Abstract
Several distinct classes of drugs, such as anticonvulsants, immunosuppressants, and calcium channel blockers, caused gingival overgrowth. One of the main drugs associated with the gingival overgrowth is the anti-epileptic such as phenytoin, which affects gingival tissues by altering extracellular matrix metabolism. In our study, we evaluate the effect of phenytoin, a drug whose active substance is phenytoin, on gingival fibroblasts of healthy volunteers. Gene expression of 29 genes was investigated in gingival fibroblasts’ cell culture treated with phenytoin compared with untreated cells. Among the studied genes, only 13 genes (CXCL5, CXCL10, CCR1, CCR3, CCR5, CCR6, IL-1A, IL-1B, IL-5, IL-7, IL-6R, BMP-2, and TNFSF-10) were statistically significant. All but one gene resulted downregulated after 24 h of treatment with phenytoin. BPM2 was the only, although weakly, up-expressed gene. Probably, we have not highlighted overexpression of the other inflammatory molecules because the study was performed on healthy people. Many studies show that phenytoin induces the overexpression of these cytokines but, probably, in our study, the drug does not have the same effect because we used gingival fibroblasts of healthy people.
Introduction
The term “gingival hyperplasia” usually used to describe an oral phenomenon seems nowadays used inappropriately because enlargement is not the result of an increase in the number of cells, but rather an increase in extracellular tissue volume. 1
“Gingival enlargement” is the term used to describe medication-related gingival overgrowth or gingival hyperplasia. It can be defined as an abnormal growth of the periodontal tissue. 2
This pathology causes not only aesthetic changes and clinical symptoms, such as pain, tenderness, bleeding, speech disturbances, and abnormal tooth movement, but also dental occlusion problems, enhancement of caries development, and periodontal disorders. 3
Drug-induced gingival enlargement (DIGE) connected with the chronic use of the anti-epileptic drug (AED) phenytoin (PHT) was reported first in 1939 by Kimball. 4 In the same year, Faurbye 5 and in 1959, Strean and Dilantin 6 suggested that the alkalinity of PHT could be the cause of the gingival side effect.
The onset of excessive gingival growth can take place after first month of treatment with drugs such as PHT; however, it usually occurs not earlier than 3 months after the start of therapy. Generally, the expansion of the gingival tissues begins around the lip and the surfaces of the mandibular anteriors. Clinically, the expansion of the gingival tissues begins in the region of interdental papillae, which gradually increase in size. The pathology then extends more sideways to adjacent papillae which merge. All drugs have unwanted effects and the morbidity of gingival enlargement needs to be compared with other possible side effects arising from alternative medication. 7
PHT is the first anticonvulsant choice for controlling the convulsive seizure disorders in patients experiencing epilepsy.8,9 Excessive gingival growth is one of the most common side effects associated with the administration of PHT, which is the currently used AED. The prevalence of PHT-induced gingival hyperplasia has shown variation ranging from 13% to 50%, in community-based studies on institutionalized patients. 10
To determine whether PHT, a drug whose active substance is PHT, can alter the inflammatory responses, we investigated its effects on gingival fibroblast cell culture analyzing the gene expression profiling of treated cells.
Materials and methods
This study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Ethics Committee of 06.09.2013 prot. n. 29579 University Study of L’Aquila.
We analyzed the gingival tissues of 99 healthy volunteers who have expressed their consensus to participate to this study.
Primary human fibroblast cells culture
Fragments of gingival tissue of healthy volunteers (11-year-old boy, 68-year-old woman, and 20-year-old man) were collected during operation. The pieces were transferred in 75-cm2 culture flasks containing Dulbecco’s Modified Eagle Medium (DMEM) (Sigma-Aldrich, Inc., St. Louis, MO, USA) supplemented with 20% fetal calf serum and antibiotics (penicillin 100 U/mL and streptomycin 100 µg/mL; Sigma-Aldrich, Inc.).
Cells were incubated in a humidified atmosphere of 5% CO2 at 37°C. The medium was changed the next day and twice a week. After 15 days, the pieces of gingival tissue were removed from the culture flask. Cells were harvested after additional 24 h of incubation.
Cell viability test
A stock solution of PHT 1 mg/mL was prepared. Further dilutions were made with the culture medium to the desired concentrations just before use.
Cell lines were seeded into 96-well plates at a density of 104 cells per well containing 100 µL of cell culture medium and incubated for 24 h to allow cell adherence.
Serial dilution of PHT (5000, 2000, 1000, 500, and 100 ng/mL) was done and PHT was added to the wells (three wells for each concentration). The cell culture medium alone was used as a negative control.
After 24 h of incubation, cell viability was measured using PrestoBlue™ Reagent Protocol (Invitrogen, Carlsbad, CA, USA) according to the manufacturer’s instructions. Briefly, the PrestoBlue™ solution (10 µL) was added into each well containing 90 µL of treatment solution. Plates were then placed back into the incubator for 1 h, after which absorbance was measured at wavelengths of 570 nm excitation and 620 nm emission by an automated microplate reader (Sunrise™; Tecan Trading AG, Männedorf, Switzerland). The percentage of viable cells was determined by comparing the average absorbance in drug-treated wells with average absorbance in control wells exposed to vehicle alone. The results were presented as the mean ± standard deviation of three measures.
Cell treatment
Cell lines were seeded at a density of 1.0 × 105 cells/mL into 9-cm2 (3 mL) wells and subjected to serum starvation for 16 h at 37°C.
Cells were treated with 1000 ng/mL PHT solution for 24 h. This solution was obtained in DMEM supplemented with 2% fetal bovine serum (FBS), antibiotics, and amino acids. Cell medium alone was used as a control negative. The cells were maintained in a humidified atmosphere of 5% CO2 at 37°C. After the end of the exposure time, cells were trypsinized and processed for RNA extraction.
RNA isolation, reverse transcription, and quantitative real-time reverse transcription polymerase chain reaction
Total RNA was isolated from cell lines using GenElute mammalian total RNA purification Miniprep Kit (Sigma-Aldrich) according to the manufacturer’s instructions. Pure RNA was quantified at NanoDrop 2000 spectrophotometer (Thermo Scientific, Waltham, MA, USA).
Complementary DNA (cDNA) synthesis was performed starting from 500 ng of total RNA, using PrimeScript RT Master Mix (Takara Bio Inc., Kusatsu, Japan). The reaction was incubated at 37°C for 15 min and inactivated by heating at 70°C for 10 s. cDNA was amplified by real-time quantitative polymerase chain reaction (PCR) using the ViiA™ 7 System (Applied Biosystems, Foster City, CA, USA).
All PCR reactions were performed in a 20-µL volume. Each reaction contained 10 µL of 2× qPCRBIO SYGreen Mix Lo-ROX (PCR Biosystems, London, UK), 400 nM concentration of each primer, and cDNA.
Custom primers belonging to the “Inflammatory Cytokines and Receptors” pathway were purchased from Sigma-Aldrich. All experiments were performed including non-template controls to exclude reagents contamination. PCR was performed including two analytical replicates.
The amplification profile was initiated by 10 min incubation at 95°C, followed by two-step amplification of 15 s at 95°C and 60 s at 60°C for 40 cycles. As a final step, a melt-curve dissociation analysis was performed.
Statistical analysis
The gene expression levels were normalized to the expression of the reference gene (RPL13) and were expressed as fold changes relative to the expression of the untreated cells. Quantification was carried out with the delta/delta Ct calculation method. 11
Results
The optimal concentration of PHT to be used for cell treatment has been obtained using PrestoBlue™ cell viability test. Based on this test, the concentration used for the treatment was 1000 ng/mL.
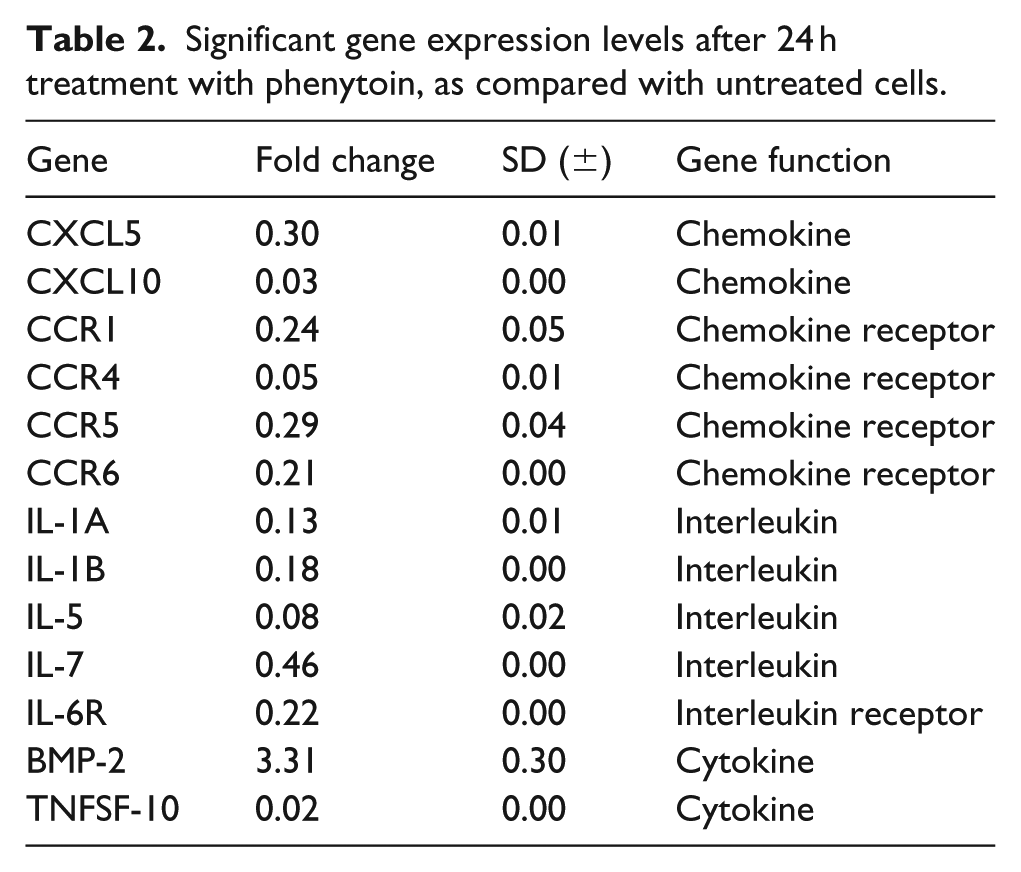
Fibroblasts’ behavior was evaluated by measuring the gene expression profile of 29 genes belonging to the “Inflammatory Cytokines and Receptors” pathway (Table 1, Figure 1). In total, 13 genes (CXCL5, CXCL10, CCR1, CCR4, CCR5, CCR6, IL-1A, IL-1B, IL-5, IL-7, IL-6R, BMP-2, and TNFSF-10) were statistically significant (Table 2). All but one gene resulted downregulated after 24 h of treatment with PHT; BPM2 was the only, although weakly, up-expressed gene (Figure 2). Table 1 reports the list gene and their fold change.
Selected genes used in real-time PCR belonging to “Inflammatory Cytokines and Receptors” pathway.

Bold values indicate fold change of significant gene expression level.

Amplification plot of fibroblast treated with Dintoina 1000 ng/mL.
Significant gene expression levels after 24 h treatment with phenytoin, as compared with untreated cells.

Gene expression profile of fibroblast treated with Dintoina 1000 ng/mL.
Significantly deregulated genes are those with a fold-change value superior to 2 (upregulated) and less than 0.5 (downregulated).
Discussion
Gingival enlargement is a term now used to describe medication-related gingival overgrowth or gingival hyperplasia, a common reactionary phenomenon that occurs with use of several types of therapeutic agents, including AEDs. 2 This disorder has been recognized since 1939, shortly after the introduction of PHT. Gingival enlargement is associated with multiple factors, including inflammatory (acute and chronic), idiopathic, drug-induced, neoplasia (benign and malignant tumors), hormonal disturbances, ascorbic acid (vitamin C) deficiency, and with dental eruption. 3 Priyadharshini et al. 12 reviewed the etiology, pathogenesis of AEDs associated with gingival overgrowth, in addition to its multidisciplinary management and prevention.
Despite noticeable advances in the management of epilepsy in the recent decade, the AED PHT remains the drug of choice in the management of epileptic patients. PHT is useful in generalized, complex, partial epilepsy and in cortical focal epilepsy. It is absorbed slowly and incompletely after oral administration in the intestine. Gastrointestinal absorption is changed both by age-related physiologic changes and by drugs that alter gastrointestinal motility. In particular, anti-acids decrease PHT gastrointestinal absorption.
Several studies presented in literature suggest that PHT-induced gingival hyperplasia is more commonly found in younger age groups. 13 This is in accordance with the observations of the various epidemiological studies. 3
In the study by Arya et al., 14 pediatric patients who developed gingival hyperplasia due to PHT use were included and followed up for 6 months. During the study, some of them were treated with folic acid against other patients who were kept on placebo. Gingival hyperplasia was found to be significantly reduced in patients treated with folic acid. Reviewing the literature in PubMed, we wanted to try to study which kind of gene could be responsible of the gingival hyperplasia, and how PHT could stimulate fibroblast growth.
To determine whether PHT alters the inflammatory responses, we investigated its effects on gingival fibroblast gene expression as compared with untreated cells.
Fibroblasts derived from gingiva of healthy volunteers were treated for 24 h with a solution of 1000 ng/mL and subsequently we have analyzed 29 genes belonging to the “Inflammatory Cytokines and Receptors” pathway.
In total, 13 genes (CXCL5, CXCL10, CCR1, CCR4, CCR5, CCR6, IL-1A, IL-1B, IL-5, IL-7, IL-6R, BMP-2, and TNFSF-10) were statistically significant. All but one gene resulted downregulated after 24 h of treatment with PHT. BPM2 was the only, although weakly, up-expressed gene (Figure 2). Probably, we have not highlighted overexpression of the other inflammatory molecules because the study was performed on healthy people.
DIGE may occur in patients taking therapeutic dosages of AED, mainly, but not only PHT. Its clinical appearance is similar in most cases and the comprehensive management may be challenging and multidisciplinary in nature.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
