Figures 2–4). The tissues that lacked expression and those without cytoplasmic staining indicated suppressive activity on VEGFRs by the drugs. Photomicrograph of NMU-induced rat’s breast tumour tissue. The stain on tumour cells showed the expressions of Flt-1 marker on rapamycin-treated rat’s breast carcinoma. There was a medium intensity of immunohistochemistry stain determined. Flt-1 immunostaining (a) x100 and (b) x400. Tumour cell (TC), blood vessel (BV). Photomicrograph of NMU-induced rat’s breast tumour tissue. The stain on tumour cells showed the expressions of Flk-1 marker on PF-4-treated rat’s breast carcinoma. There was a high intensity of immunohistochemistry stain determined reflected with high scoring. Flk-1 immunostaining (a) x100 and (b) x400. Tumour cell (TC), blood vessel (BV). Photomicrograph of NMU-induced rat’s breast tumour tissue. The stain on tumour cells showed the expressions of Flt-4 marker on rapamycin+PF-4-treated rat’s breast carcinoma. There was a low intensity of immunohistochemistry stain determined. Flt-4 immunostaining (a) x100 and (b) x400.


A semiquantitative scoring system was used to determine the protein expression and included counts of positive cells and protein expression intensity. The counting was performed on three selected regions of interest based on the region with the highest protein expressions. The control group had a score of 9 points for both the Flt-1 and Flk-1 markers and 7.5 points for Flt-4, indicating that tumour growth was primarily dependent on the formation of new blood vessels to supply the nutrients needed for that growth. Moreover, the high expression of Flt-4 indicated lymphangiogenesis, which is used by tumour cells as a passageway for metastases.
Effects of angiogenesis inhibitors
Semiquantitative score of VEGFR expression in NMU-induced mammary carcinoma in rats. The rapamycin and rapamycin+PF-4 groups exhibited lower scores, whereas the PF-4 group exhibited less effective suppressive activity on VEGFR markers.

Two-way ANOVA multiple comparisons showed significant value with an untreated group as control. Both rapamycin and drug combination of rapamycin and PF-4 showed significantly different while the PF-4-treated group was found not significant compared to the control. p value < 0.05 is considered as significant.
RNA expression
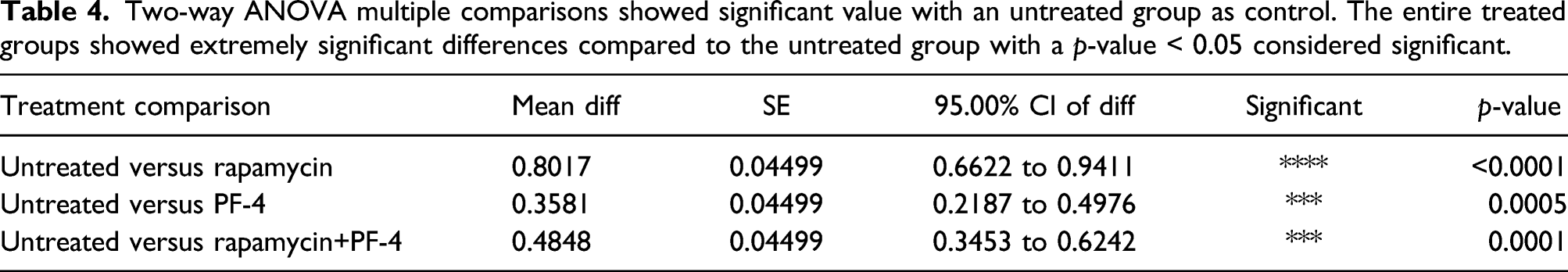
The mRNA expression levels of Flt-1, Flk-1 and Flt-4 genes from the rapamycin, PF-4 and rapamycin+PF-4-treated groups were significantly downregulated compared with the untreated group (Figure 5), with p-values of 0.036, 0.018 and 0.000, respectively. Based on these findings, the expression ratio of genes in all intervention groups was downregulated by a mean factor of 0.591 (SE range was 0.441–0.789), 0.458 (SE range was 0.311–0.669) and 0.134 (SE range was 0.078–0.267) with p-values < 0.01. Moreover, we found that the drugs used for intervention had suppressed angiogenesis and lymphangiogenesis at the mRNA level with varying intensities. Two-way ANOVA multiple comparisons (Table 4) showed the gene expression was significantly different from the untreated control group with a p value<0.05 in all treated groups. This indicated the VEGFR factors were highly suppressed at the gene level, resulting in downregulation of the VEGFR factors gene. As demonstrated at the protein level, genes expression in the rapamycin group was significantly downregulated compared to the untreated group, whereas PF-4 and rapamycin+PF-4 groups genes expression was found to have less downregulated compared to the untreated group. This finding suggests intervention with PF-4 led to a less suppressive activity on tumour growth indicated by high genes expression, specifically with respect to VEGFRs. The expression ratio of Flt-1, Flk-1 and Flt-4 genes. The mRNAs were isolated from NMU-induced mammary carcinoma under the influence of rapamycin, PF-4 and rapamycin+PF-4. Rapamycin treated group was determined to show massive gene downregulation compared to other treated groups. In addition, all VEGFRs markers showed significant downregulation of gene expression compared to the untreated group. Two-way ANOVA multiple comparisons showed significant value with an untreated group as control. The entire treated groups showed extremely significant differences compared to the untreated group with a p-value < 0.05 considered significant.
Discussion
In this study, we elucidate the effects of rapamycin and PF-4 as a single dose and in combination on breast carcinoma in vivo. We investigated four intervention groups and a control group (untreated). Rapamycin has been used in several medical applications, including as an antifungal agent, an anti-rejection drug after organ transplants, as a treatment for lymphangioleiomyomatosis (LAM) and as an anti-proliferative agent (anticancer). 18 Contrarily, PF-4 is a molecule responsible for inducing blood coagulation and activating platelets during platelet aggregation. PF-4 plays a significant role in wound repair and inflammatory reactions. 19 In this study, we observed the relation of synergistic strength between interventional drugs, which are rapamycin and PF-4 against the proliferative and angiogenic activities in breast carcinoma.
Angiogenesis has long been known as fundamental for such diverse physiological processes as embryonic and postnatal development, reproductive functions and wound repair. The blood vessels provide oxygen and nutrients and carry key regulatory signals to growing tissues. 20 Studies on tumorigenesis have shown that the inflammatory response is sometimes accompanied by increased vascular proliferation. This finding led to the proposal that new blood vessels play a crucial pathogenic role in regulating inflammatory processes and fulfilling the demands of proliferating cells. Tumours need nutrients to grow, and their only way to obtain sufficient nutrients is through blood vessels. 21 The mechanisms of the development of new vasculature from the existing vessels require the activation of some upstream signalling receptors, which will then trigger some angiogenic pathways, including the mTOR and PI3K/Akt pathways. Based on these findings, we hypothesize that aggressive tumour cells express more angiogenic proteins than non-aggressive tumours. This indicates that tumours are highly dependent on angiogenesis, which provides them with additional vessels to fulfil the demands for nutrients and oxygen needed by growing tumour cells. In addition, the vessels also function as a metastatic passageway for tumour cells to spread primarily via lymphatic vessels.
Based on the literature, rapamycin acts on mTOR through several upstream pathways. mTOR can be classified into two distinct types, mTOC1 and mTOC2, which are characterized by the presence of raptor and rictor, respectively. Signalling through mTORC1 involves Grb2 and Ras–Raf–MEK–Erk, whereas signalling through mTORC2 involves two different pathways that involve IRS-1 and Grb10. 22 The VEGF and mTOR pathways are connected by IRS-1 and PI3K/Akt. Inhibition of mTOR pathways will affect the production of VEGF, thus suppressing angiogenesis. In contrast to rapamycin, PF-4 affects the CXCR3 ligand. Activated CXCR3 protein will activate the PI3K/Akt pathway downstream. 23 With the combination of both drugs (rapamycin and PF-4), we hypothesized that they would function synergistically against the PI3K/Akt pathway, which would result in a better prognosis.
This study has noted that after the treatment of rapamycin or rapamycin+PF-4, the expression of VEGFRs was significantly suppressed, whereas PF-4 treatment alone resulted in less suppression of targeted markers. Based on these findings, we report that PF-4 and rapamycin do not function synergistically on VEGFRs, but some evidence suggests that they function antagonistically against the ligands. VEGF-A (Flt-1 and Flk-1) was discovered to be a survival factor for endothelial cells, and it exerts its effects through the PI3K/Akt pathway. High VEGF-A expression in tumour cells suggests that VEGF prevents endothelial cell apoptosis induced by serum starvation.
The expression of angiogenic markers, primarily Flt-1, Flk-1 and Flt-4, was associated with the size and aggressiveness of tumours. Tumour cells in larger, more aggressive tumours require more nutrients and oxygen for their growth. 24 Based on previous studies, Flt-1 protein was highly associated with the production of Flk-1/KDR proteins, which play a significant role in the regulation of angiogenesis and vasculogenesis. Interestingly, with their roles as signalling receptors, Flt-1, Flk-1 and Flt-4 were reported to be responsible for such disorganization and lethality and were found to be negative regulators of VEGF, at least during the early development. In our rat model, Flt-1 was highly expressed in aggressive tumour cells. This indicates that Flt-1 mediates chemotactic signals and potentially extends the role of the receptor, suggesting that this protein promotes mechanisms other than angiogenesis, such as increasing the survival rate of tumour cells, mitogenesis and permeability-enhancing effects of VEGF rather than being a non-functional or decoy receptor. In the current study, we found that both receptors (Flt-1 and Flk-1) were potentially elevated in epithelial–mesenchymal transition in breast carcinoma and resulted in a rapid proliferation of tumour cells in this tissue.
Angiogenesis highly depends on VEGF protein production, which relies on Flk-1/KDR to activate its pathways. 6 Several previous studies reported that Flk-1 signals act through the PI3K/Akt and PLC-y/Raf–MEK–ERK signalling cascades, which play a significant role in survival, permeability, migration and proliferation. 25 Moreover, bulk production of these proteins is important to sustain a rapidly growing tumour, requiring additional oxygen and nutrients. In this study, we found that the expression of Flk-1/KDR was prominent, indicating that angiogenesis was very aggressive. Conversely, the reduced expression of VEGF markers suggests the suppression of angiogenesis.
Fms-like tyrosine-4 is the receptor of VEGF-C and VEGF-D and acts as a pivotal regulator of lymphangiogenesis. In this study, we found that the expression of Flt-4 was closely associated with tumour aggressiveness. Previous findings in human breast carcinoma with overexpression of VEGF-C revealed the induction of lymphangiogenesis in and around the tumour. This finding confirms preliminary evidence that Flt-4 is a vital receptor that regulates the development of new lymphatic vessels, an essential route for tumour metastases. 26 In this study, we found that Flt-4 was highly expressed in aggressive breast carcinoma but expressed to a lesser degree in the rapamycin- and rapamycin+PF-4-treated groups. These results suggested that both rapamycin and rapamycin+ PF-4 were useful in treating metastatic carcinoma but that the use of PF-4 alone did not result in good outcomes. Furthermore, we found that the use of antiangiogenic drugs suppressed the development of lymphatic vessels more so than blood vessels. This demonstrates that these drugs can potentially block the interaction between VEGF-C- and Flt-4-soluble VEGFR3 fusion proteins.
In this experiment, we had encountered some limitations of the study. Those limitations include not using power calculation to determine the sample size for in vivo study. However, the method resource equation used to determine the sample size was proven good based on many in vivo cancer studies. 27 Secondly, the western blot method is a useful method to strengthen protein expression analysis of immunohistochemistry staining. However, in this experiment, we have conducted qRT-PCR gene expression analysis and MVD calculation to enhance the evidence of immunohistochemistry findings. According to a study by Gheni and Westernberg (2020), 28 the evaluation of HER2/neu oncogene in breast cancer patients was carried out by using immunohistochemistry protein expression and qRT-PCR gene expression methods. 28 Overall, the details of the study may be useful for further investigation of rapamycin drug towards its anticancer property.
Conclusion
In conclusion, rapamycin is a potent antiangiogenic drug acting on breast carcinoma. The downregulation of VEGFR expression at both protein and mRNA levels in the rapamycin-treated group indicates that rapamycin is best used against an angiogenic stimulant. Conversely, PF-4 has been demonstrated to be a less effective antiangiogenic drug in this breast carcinoma model and has played an antagonistic role for rapamycin.
Footnotes
Acknowledgements
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Research University grants from Universiti Sains Malaysia (grant number 1001/PPSP/813035).
Ethical approval
Ethical approval for this study was obtained from Animal Ethics Committee of Universiti Sains Malaysia (PPSG/07(A)/044/ (2010) (56)).
Animal Walfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
