Abstract
As adrenaline, serotonin and norepinephrine are two other vasoconstrictors and both of which have been proved to increase the quality and duration of local anesthetics when added as adjuvants. However, the difference in the improvement of the nociception of local anesthetics between the two adjuvants remains unclear. The purpose of this study was to assess the cutaneous nociception of mexiletine by coadministration with serotonin and norepinephrine. Subcutaneous injection of drugs or combinations includes mexiletine 0.6, 1.8, 6.0 μmol, serotonin 1.6500 μmol, noradrenaline 0.8895 nmol, saline, mexiletine 1.8 and 6.0 μmol, respectively combined with serotonin 0.4125, 0.8250, 1.6500 μmol and noradrenaline 0.0356, 0.1779, 0.8895 nmol, with each injection dose of 0.6 ml. The nociception of mexiletine alone and mexiletine coadministered with serotonin and norepinephrine was assessed after subcutaneous injection. Subcutaneous injections of mexiletine elicited dose-related cutaneous antinociception (P < 0.05, 0.01, or 0.001). Compared with mexiletine (1.8 μmol), adding norepinephrine (except for lowest dose) and serotonin to mexiletine (1.8 μmol) solutions for skin nociceptive block potentiated and prolonged the action (P < 0.01 or 0.001). Mexiletine (6.0 μmol) combined with norepinephrine and serotonin extended the duration of cutaneous antinociception when compared with mexiletine (6.0 μmol) alone (P < 0.05, 0.01, or 0.001). Both serotonin and norepinephrine improve the sensory block and enhances the nociceptive block duration of mexiletine, and serotonin is superior to that of norepinephrine.
Keywords
Introduction
As one of the most commonly used class Ib antiarrhythmic agents, mexiletine is used to treat acute and chronic ventricular arrhythmia in clinical practice. However, it produces a local anesthetic effect as a result of a sodium ion channel block effect, 1 besides its antiarrhythmic effect. Local analgesia is an easy and economical approach and the analgesic technique involves infiltration of the surgical wound and the surrounding tissues with a high-volume, long-acting local anesthetic solution, and frequently, with adjuvants. 2 It has been frequently performed for surgical procedures and postoperative pain control after various surgeries, such as total hip arthroplasty, laparoscopy, and deep brain stimulation surgery.3–5
The administration of local anesthetics in various types of pain is limited by its duration and dose-dependent adverse reactions. Adjuvants are commonly used with local anesthetics to synergistically enhance the duration of sensory-motor block and limit its cumulative dose requirement. Vasoconstrictors are most commonly used as adjuvants to extend the duration and improve the quality of analgesia as a result of the reduced absorption. As adrenaline, serotonin, and norepinephrine are two other vasoconstrictors and both of which have been proved to increase the quality and duration of local anesthetics when added as adjuvants.6–8 However, the difference in the improvement of the analgesia of local anesthetics between the two adjuvants remains unclear. The objective of the research was to compare the improvement of cutaneous nociception of mexiletine respectively by coadministration with different doses of serotonin and norepinephrine in a rat model of cutaneous trunci muscle reflex (CTMR) in response to local cutaneous pinpricks.
We are engaged in the research and development of local anesthetics. As far as the current local anesthetics are concerned, short-acting and long-term drugs are represented by lidocaine and bupivacaine, respectively. Mexiletine not only has a local anesthetic effect that lasts longer than lidocaine, but also has no cardiotoxicity compared with bupivacaine. Therefore, before the development of mexiletine as a local anesthetic, it is the significance of this study to clarify its compatibility with adjuvants.
Materials and methods
Animals
Male Sprague Dawley rats, weighing 200–250 g, were obtained from the Anhui Laboratory Animal Center (Hefei, China). They were kept three per cage in the animal housing facility at the Laboratory Animal Center of the First Affiliated Hospital of USTC with controlled room temperature (23°C ± 2°C) and a relative humidity of approximately 40%–60% under a natural light-dark cycle (12 h light/dark cycle (light from 7:00 AM–7:00 PM)) with ready access to food and water.
Drugs
Mexiletine hydrochloride was obtained from Sigma-Aldrich Chemical Co. (St. Louis, MO, USA). Serotonin hydrochloride and noradrenaline bitartrate were purchased from Target Molecule Corp. (Boston, MA, USA). All the drugs were dissolved in normal saline and were freshly prepared.
Subcutaneous injection
The rats were handled daily for 7 days before the experiment to familiarize them with the experimental environment and behavioral investigator. There were eight rats for each treatment. The detailed method of subcutaneous injection was on the basis of the previous reports.9,10 Before injection, the hair on the dorsal surface of thoracolumbar segment (10 cm × 6 cm) was mechanically shaved. Prepared drug 0.6 ml was injected into the shaved area with a 30-gauge needle. Immediately after injection, a circular bulge of approximately 20 mm in diameter was formatted on the injection area. Marked with ink within 1 min.
Subcutaneous injection of drugs or combinations includes mexiletine 0.6, 1.8, 6.0 μmol, serotonin 1.6500 μmol, noradrenaline 0.8895 nmol, saline, mexiletine 1.8 and 6.0 μmol respectively combined with serotonin 0.4125, 0.8250, 1.6500 μmol and noradrenaline 0.0356, 0.1779, 0.8895 nmol, with each injection dose of 0.6 ml.
Cutaneous nociception evaluation
The CTMR, as previously reported,9,10 characterized by the reflex movement of the skin over the back produced by twitches of the lateral thoracospinal muscle in response to local dorsal cutaneous stimulation, was used to assess the effect of cutaneous nociception. The CTMR responses were induced by a testing tool that affixed to the No. 15 von Frey filament (Somedic Sales AB, Stockholm, Sweden) with an 18-gauge end-cut needle that produced a standardized noxious stimulus (19 ± 1 g).
After observing the normal reaction to pinpricks applied outside the wheal and on the contralateral side, a total of six pinpricks at six different points was applied inside the wheal, with a frequency of 0.5–1.0 Hz. The testing of the six pinpricks was performed at 0, 2, and 5 min and every 5 min for the first 30 min, followed by every 10 min for 30–60 min after injection and every 15–30 min afterwards until the CTMR fully recovered from the block. Record the number of pinpricks that the rat failed to respond to after nociceptive stimulus. The antinociception of each dose of drug or combination was quantitatively assessed for the number of times that the pinprick failed to elicit a response.
The nociceptive block (percent of possible effect; %PE) was defined as the number of the pinpricks that the rat failed to react, with, for example, the complete absence of six responses was recorded as a complete nociceptive block (100% of possible effect; 100% PE). 11 The maximal blocking effect was presented as the percentage of maximal possible effect (%MPE), and the full recovery time was defined as the period from subcutaneous injection to complete recovery of the CTMR reactions. Record the full recovery time and %MPE of each dose of drug and combination of drugs. The investigator who was responsible for assessing the cutaneous antinociceptive effect was blinded to the drug or combination injected. To reduce the number of rats used, the shaved areas on the rats’ backs were averagely divided into four parts. Each area allowed for the injection of one drug or combination only, and the injection wheal with marked ink did not overlap with the neighbors. Each rat was given a recovery day after the first day of the experiment.
There were three specific experiments performed. In experiment 1, the dose-dependent effect of mexiletine was evaluated (n = 8 for each group). Mexiletine 0.6, 1.8, and 6.0 μmol were selected, based on a previous report. 1 In experiment 2, the indexes of cutaneous antinociception, including the %MPE, duration of action, and areas under the curve (AUCs) of mexiletine (1.8 or 6.0 μmol) alone or a mixture of mexiletine (1.8 or 6.0 μmol) with serotonin (0.4125, 0.8250, 1.6500 μmol) or noradrenaline (0.0356, 0.1779, 0.8895 nmol) were compared (n = 8 for each group). In experiment 3, in order to eliminate the possibility of systemic effects of mexiletine alone and the combinations, two control groups were designated. In one of the groups (n = 8 for each group), rats were intraperitoneally injected with mexiletine 6.0 μmol in combination with serotonin 1.6500 μmol or noradrenaline 0.8895 nmol. In the other group (n = 8 for each group), the rats underwent intraperitoneal administration of mexiletine at 6.0 μmol, serotonin 1.6500 μmol, or noradrenaline 0.8895 nmol.
Statistical analysis
The data are expressed as the means ± SEMs. The %MPE, duration and AUCs among the groups were analyzed using one-way ANOVA followed by Tukey’s honestly significant difference (HSD) test for paired comparisons. The AUCs of nociceptive block were calculated by using GraphPad Prism 7 for Windows (GraphPad Software Inc., CA, USA). SPSS for Windows (version 17.0, SPSS Inc., Chicago, IL, USA) was used for the statistical analysis, and a P-value less than 0.05 was considered significant.
Results
Subcutaneous injections of mexiletine elicited dose-dependent cutaneous antinociception in rats (Figure 1). Subcutaneous mexiletine at 1.8 μmol showed 50% blockade (%MPE), whereas mexiletine at 6.0 μmol exhibited 100% blockade, as depicted in Figure 1. Mexiletine (1.8, or 6.0 μmol) co-administrated with any dose of serotonin (0.4125, 0.8250, 1.6500 μmol) reached full sensory block (%MPE) (Figure 2a and b). Serotonin 1.6500 μmol exhibited 50.00% of subcutaneous antinociception, and saline vehicle did not elicit cutaneous antinociceptive effects (Figure 2a and b).
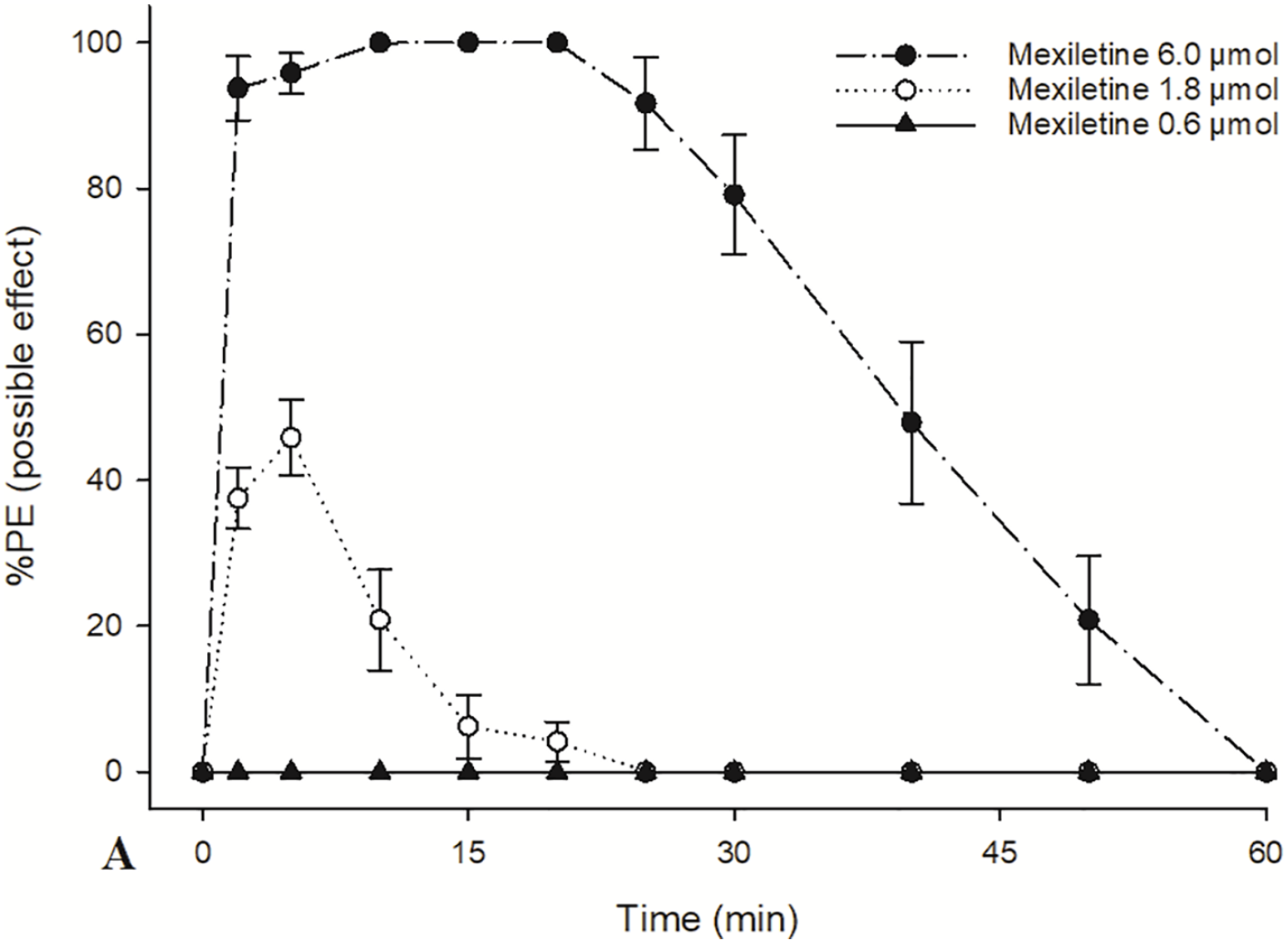
Time courses of cutaneous antinociception after three-dose treatment of mexiletine. Mexiletine at 6.0 μmol produced complete (100%) nociceptive block.

The addition of serotonin (0.4125, 0.8250, or 1.6500 μmol) to mexiletine (1.8 or 6.0 μmol) preparations at producing cutaneous antinociception. (a) Mexiletine 1.8 μmol respectively combined with different doses of serotonin and saline alone. (b) Mexiletine 6.0 μmol respectively combined with different doses of serotonin and serotonin 1.6500 μmol alone.
When mexiletine (1.8 μmol) co-administrated with noradrenaline (0.0356 or 0.1779 nmol), the sensory block (%MPE) reached 64.58%–85.42%, whereas when co-injected with noradrenaline (0.8895 nmol), it reached full subcutaneous antinociception (Figure 3a). Saline vehicle showed no cutaneous antinociceptive effects (Figure 3a). Mexiletine at 6.0 μmol achieved complete nociceptive block with any combination, whereas noradrenaline 0.8895 nmol alone exhibited 33.33% of subcutaneous analgesia (Figure 3b).

The addition of norepinephrine (0.0356, 0.1779, or 0.8895 nmol) to mexiletine (1.8 or 6.0 μmol) preparations at producing cutaneous antinociception. (a) Mexiletine 1.8 μmol respectively combined with different doses of norepinephrine and saline alone. (b) Mexiletine 6.0 μmol respectively combined with different doses of norepinephrine and norepinephrine 0.8995 nmol alone.
The %MPE, duration, and AUCs of each drug alone or combinations are presented in Tables 1 and 2. The drugs or combinations that intraperitoneal administrated including mexiletine 6.0 μmol, serotonin 1.6500 μmol, noradrenaline 0.8895 nmol, mexiletine 6.0 μmol combined with serotonin 1.6500 μmol or noradrenaline 0.8895 nmol did not provoke cutaneous analgesia. All the rats recovered completely after each injection.
The percent of maximum possible effect (%MPE), duration, and the area under the curves (AUCs) of mexiletine alone or co-administration of low dose mexiletine with serotonin and norepinephrine at producing cutaneous antinociception.

Data are expressed as mean ± SEM (n = 8 in each group).
Compared with Mex 1.8 μmol, aP < 0.001, b P <0.01; compared with Mex 1.8 μmol + Nor 0.0356 nmol, cP < 0.001, dP < 0.01; compared with Mex 1.8 μmol + Nor 0.1779 nmol, eP < 0.001, fP < 0.01; compared with Mex 1.8 μmol + Nor 0.8895 nmol, gP < 0.001, hP < 0.01; compared with Mex 1.8 μmol + Ser 0.4125 μmol, iP < 0.001, jP < 0.01; compared with Mex 1.8 μmol + Ser 0.8250 μmol, kP < 0.001.
The data among the groups were analyzed using one-way ANOVA followed by Tukey’s honest significant difference (HSD) test for paired comparisons.
Mex: mexiletine; Nor: norepinephrine; Ser: serotonin.
The percent of maximum possible effect (%MPE), duration, and the area under the curves (AUCs) of mexiletine alone or co-administration of high dose mexiletine with serotonin and norepinephrine at producing cutaneous antinociception.

Data are expressed as mean ± SEM (n = 8 in each group).
Compared with Mex 6.0 μmol, aP < 0.001, bP < 0.01, cP < 0.05; compared with Mex 6.0 μmol + Nor 0.0356 nmol, dP < 0.001; compared with Mex 6.0 μmol + Nor 0.1779 nmol, eP < 0.001; compared with Mex 6.0 μmol + Nor 0.8895 nmol, fP < 0.001, gP < 0.01; compared with Mex 6.0 μmol + Ser 0.4125 μmol, hP < 0.001, iP < 0.01; compared with Mex 6.0 μmol + Ser 0.8250 μmol, jP < 0.001.
The data among the groups were analyzed using one-way ANOVA followed by Tukey’s honest significant difference (HSD) test for paired comparisons.
Mex: mexiletine; Nor: norepinephrine; Ser: serotonin.
Compared with mexiletine (1.8 μmol), adding serotonin and noradrenaline to mexiletine (1.8 μmol) solutions for skin nociceptive block potentiated and prolonged the action. Mexiletine (6.0 μmol) combined with serotonin and noradrenaline extended the duration of cutaneous analgesia when compared with mexiletine (6.0 μmol) alone.
Compared with mexiletine 1.8 μmol+noradrenaline 0.0356 nmol, the %MPE, complete block time, full recovery time and AUCs increased in mexiletine 1.8 μmol+ serotonin 0.4125 μmol (P < 0.001). Compared with mexiletine 1.8 μmol+noradrenaline 0.1779 nmol, the %MPE, complete block time, full recovery time and AUCs increased in mexiletine 1.8 μmol+ serotonin 0.8250 μmol (P < 0.001). Compared with Mexiletine 1.8 μmol+noradrenaline 0.8895 nmol, the %MPE, complete block time, full recovery time and AUCs increased in mexiletine 1.8 μmol+ serotonin 1.6500 μmol (P < 0.001).
Compared with mexiletine 6.0 μmol+noradrenaline 0.0356 nmol, the complete block time, full recovery time and AUCs increased in mexiletine 6.0 μmol+serotonin 0.4125 μmol (P < 0.001). Compared with mexiletine 6.0 μmol+noradrenaline 0.1779 nmol, the complete block time and AUCs increased in mexiletine 6.0 μmol+serotonin 0.8250 μmol (P < 0.001). Compared with Mexiletine 6.0 μmol+noradrenaline 0.8895 nmol, the complete block time, full recovery time and AUCs increased in mexiletine 6.0 μmol+serotonin 1.6500 μmol (P < 0.001).
Discussion
Our results indicate that both serotonin and norepinephrine improve the sensory block and enhances the nociceptive block duration of mexiletine, and serotonin works much better than that of norepinephrine. The results of the study also show mexiletine exhibited a dose-dependent effect on subcutaneous antinociception, in agreement with previous studies.1,11
In addition, all the three doses of serotonin and norepinephrine improved the sensory block and enhanced the nociceptive block duration of mexiletine. Similar studies showed that serotonin and norepinephrine could intensify the analgesic effect of local anesthetics when used in combination.6–8 Local anesthetics block neural impulses by inhibiting sodium currents in nerve tissues.12,13 Lidocaine and its analog mexiletine are sodium channel blockers,14,15 both of them produced dose-dependent cutaneous analgesia.1,16
Postoperative pain brings a lot of harm to patients, such as cardiopulmonary function, coagulation function, abnormal gastrointestinal function, endocrine metabolism and other complications that seriously affect the recovery. Cutaneous analgesia via injection of local anesthetics is an acceptable way for the management of anesthesia and postoperative pain with fewer adverse reactions. 17
As another vasoconstrictor, phenylephrine has been recommended to be mixed with local anesthetics at a rate of 1:20,000 (0.1473 μmol) for a potent and extended local anesthetic effect in clinical practice, according to its instructions. Our preliminary results showed that the improved effect of norepinephrine 1:2,000,000 (0.8895 nmol) on the efficacy of local anesthetics was similar to that of phenylephrine 0.1473 μmol. Therefore, the dose of norepinephrine selected was 0.8895 nmol, and then 5-times diluted in sequence. Based on the previous study, 6 the doses of serotonin selected were 0.4125, 0.8250, and 1.6500 μmol.
As for the reason that the two adjuvants improve the local analgesia, we infer that it may be their vasoconstrictive effects, like adrenaline. Adrenaline is one of the vasoconstrictors that are traditionally used as adjuvants to improve the analgesia of local anesthetics 18 as a result of the reduced absorption into the blood. Interestingly, in our study, noradrenaline 0.8895 nmol alone exhibited 33.33% of subcutaneous antinociception, probable via mixed subtypes of α1-adrenoceptors. 19 As for the reason that subcutaneous serotonin at 1.6500 μmol exihibited 50.00% subcutaneous antinociception, probably due to serotonergic 5-HT1A and 5-HT4 receptors involve in antinociception.20,21 Norepinephrine, as an alpha-2 agonist, produces a vasoconstrictive effect, which causes a decrease in the spread of local anesthetics as well as the postponed absorption and the removal of local anesthetics surrounding the nerves. 11 Our results showed that as adjuvants, adrenaline, 22 dexmedetomidine, 22 clonidine, 11 serotonin, and norepinephrine can prolong the action time of mexiletine. Based on these results, the order of adjuvants strength from highest to lowest is: adrenaline > serotonin > dexmedetomidine > norepinephrine > clonidine. The difference in the degree of action of these adjuvants may be due to their different mechanisms. Adrenaline and norepinephrine are powerful vasoconstrictors, dexmedetomidine and clonidine are alpha2 receptor agonists. Besides acting on serotonergic 5-HT1A and 5-HT4 receptors, serotonin is also a potent vasoconstrictor. Since systemic administration of lidocaine exhibited central antinociceptive effects, 23 the two control groups were designed to banish the possibility of central antinociception. The results that intraperitoneal injection of mexiletine or combinations did not elicit cutaneous antinociception could eliminate this concern.
There are limitations in our study. We only compared the extent to which serotonin and norepinephrine enhance and prolong the local anesthetic effect of mexiletine. In fact, the mechanisms of action of the two adjuvants are different. This study did not clarify the mechanism. On the other hand, the serotonin doses we used can cause itching at the injection sites, 24 however, other analgesic drugs, such as morphine, produce the side effects of itching while producing analgesia. As a popular analgesic, morphine indeed has a good analgesic effect in spite of the side effects of itching. Therefore, we do not think that itching will affect the analgesic effect. Power calculation for estimation of sample size was not performed in our study, however, it is generally accepted that 5–8 rats in each group are sufficient for pain behavior testing.
Conclusion
In conclusion, subcutaneous mexiletine produces cutaneous antinociception in a dose-related manner. Both serotonin and norepinephrine improve the sensory block and enhances the nociceptive block duration of mexiletine, and serotonin is superior to that of norepinephrine.
Footnotes
Acknowledgements
Conceived and designed the experiments: JL, MH; Performed the experiments: MH, FK, CY; Analyzed statistic: MZ, KZ, TW; Wrote the paper: MH, ZL; Performed the revision: JL. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
The experimental protocols were approved by the Institutional Animal Care and Use Committee of the University of Science and Technology of China (USTCACUC1902025).
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation. All the experimental procedures strictly observed the guidelines of the International Association for the Study of Pain; Or The present study involved client-owned animals; it demonstrated a high standard (best practice) of veterinary care and involved informed client consent; Or Guidelines for humane animal treatment did not apply to the present study because *REASON*.
