Abstract
Background
Hypercoagulability is a risk factor of thromboembolic events in COVID-19. Anti-phospholipid (aPL) antibodies have been hypothesized to be involved. Typical COVID-19 dermatological manifestations of livedo reticularis and digital ischemia may resemble cutaneous manifestations of anti-phospholipid syndrome (APS).
Objectives
To investigate the association between aPL antibodies and thromboembolic events, COVID-19 severity, mortality, and cutaneous manifestations in patients with COVID-19.
Methods
aPL antibodies [anti-beta2-glycoprotein-1 (B2GP1) and anti-cardiolipin (aCL) antibodies] were titered in frozen serum samples from hospitalized COVID-19 patients and the patients’ clinical records were retrospectively analyzed.
Results
173 patients were enrolled. aPL antibodies were detected in 34.7% of patients, anti-B2GP1 antibodies in 30.1%, and aCL antibodies in 10.4%. Double positivity was observed in 5.2% of patients. Thromboembolic events occurred in 9.8% of patients, including 11 pulmonary embolisms, 1 case of celiac tripod thrombosis, and six arterial ischemic events affecting the cerebral, celiac, splenic, or femoral-popliteal arteries or the aorta. aPL antibodies were found in 52.9% of patients with vascular events, but thromboembolic events were not correlated to aPL antibodies (adjusted OR = 1.69, p = 0.502). Ten patients (5.8%) had cutaneous signs of vasculopathy: nine livedo reticularis and one acrocyanosis. No significant association was observed between the presence of cutaneous vasculopathy and aPL antibodies (p = 0.692).
Conclusions
Anti-phospholipid antibodies cannot be considered responsible for hypercoagulability and thrombotic events in COVID-19 patients. In COVID-19 patients, livedo reticularis and acrocyanosis do not appear to be cutaneous manifestations of APS.
Keywords
Introduction
Patients affected by COVID-19 may develop a hypercoagulation state that is associated with a significantly increased risk of thromboembolic events, including microthrombosis of small vessels, major venous 1 or arterial thrombosis, 2 and even disseminated intravascular coagulopathy (DIC). 3 The pathophysiology of these phenomena has not yet been fully elucidated. Various mechanisms have been suggested, including the development of systemic inflammation and cytokine storm4,5 and a specific vasculopathy due to direct vascular endothelial cell infection by SARS-CoV-2 through the angiotensin-converting enzyme two receptor. 6 Anti-phospholipid (aPL) antibodies are well-known pro-thrombotic factors and have also been hypothesized as possible culprits. 7 Indeed, clinical manifestations of thrombosis in COVID-19 might evoke an anti-phospholipid syndrome (APS). Moreover, DIC may be substantially indistinguishable from a catastrophic APS. In COVID-19, skin is not spared by vasculopathy and microthrombotic events. 8 Typical COVID-19 dermatological manifestations range from mild forms of livedo reticularis to severe digital ischemia and may also resemble the cutaneous manifestations of APS. 8 aPL antibodies not only promote platelet aggregation and activation but also induce a proinflammatory and procoagulant endothelial phenotype, upregulating cellular adhesion molecules and promoting the synthesis of endothelial nitric oxide synthase and of proinflammatory cytokines such as interleukin-6 and tumor necrosis factor-α. 9 Infections, such as pneumonia, human immune deficiency, hepatitis C, and skin and urinary tract infections, are the most common infectious triggers of APS.9,10 In these cases, it has been hypothesized that molecular mimicry between proteins of infecting pathogens and beta2-glycoproteins-1 (B2GP1) (or other molecules) may elicit aPL antibodies. Remarkably, over 1/3 of the immunogenic proteins in SARS-CoV-2 have potentially problematic homology to proteins that are key to the human adaptive immune system. 11 Moreover, SARS-CoV2 seems to induce organ injury through alternative mechanisms beyond direct viral infection, including immunological injury. 12
However, no evidence has so far truly confirmed this hypothesis, and it is currently a hot topic of debate whether aPL antibodies are responsible for hypercoagulability or mere bystanders in COVID-19 patients. 13 Zhang et al. were the first to report 3 cases of ischemic stroke associated with anti-cardiolipin (aCL) and anti- B2GP1 in COVID-19 patients. 14 Since then, a huge number of studies have been published on COVID-19 patients, lupus anticoagulant,15,16 aPL antibodies, and thrombotic events.17–27
Notably, these studies have yielded somewhat variable results, reporting a prevalence of aPL antibodies in COVID-19 patients ranging from an unremarkable 3.7% 25 up to an impressive 57%. 28 Moreover, some papers have assessed only LA and have not further investigated aPL antibodies.2,15 Most importantly, not all authors have correlated circulating aPL antibodies with actual thrombotic events and have not tested aPL antibodies sequentially in time, thus not fulfilling the diagnostic criteria of APS. 29 This latter limitation, regarding repeated aPL antibody testing after 12 weeks, might be explained by the high mortality rate in the patients tested and by design biases of the studies.
Zhang et al. themselves initially cautioned that aPL antibodies may arise transiently in patients with critical illness and infections, and that the presence of these antibodies rarely leads to thrombotic events. 14 Also, false-positive LA test results in patients with COVID-19 might be expected, given the marked elevation in measured C-reactive protein and the common use of low molecular weight heparin and unfractionated heparin for thromboprophylaxis.30–32
The aim of the present study was to investigate the prevalence of aPL antibodies in hospitalized patients with COVID-19 and the possible correlation between aPL antibodies, hypercoagulability, thromboembolic events, COVID-19 severity, mortality, and cutaneous manifestations of vasculopathy.
Methods
Study population
We conducted a retrospective cohort study on patients affected by COVID-19, who were hospitalized between March and May 2020 at the Ospedale Policlinico San Martino IRCCS, in Genoa, Italy. All patients tested positive for SARS-CoV-2 on naso-pharyngeal swab samples and clinically presented full-blown COVID-19. No other specific inclusion or exclusion criteria were adopted in selecting the sample. Frozen serum samples, which were used for aPL antibody titration, were obtained at the beginning of hospitalization (within 10 days). Abnormalities in laboratory parameters were evaluated on the same date as serum samples were drawn. By contrast, thrombotic events and cutaneous manifestations were evaluated throughout the entire hospitalization. Demographics, pre-existing comorbidities, and the course of the disease during hospitalization were obtained by retrospectively analyzing clinical records. Patients were subdivided into three severity groups: mild, moderate, and severe, based on the clinical course of the disease and taking into account respiratory insufficiency, pneumonia severity, and the need for invasive ventilation. Severe cases included critically ill patients with extensive acute respiratory distress syndrome, endotracheal intubation, and mechanical ventilation; moderate cases included patients with severe pneumonia and who needed the respiratory support of continuous positive airway pressure; and mild cases were patients needing hospitalization, but in whom pneumonia was milder and oxygen was administered via nasal cannula or face masks.
Recorded thromboembolic and ischemic events included deep venous thrombosis, pulmonary embolism, myocardial infarction, stroke, and disseminated intravascular coagulation. The cutaneous manifestations studied were mainly vasculopathy-related, such as acrocyanosis, digital ischemia, and livedo reticularis, but also other COVID-19-related cutaneous manifestations, such as rashes. Local ethics committee approval was obtained (N. CER Liguria 521/2020).
Laboratory analysis
Serum samples were evaluated for anti-B2GP1 (IgG, IgA, and IgM) and anti-cardiolipin (aCL) (IgG, IgA, and IgM) antibodies by means of a commercial enzyme-linked immunosorbent assay (Euroimmun Mediziniske Labordiagnostika, Germany). Results were expressed as arbitrary units, with a cut-off of positivity according to the manufacturer’s instructions.
Statistics
Continuous variables were summarized in terms of mean and standard deviation and median and interquartile range (IQR) according to their distribution; when in any group the single IQR was equal to the mean, the minimum–maximum range was used. Categorical variables were summarized in terms of total count and percentage. Continuous variables reported with means were compared by means of two-sample t-test, and continuous variables reported with medians and ordered categories were compared by means of Mann–Whitney test, and categorical variables by means of chi-square test.
Correlations were estimated by using Spearman’s Rho correlation coefficient over pairwise complete observations: we adjusted the p values of the correlations for multiple comparisons by means of the false-discovery rate method and considered p lower than 0.05 as significant.
We modelled the risk of death, thromboembolic events, or livedo reticularis or acrocyanosis occurring within the study by using a binomial logistic model. We estimated the effect of aPL antibodies, with and without adjustments for age and clinical status, to mitigate differences between patients with anti-B2GP1 or aCL antibodies and those without these antibodies (details on variable selection procedures are reported in Supplementary Table 1). An ordinal logistic regression model was used to assess the association between clinical state and aPL antibodies, with and without adjustments for age and vasculopathy discrepancies.
All analyses were performed by means of R statistical software, version 4.0.2.
Results
General characteristic of the sample, N = 173.
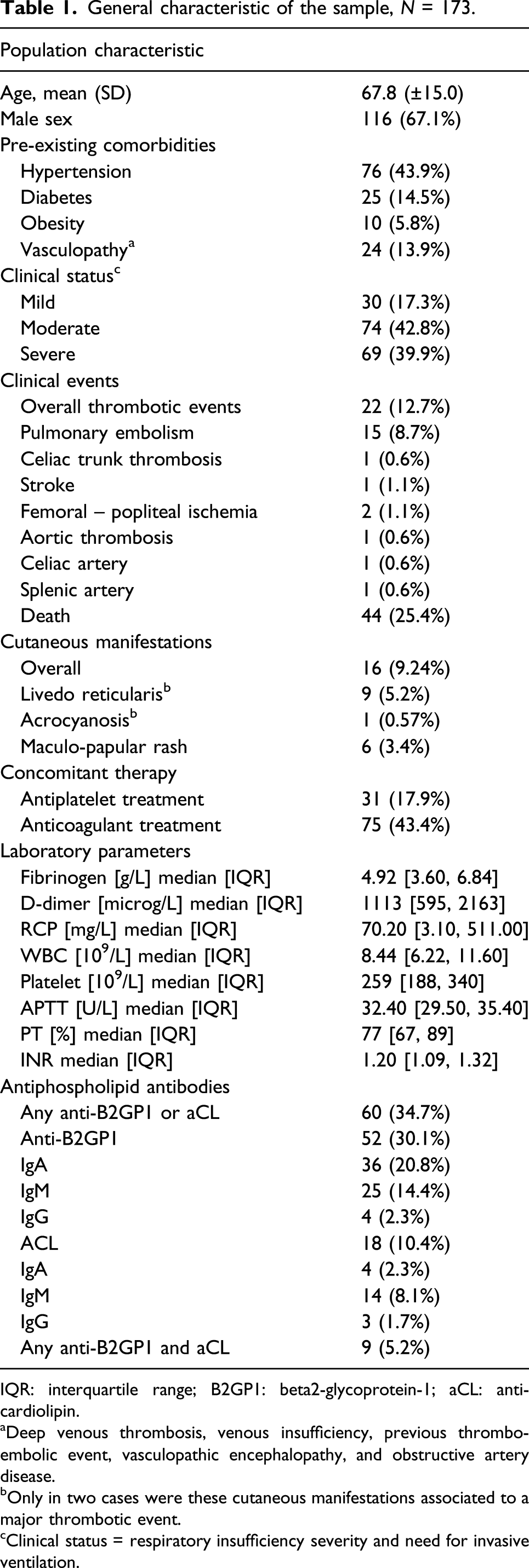
IQR: interquartile range; B2GP1: beta2-glycoprotein-1; aCL: anti-cardiolipin.
aDeep venous thrombosis, venous insufficiency, previous thrombo-embolic event, vasculopathic encephalopathy, and obstructive artery disease.
bOnly in two cases were these cutaneous manifestations associated to a major thrombotic event.
cClinical status = respiratory insufficiency severity and need for invasive ventilation.
The female-to-male ratio across the three groups with different disease severity was similar (n. of males in mild group: 60.0% vs moderate: 70.3% vs severe: 66.7%, p = 0.598). Patients with milder clinical status were on average younger (mean age in mild group: 63 ± 17 vs moderate: 64 ± 13 vs severe: 74 ± 14 years, p < 0.001).
Prevalence of anti-phospholipid antibodies
At least one type of aPL antibody was detected in 60/173 patients (34.7%); at least one type of anti-B2GP1 antibody in 52/173 patients (30.1%); and at least one type of aCL antibody in 18/173 patients (10.4%) (Table 1). Anti-B2GP1 IgA antibodies were detected in 36/173 patients (20.8%) (Table 1), IgM in 25/173 (14.4%), and IgG in 4/173 (2.3%). Anti-CL IgA antibodies were detected in 4/173 patients (2.3%), IgM in 14/173 (8.1%), and IgG in 3/173 (1.7%). Of the 60 patients with circulating aPL antibodies, 50% had severe, 25% had moderate, and 25% had mild COVID-19. Double positivity for both aCL and anti-B2GP1 antibodies was found in 9/173 (5.2%) of patients.
Thromboembolic events, COVID-19 severity, and mortality
We observed 22 thromboembolic/ischemic events in 17/173 patients (9.8%). In detail, we observed 16 venous thrombo-embolic events, namely, 15 cases of pulmonary embolism, 1 case of thromboembolism of the celiac tripod, and six arterial ischemic events—affecting the splenic artery in one case, the celiac trunk in one case, the cerebral arteries in one case, the aorta in one case, and the femoral-popliteal arteries in two cases (Table 1).
Comparison between aPL+ and aPL− patients.

aPL: anti-phospholipid; IQR: interquartile range; B2GP1: beta2-glycoprotein-1; aCL: anti-cardiolipin.
aDefined as respiratory insufficiency severity and need for invasive ventilation.
bConsidered as ordered categories.
cp calculated by multivariate analysis.
Moreover, aPL antibodies were not significantly associated with greater disease severity [OR = 1.24 (95%CI 0.68, 2.29), p = 0.486; adjusted by age and prior vasculopathies OR = 1.24 (95%CI 0.45, 3.48), p = 0.678].
Apparently, higher mortality rates occurred in patients with aPL antibodies [OR = 2.72 (95%CI 1.35, 5.55)], but after adjustments for age and clinical status [OR = 2.11 (95%CI 0.71, 6.40), p = 0.180], the effect was reduced in size and significance.
Laboratory findings and anti-phospholipid antibodies
Univariate correlations between lab values.

Spearman’s Rho, (p-value) [number of patients].
aSignificant at p < 0.05.
b36 patients (20.8%) with value equal to or higher than 20 R/ml.
Cutaneous manifestations
Univariate associations between cutaneous vasculopathy manifestations, laboratory parameters, and clinical events.
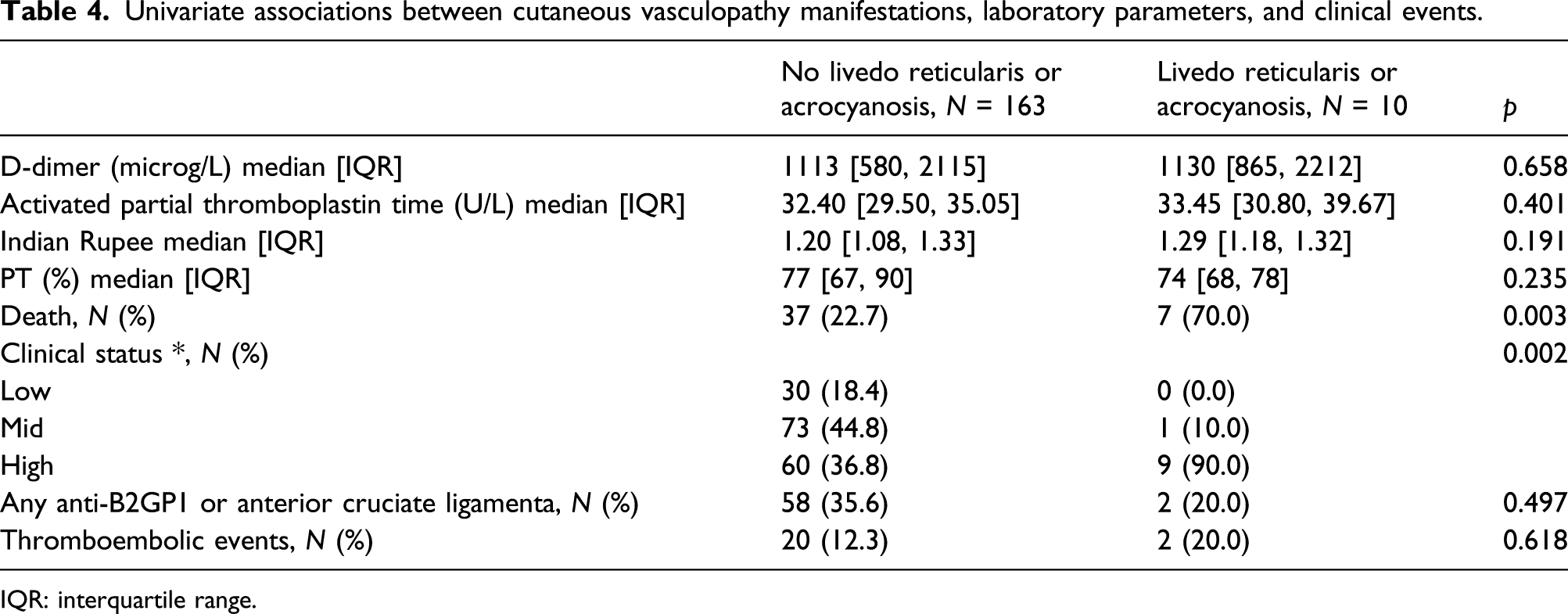
IQR: interquartile range.
Discussion
The prevalence of aPL antibodies (34.7%) in our cohort of hospitalized COVID-19 fell into the range reported in previous studies25,28 and was lower than the prevalence (58%) reported by a review pooling 23 studies with a total of 250 patients, 13 and it is considerably higher when compared to the expected prevalence in healthy controls. Our patients with aPL antibodies did not show a higher risk of thrombotic events during hospitalization. In other words, half of the patients who actually suffered thrombotic events had circulating aPL antibodies, while the other half did not, making the association between aPL antibodies and thrombotic events purely arbitrary.
Furthermore, the prevalence of anti-aPL antibodies in COVID-19 patients is significantly less frequent compared to the prevalence reported in actual APS patients. 32
It has been reported that aCL and anti-B2GP1 IgA antibodies are the most frequent circulating aPL antibodies in COVID-19 patients, and their presence has been associated with more severe COVID-19. 24 On the other hand, a review of the existing literature reported anti-B2GP1 IgM antibodies as the most common isotype. 13 In the present study, IgAs were the most common isotype of anti-2-GP1 antibodies, while IgMs were the most frequent isotype of aCL antibodies. Overall, IgA antibodies were the most frequently found Ig isotype, but no correlation with disease severity, mortality rate, or thrombotic events was found, unlike what has previously been reported. 24 It is, however, interesting that IgA immunoglobulins are the first and main line of humoral response elicited by the bronchial-associated lymphoid tissue (BALT). 24 It could perhaps be speculated that the higher prevalence of IgA isotype could be related to epitope-spreading phenomena in BALT, in an effort to produce a valid humoral defense against SARS-COV-2.
In line with the results of a previous study, 33 multiple aPL antibody positivity and high aCL/anti-B2GP1 titers were not common in the present study. However, in patients with APS, the risk of recurrence of a thrombotic event has been seen to be greatly increased in those who have multiple subtypes of aPL antibodies (LA, aCL, anti-b2-GPI, and aPS/PT), that is, double- or triple-positive patients. 34
Another difference between COVID-19-associated aPL antibodies and APS is that patients with COVID-19 exhibit low titers of anti-B2GP1, with specificity against domains 4 and 5, while patients with APS display high aPL antibody titers, with specificity against domain 1 or b2-GP1. 20
In addition, aPL antibodies in our study did not correlate with either disease severity or with mortality rates. The only significant correlation was found between the presence of aPL antibodies and PT and INR prolongation, as expected. Indeed, it is well known that aPL antibodies in plasma determine these abnormal laboratory values, causing the lupus anticoagulant effect. 9
A limitation of the present study, and of previous studies, is that aPL antibodies were not re-assayed after 12 weeks. Thus, it is not clear whether the presence of aPL antibodies in COVID-19 patients was transient or persistent. One study, in which aPL testing was repeated after 1 month, suggested that aPL antibodies in COVID-19 patients were mostly a transient phenomenon that occurred during the acute phase. 33 Moreover, owing to the retrospective design of the study, aPL antibody titration was performed on serum samples drawn at the beginning of hospitalization, and did not always coincide with the occurrence of thrombotic events. An additional limitation was the absence of a control group. Notably, a recent prospective study on patients with respiratory insufficiency hospitalized in the ICU found no significant difference in aCL antibody prevalence between COVID-19-positive patients and controls with respiratory insufficiency due to other causes. 35 LAC was not tested in our cohort of patients, and this might be a further limitation. This decision was based on the fact that LAC values may be distorted in the case of an increase in CRP, which is also to be expected in severe COVID-19, and that it is difficult to reasonably interpret LAC values that are measured during concomitant anticoagulation therapy, such as unfractionated heparin or vitamin K antagonists. 31 Finally, sample size was not calculated in this study, thus limiting statistical significance.
Notably, cutaneous signs of vasculopathy, including livedo reticularis and acrocyanosis, were seen both in patients with and without aPL antibodies, but were actually more common in patients without aPL antibodies. This implies that such manifestations are the cutaneous expression of a generic vasculopathy and are not exquisitely referable to aPL antibody presence or to APS. Additionally, livedo reticularis and acrocyanosis were not relatable to coagulation abnormalities. However, patients with cutaneous vasculopathy seem to have higher mortality rates and more severe COVID-19. This finding should be confirmed by further studies on higher numbers of patients.
Conclusions
On the basis of the findings of the present study, anti-phospholipid antibodies cannot be truly considered to be responsible for hypercoagulability and thrombotic events in COVID-19 patients, but rather mere bystanders. Moreover, in COVID-19 patients, livedo reticularis and acrocyanosis do not seem to be cutaneous signs of an actual APS, but rather an unspecific manifestation of vasculopathy.
Further studies, if possible, with a prospective design that involve repeated aPL antibody testing after 12 weeks and which include control groups (e.g., patients with pneumonia due to other infectious agents) are necessary in order to unquestionably elucidate the role of aPL antibodies in COVID-19-associated coagulopathy.
Supplemental Material
sj-pdf-1-iji-10.1177_20587384211042115 – Supplemental Material for A retrospective study on the prevalence of anti-phospholipid antibodies, thrombotic events and cutaneous signs of vasculopathy in 173 hospitalized COVID-19 patients
Supplemental Material, sj-pdf-1-iji-10.1177_20587384211042115 for A retrospective study on the prevalence of anti-phospholipid antibodies, thrombotic events and cutaneous signs of vasculopathy in 173 hospitalized COVID-19 patients by Giulia Gasparini, Paola Canepa, Simonetta Verdiani, Luca Carmisciano, Emanuele Cozzani, Denise De Grazia, Orsi Andrea, Giancarlo Icardi and Aurora Parodi in International Journal of Immunopathology and Pharmacology
Footnotes
Author Contributions
Paola Canepa, Simonetta Verdiani. These authors equally contributed.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
