Abstract
Uridine 5’-diphospho-glucuronosyl-transferase 1A1 (UGT1A1) plays an important role in the biliary excretion of bilirubin, suggesting genetic polymorphisms of UGT1A1 may have an impact on bile acid metabolism, which may be related to the development of anti-tuberculosis drug-induced liver injury (ATLI). This study explores the associations between genetic polymorphisms of UGT1A1 and ATLI in a Chinese anti-tuberculosis population. A 1:2 matched case–control study was conducted among 290 ATLI cases and 580 controls, of which causality assessment of ATLI cases was based on the updated Roussel Uclaf Causality Assessment Method (RUCAM). Conditional logistic regression was applied to calculate odds ratio (OR) and 95% confidence intervals (CIs), with weight and use of hepatoprotectant as covariates. The Bonferroni correction was used to adjust P values for multiple testing. Compared with those carrying rs6719561 TT genotype, patients with TC genotype had lower risk of ATLI (adjusted OR = 0.723, 95% CI: 0.531–0.985, P = 0.040). The haplotype TAG (rs3755319-rs2003569-rs4148323) could marginally significantly increase the risk of ATLI (adjusted OR = 5.071, 95% CI: 1.007–25.531, P = 0.049), while haplotype TC (rs4148329-rs6719561) could reduce the risk of ATLI (adjusted OR = 0.719, 95% CI: 0.527–0.982, P = 0.038). Patients with polymorphisms at rs4148328 or rs3755319 were at a reduced risk of moderate and severe liver injury under different genetic models. Based on this case–control study, genetic polymorphisms of UGT1A1 may be associated with susceptibility to ATLI in the Chinese anti-tuberculosis population.
Introduction
Tuberculosis (TB) remains a devastating disease and major global health scourge. Although first-line anti-TB regimens are effective in treating active TB, they are associated with many adverse drug reactions (ADRs). Anti-TB drug-induced liver injury (ATLI) is one of the most prevalent ADRs. 1 However, the pathogenesis of ATLI is still unclear, this idiosyncratic reaction has been found to have an association with genetic polymorphisms. 1
Uridine 5’-diphospho-glucuronosyl-transferases (UGTs) are membrane proteins that bind to the endoplasmic reticulum and are very important phase II metabolic enzymes responsible for approximately 40%–70% of endo and xenobiotic reactions. 2 UGTs catalyzes the transfer of glucuronic acid from UDP-glucuronic acid to other molecules (usually hydrophobic substrates). As one of the most crucial phase II metabolizing enzymes, UGT1A1 plays a major role in the biliary excretion of bilirubin. Hydrophobic bilirubin is a toxic product of heme metabolism that can be transformed into hydrophilic bilirubin in the liver through conjugation with uridine diphosphate glucuronic acid under the action of UGT1A1. The excretion of bilirubin from the body will be promoted if hydrophilicity of bilirubin is enhanced. Injured or reduced activity of UGT1A1 may induce unconjugated hyperbilirubinemia. 3 Thus, the function of glucuronidation reaction is accelerating the elimination of the toxic compounds, which play an important role in metabolic detoxification. 4 It is not difficult to speculate that genetic polymorphisms in UGT1A1 may affect an individual patient’s susceptibility to ATLI.
Since 2014, a cohort of anti-TB treatment patients was founded and followed according to the ADACS cohort research protocol 5 in Jiangsu province of China. Using this cohort, we performed a matched case–control study to explore the role of UGT1A1 polymorphisms in ATLI.
Materials and methods
Study population
The anti-TB treatment patients enrolled in this study were recruited from Jiangsu Province in China. From April 2014 to September 2016, newly diagnosed TB patients were recruited from the outpatients of hospitals (The Third People’s Hospital of Zhenjiang, The People’s Hospital of Taixing, and The Second People’s Hospital of Changshu). All patients were treated at home following a standard chemotherapy regimen with a combination of anti-TB drugs including isoniazid (INH), rifampin (RMP), pyrazinamide (PZA), ethambutol (EMB), and/or streptomycin (SM) for 6–9 months. Because ATLI primarily occurs in the period of starting treatment, liver enzyme tests were performed once every 2 weeks within 2 months of the initial treatment in all patients. If patients with continued sputum tested TB positive after the first 2 months, the period of strengthen treatment would be extended a month, and liver enzyme tests would be performed two more times. In addition, whether patients were using hepatoprotectants or not was recorded. Patients who preventively took hepatoprotectants along with anti-TB treatment before the occurrence of ATLI were categorized as using hepatoprotectants, while others were defined as not using hepatoprotectants.
Newly diagnosed pulmonary TB patients who received standard short-course chemotherapy, finished the anti-TB treatment, and signed the informed consent by themselves or surrogate, would be included in this study. In addition, patients were excluded if they had any of the following criteria: (1) patients with mental illness who could not finish the related epidemiological investigation; (2) Patients with a history of chronic diseases that may cause liver dysfunction, such as tumor and diabetes; (3) patients with critical medical conditions, such as cardiovascular and cerebrovascular, serious infection, trauma, surgery, and wasting disease; (4) patients with serious TB and/or coinfected with other diseases and survival time shorter than 6 months; (5) patients with abnormal serum alanine aminotransferase (ALT), aspartate aminotransferase (AST), or total bilirubin (TBIL) before anti-TB treatment; and (6) patients with chronic hepatitis B or other liver diseases that may cause liver dysfunction.
ATLI was defined as follows: (1) an increase to over two times the upper limit of normal (ULN) in ALT or a combined increase in AST and TBIL, provided one of them was more than two times above the ULN 6 and (2) causality assessment result was highly probable, probable, or possible based on the updated Roussel Uclaf Causality Assessment Method (RUCAM). 7 The clinical pattern of liver injury was classified into hepatocellular, cholestatic, or mixed types based on the ratio (R) of elevation of baseline ALT to baseline alkaline phosphatase (ALP). The severity of liver injury was classified into mild (ALT < 5 ULN), moderate (ALT 5–10 ULN), and severe (ALT ⩾ 10 ULN) according to the World Health Organization (WHO) toxicity classification standards.
In this study, patients meeting the criteria of ATLI were assigned into the case group. Controls were selected from those with sustained normal liver function throughout therapy. For each ATLI case, two controls were matched with the same time, place, age (within 5 years), sex, treatment history, disease severity, and drug dosage.
Informed consent was obtained from all individual participants included in the study. The study was approved by the ethical committee of the Nanjing Medical University.
SNP selection and genotyping
All eligible single-nucleotide polymorphisms (SNPs) in UGT1A1 gene including 2 kb upstream and downstream were downloaded from Chinese Han population (CHB) data available on International HapMap Project (http://hapmap.ncbi.nlm.nih.gov, HapMap Gene Brower release #24). Haploview 4.2 software (Broad Institute of MIT and Harvard, Cambridge, MA, USA) was applied to selected SNPs. SNPs were selected based on the following criteria: (1) minor allele frequency (MAF) ⩾0.10; (2) with r2 of pairwise linkage disequilibrium ⩾0.80. As much as possible, functional sites such as exons, promoter start sites, transcription factor binding sites, and so on were selected. As a result, nine tagSNPs (rs2003569, rs887829, rs8330, rs3755319, rs4148329, rs4148328, rs11563250, rs4148323, and rs6719561) in the UGT1A1 gene were selected.
Total genomic DNA was extracted by phenol-chloroform method and genotyped by Sequenom Mass Array allelic discrimination technology (Sequenom corporation, USA), blind to the case or control status. Around 10% of the samples were repeated using the same assay, and the results were completely concordant. The overall call rate of genotyping was more than 99%.
Statistical analysis
Normal distribution data were described as the mean ± standard deviation (SD), while skewed distribution data were described as median (inter-quartile range (IQR)). Differences between groups were analyzed by using two-factor analysis of variance test or non-parametric test. Categorized variables were described as a percentage and analyzed using the χ2 test. Hardy-Weinberg equilibrium was tested by a goodness-of-fit χ2 test. Haplotype blocks were selected with Haploview 4.2 software by considering linkage disequilibrium. Haplotype analysis was performed by PHASE 2.1 software. Genotype frequencies were compared in the case and control groups by conditional logistic regression analysis, and results were shown with odds ratios (ORs) and 95% confidence intervals (95% CI), with weight and use of hepatoprotectant as covariates. Subgroup analysis was conducted on the basis of the severity of liver injury. Bonferroni correction method was applied to adjust the P value for multiple comparisons of SNPs. A two-tailed P value of less than 0.05 was regarded as statistically significant. Statistical analyses were performed by SPSS for Windows software (version 20.0, IBM Inc).
Results
Characteristics of patients
A total of 290 patients with ATLI cases and 580 matched controls were included in the study. Among 290 ATLI cases, 24 cases got 4 score (8.3%), 150 cases got 5 score (51.7%), 100 cases got 6 score (34.5%), and 16 cases got 7 score (5.5%) based on the updated RUCAM. A total of 147 (50.7%) were classified as hepatocellular type, 16 (5.5%) as cholestatic type, and 32 (11.0%) as mixed type of liver injury, with the rest of 95 cases classified as unclear type due to lack of the ALP test results. A total of 217 (74.8%) ATLI cases had mild liver injury, 54 (18.6%) had moderate liver injury, and 19 (6.6%) had severe liver injury. Their baseline characteristics were summarized in Table 1. No significant difference was observed involving sex, age, treatment history, and baseline values of liver biochemical parameters (ALT, AST, and TBIL) between the two groups. However, the peak ALT, AST, and TBIL levels during the treatment were significantly higher in ATLI cases than those in controls (P < 0.001).
Characteristics of patients in ATLI cases and controls.
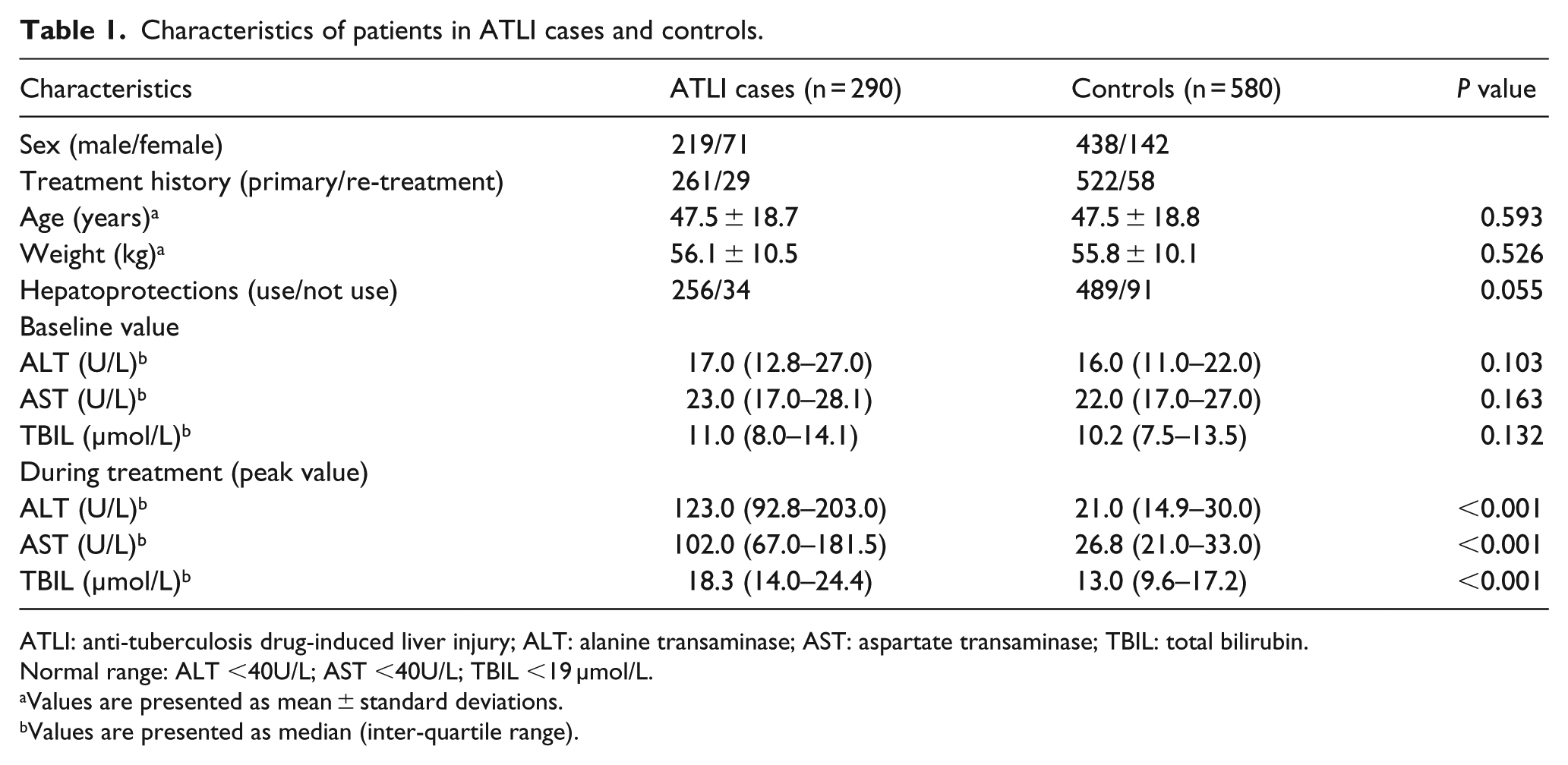
ATLI: anti-tuberculosis drug-induced liver injury; ALT: alanine transaminase; AST: aspartate transaminase; TBIL: total bilirubin.
Normal range: ALT <40U/L; AST <40U/L; TBIL <19 µmol/L.
Values are presented as mean ± standard deviations.
Values are presented as median (inter-quartile range).
Genotype analysis
Genotyping call rate was over 99.7% and all tagSNPs studied in UGT1A1 gene were in HWE in the controls, except the SNP rs8330. The distributions of genotypes for the selected tagSNPs of the UGT1A1 gene are shown in Table 2. Compared with those carrying rs6719561 TT genotypes, the patients with TC genotypes had a significantly lower risk of ATLI (adjusted OR = 0.723, 95% CI: 0.531–0.985, P = 0.040). In the dominant model, rs6719561 genotypes TC + CC were also associated with a lower risk of ATLI (adjusted OR = 0.727, 95% CI: 0.543–0.974, P = 0.032). How-ever, the above significance disappeared after Bonferroni correction.
Genotypes distribution of UGT1A1 in two groups and the risk of ATLI.

tagSNPs: tag single-nucleotide polymorphisms; UGT1A1: UDP glucuronosyl-transferase 1A1; ATLI: anti-tuberculosis drug-induced liver injury; Dom: dominant model; Rec: recessive model; Add: additive model; OR: odds ratio; 95% CI: 95% confidence interval.
Conditional logistic regression model analysis and adjusted for weight and use of hepatoprotectant.
Haplotype analysis
Linkage disequilibrium plot was reconstructed by using Haploview 4.2 software among nine tagSNPs of the UGT1A1 gene in 580 control subjects. By considering both D’ and r2, two haplotypes were analyzed. Compared with those carrying haplotype TGG (rs3755319-rs2003569-rs4148323), patients with haplotype TAG were discovered to have a higher risk of ATLI (adjusted OR = 5.071, 95% CI: 1.007–25.531, P = 0.049). In addition, patients with the haplotype TC (rs4148329-rs6719561) were at a lower risk of ATLI than those with the haplotype TT (adjusted OR = 0.719, 95% CI: 0.527–0.982, P = 0.038).
Subgroup analysis
Extended subgroup analysis was conducted according to different severity of liver injury. Patients with polymorphism at rs3755319 were at a reduced risk of moderate and severe liver injury under recessive model (adjusted OR = 0.261, 95% CI: 0.075–0.902, P = 0.034). Furthermore, patients with polymorphism at rs4148328 were also at a reduced risk of moderate and severe liver injury under recessive and additive model (adjusted OR = 0.359, 95% CI: 0.140–0.918, P = 0.032; adjusted OR = 0.632, 95% CI: 0.416–0.960, P = 0.032, respectively).
Discussion
In this study, genotype and haplotype analysis showed genetic polymorphisms in UGT1A1 might be associated with susceptibility to ATLI. However, only a few similar studies have been reported, with inconsistent results.8 –11 This discrepancy could be attributed to four reasons. First, different studies were conducted with different study designs and sample size, for example, a case–control study with 67 cases in Kim et al.’s 8 study, a 1:4 matched nested case–control study with 89 cases in Chen et al.’s 11 study, while 1:1 matched case–control study with 461 cases in Sun et al.’s 10 study. Second, the criteria for ATLI diagnosis were not identical across the studies. Although the criteria based on the international consensus from a 1989 meeting 6 were used for most of the related studies,8,9,11 including our study, the criteria in Sun et al.’s 10 study was according to the criteria established by the Chinese Medical Association. Third, a valid causality assessment is required to verify accurate drug-induced liver injury (DILI) cases. Because abnormal liver tests in patients under a drug therapy are more likely due to chronic liver disease than to DILI, the suspected DILI was probably not DILI. 12 The RUCAM was used in our study and Sun’s study, while causality assessment were not described in Kim et al.’s 8 study and Chang et al.’s 9 study. WHO-UMC used by Xia et al. 5 was another method which was suitable for all kinds of ADRs, but not liver specific. Fourth, the correction of multiple comparisons played a crucial role. Bonferroni correction was used in our study and Kim et al.’s 8 study, while no adjustment for multiple comparisons was used in Sun et al.’s 10 study.
The major strength of this study is a 1:2 matched case–control study which could control for the effects of some potential confounds as well as increase the statistical power. However, some limitation cannot be ignored. For example, there was a significant departure from the HWE in rs8330. Detection of ALP was not performed in all patients. We did not collect patients’ history of prior hepatitis C infection, a known risk factor for hepatotoxicity.
In conclusion, based on this matched case–control study, genetic polymorphisms of UGT1A1 might be associated with susceptibility to ATLI in the Chinese population.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by grants from the National Nature Science Foundation of China (grant no. 81373066).
