Abstract
Bisphenol A (BPA) is used in the production of polycarbonate plastics and epoxy resins and found in many consumer products. Previous studies have reported that perinatal exposure to BPA through the oral route promotes the development of allergic airway inflammation. We investigated the effects of exposure to low-dose BPA during the juvenile period of development on allergic airway inflammation. Six-week-old male C3H/HeJ mice were intratracheally administered ovalbumin (OVA, 1 μg) every 2 weeks and/or BPA (0, 0.0625, 1.25, and 25 pmol/animal/week) once per week for 6 weeks. Following the final intratracheal instillation, we examined the cellular profile of the bronchoalveolar lavage fluid, histological changes and expression of inflammatory/anti-inflammatory mediators in the lungs, OVA-specific immunoglobulin (Ig) production, serum corticosterone levels, and changes in the lymphoid tissues (mediastinal lymph node (MLN) and spleen). Exposure to OVA + BPA enhanced inflammatory cell infiltration and protein expression of Th2 cytokines/chemokines (e.g. interleukin (IL)-13 and IL-33) in the lungs, OVA-specific immunoglobulin E (IgE) production, the numbers of total cells and activated antigen-presenting cells (MHC class II+ CD86+, CD11c+), as well as the production of Th2 cytokines (i.e. IL-4 and IL-5) and stromal cell-derived factor-1α in MLN cells compared to OVA exposure alone. These effects were more prominent with 0.0625 or 1.25 pmol/animal/week of BPA. Furthermore, exposure to OVA + BPA altered serum levels of anti-inflammatory corticosterone, estrogen receptor 2 messenger RNA (mRNA) expression in the lungs and spleen functionality. These findings suggest that low-dose BPA exposure may aggravate allergic airway inflammation by enhancing Th2 responses via disruption of the immune system.
Keywords
Introduction
Bisphenol A (BPA) is a raw component of polycarbonate plastics and epoxy resins that are used to make products, such as food and drink packaging, the internal coating of cans, compact disks, building materials, and medical devices. 1 Due to its extensive number of applications, BPA is widespread in the environment and humans may be exposed to this chemical from the fetal period into adulthood.2–4
The prevalence of allergies is increasing dramatically worldwide due to changes in lifestyle and the environment, including an increased exposure to chemicals, particularly endocrine disruptors. 5 Humans can be exposed to these chemicals in conjunction with allergens. Moreover, environmental estrogens (e.g. BPA and phthalates) can potentially affect each step contributing to the development of allergic diseases (i.e. antigen presentation, Th2 polarization, immunoglobulin E (IgE) production, and mast cell degranulation). 6
In a review of published epidemiological studies, Robinson and Miller suggested that BPA exposure may lead to deleterious immunological changes, including the development of allergies and asthma via BPA exposure; albeit, the relationship between BPA exposure and the increasing incidence of allergy and asthma remains inconclusive. 5 Several human epidemiological studies have reported a positive association between BPA exposure and severe allergic asthma. 7 In addition, experimental studies using an animal model of allergic airway disease have reported that perinatal exposure to BPA through drinking water promotes allergic responses in pups, suggesting enhanced susceptibility during prenatal and breastfeeding periods.8–10 However, while Nygaard et al. 10 have also suggested that exposure to BPA through drinking water during the early stages of life augments allergic responses, these effects were observed at high doses.
The European Food Safety Authority (EFSA) has lowered the estimated safe level of BPA, known as the tolerable daily intake (TDI), from its previous level of 50 μg/kg/day to only 4 μg/kg/day. 11 Recent studies have raised concerns regarding the potential low-dose effects of BPA on the immune system, including an impaired systemic cellular immune response.4,12,13 The Dutch National Institute for Public Health and the Environment expert workshop concluded that the current EFSA TDI warrants reconsideration in the context of the currently available data on the adverse immune effects of BPA. 14
Potential exposure to BPA is predominantly through dietary ingestion; however, there are multiple routes of human exposure. BPA has been detected in indoor environments (0.2–17.6 μg/g in house dust) and the atmosphere (0.001–17.4 ng/m3); therefore, exposure via inhalation is also a concern.15–17 Thus, an evaluation of the potential health effects of BPA exposure through the intratracheal route is also required. The Ministry of the Environment of Japan has reported an estimated peak total exposure dose from the general atmosphere is 0.0003 μg/kg/day in Japanese people (concentrations ranging from less than 0.5–1 ng/m3). 18
The effects of co-exposure to allergen and low-dose BPA through the intratracheal route on allergic airway inflammation remain unclear. The aim of this study was to determine whether intratracheal exposure to BPA during the juvenile period of development at various concentrations, including low doses, has an impact on the development of allergic airway inflammation and immune cell function in mice.
Materials and methods
Animals
Five-week-old male C3H/HeJJcl mice were purchased from Japan Clea Co. (Tokyo, Japan) and housed in an animal facility maintained at 22°C–26°C and 40%–69% humidity under a 12-h light/dark cycle. The mice were fed a commercial diet (CE-2; Japan Clea) and given water ad libitum. All animal experiments followed all applicable NIH guidelines, were approved by the Animal Care and Use Committee, and conducted in accordance with the guidelines for the Care and Use of Laboratory Animals of the National Institute for Environmental Studies. All animals were treated humanely and did not suffer.
Study protocol
We developed a mouse model of allergic airway inflammation by repeatedly administrating ovalbumin (OVA) intratracheally. The three experimental doses of BPA used in this study were equivalent to 0.25, 5, and 100 times the estimated peak exposure dose from the general atmosphere in Japan of 0.0003 μg/kg/day. 18 The lowest dose was determined based on the minimum level, which is less than half of the estimated peak exposure from the general atmosphere. 18 Exposure doses were adjusted according to the body weight of the mice. Six-week-old mice were divided into eight experimental groups: (1) Vehicle; (2) 0.0625 pmol BPA/animal 25 g/week (BPA-L); (3) 1.25 pmol BPA/animal 25 g/week (BPA-M); (4) 25 pmol BPA/animal 25 g/week (BPA-H); (5) OVA; (6) OVA + BPA-L; (7) OVA + BPA-M; and (8) OVA + BPA-H. Mice were anesthetized with 4% halothane (Takeda Chemical Industries, Ltd, Osaka, Japan) and intratracheally instilled with 100-μL aliquots of an aqueous suspension. Mice were administrated the suspensions in accordance with shallow breathing under anesthesia and immediately awakened after instillation. The Vehicle group received phosphate-buffered saline (PBS, pH 7.4; Thermo Fisher Scientific, Inc., Chicago, IL, USA) containing 0.0005% ethanol (Nacalai Tesque, Inc., Kyoto, Japan) once per week for 6 weeks; the OVA group received 1 μg of OVA (10 μg/mL; Sigma-Aldrich Co., St Louis, MO, USA) dissolved in PBS containing 0.0005% ethanol every 2 weeks for a period of 6 weeks; the BPA groups received 0.0625, 1.25, or 25 pmol of BPA (Sigma-Aldrich Co., St Louis, MO, USA) in PBS containing 0.0005% ethanol once per week for 6 weeks; and the OVA + BPA groups received a combined administration of BPA and OVA (see Supplemental Figure S1).
Mice were sacrificed under anesthesia 4 h after the final intratracheal instillation and analyzed gene expression in the lung tissue by reverse transcription polymerase chain reaction (RT-PCR). We then measured several other parameters 24 h after the final intratracheal instillation, as outlined below.
Quantitation of inflammatory cell infiltration into the alveoli
We lavaged the lungs (5–8 animals per group) and collected the bronchoalveolar lavage (BAL) fluid to recover free alveolar cells, as previously described. 19 These cells were stained with Diff-Quik (International Reagents Co., Kobe, Japan), following which differential cell counts were made using Autosmear (Sakura Seiki Co., Tokyo, Japan). The lungs were retrieved immediately following BAL collection and stored at −80°C until further use.
Histopathological evaluation
We fixed the lungs in 10% phosphate-buffered formalin (pH 7.4). The tissue sections were then embedded in paraffin and cut into 4-μm-thick slices (four animals per group). Histological specimens were stained with hematoxylin and eosin (H&E) to evaluate eosinophil and lymphocyte infiltration in the airways, and periodic acid–Schiff (PAS) staining was used to evaluate goblet cell differentiation and mucus hyperproduction in the bronchial epithelium. We examined the entire area of each specimen and assessed the histological changes using an Olympus AX80 microscope (AX80; Olympus, Tokyo, Japan). The degree of eosinophil and lymphocyte infiltration or goblet cell differentiation and mucus hyperproduction was graded in a blind fashion as follows: 0 = not present, 0.5 = slight, 1 = mild, 1.5 = mild to moderate, 2 = moderate, 2.5 = moderate to marked, and 3 = marked. A score of 0.5 was defined as an inflammatory reaction affecting <10% of the airways or goblet cells stained with PAS, scores of 1 = 10%–20%, 1.5 = 20%–30%, 2 = 30%–40%, 2.5 = 40%–50%, and 3 = >50%, indicating the extent that the airways or goblet cells were affected.
Quantitation of cytokines and chemokines in the lung tissue
We removed the whole lungs (5–8 animals per group) immediately following BAL and prepared lung homogenate supernatants as previously described. 19 We converted 1 g of the homogenate suspension to 1 mL and calculated the protein levels of various cytokines and chemokines in the total lung homogenates. The levels of interleukin (IL)-4, IL-5, interferon gamma (IFN-γ), IL-12 (Thermo Fisher Scientific, Inc., Chicago, IL, USA), IL-10, IL-13, IL-33, macrophage-inflammatory protein 1-apha (MIP-1α), keratinocyte chemoattractant (KC), monocyte chemoattractant protein-1 (MCP-1), chemokine (C-C motif) ligand 5/regulated and normal T-cell expressed and secreted (CCL5/RANTES), CCL11/eotaxin (R&D Systems, Minneapolis, MN, USA), and monocyte-chemotactic protein 3 (MCP-3; PeproTech, Inc., NJ, USA) in the lung homogenate supernatants were measured using an ELISA kit, according to the manufacturer’s instructions. Cytokine and chemokine content were expressed as pg in the total lung homogenate supernatants.
Quantitation of antigen-specific Ig and corticosterone in the serum
Blood was sampled by cardiac puncture and the serum was collected (5–8 animals per group) and stored at −80°C until future use. We examined the level of OVA-specific Ig (OVA-IgE, OVA-IgG1, and OVA-IgG2a) and corticosterone in the serum. OVA-IgE was measured using a commercial mouse anti-OVA IgE ELISA kit (Shibayagi Co., Gunma, Japan) according to the manufacturer’s instructions, and OVA-IgG1 and OVA-IgG2a were measured by an ELISA with a solid-phase antigen, as previously reported. 20 Corticosterone was measured using a commercial ELISA kit (Endocrine Technologies, Inc., CA, USA), according to the manufacturer’s instructions.
Real-time RT-PCR analysis
Total ribonucleic acid (RNA) was extracted from the lungs (3–6 animals per group) using ISOGEN (Nippon Gene, Tokyo, Japan) and purified using an RNeasy® mini kit (Qiagen, Hilden, Germany) according to the manufacturer’s instructions. We assessed the total RNA concentration using a NanoDrop spectrometer (Thermo Fisher Scientific, Inc., Chicago, IL, USA). The total RNA was reverse transcribed to complementary DNA (cDNA) using a High-Capacity RNA-to-cDNA™ Kit (Thermo Fisher Scientific, Inc., Chicago, IL, USA). The messenger RNA (mRNA) expression of estrogen-related receptor gamma (
Preparation of lymph node cells and splenocytes
The mediastinal lymph nodes (MLNs; 5–8 animals per group) were pushed through a sterile stainless steel wire mesh in PBS (pH 7.4; Takara Bio Inc., Shiga, Japan). The MLN cells were collected following centrifugation at 400
Analysis of immune cell function
MLN cells (1 × 106/mL; 3–4 animals per group) or splenocytes (1 × 106/mL; four animals per group) were cultured with or without OVA (100 μg/mL) in 200 μL of R10 medium in 96-well flat-bottom plates. These cultures were performed in triplicate at 37°C in a 5% CO2/95% air atmosphere. After 3 days, we collected the culture supernatant and stored it at −80°C until future use. We added 5-bromo-2′-deoxyuridine to each well 20 h before measuring cell proliferation using an ELISA kit (Roche Molecular Biochemicals, Mannheim, Germany), according to the manufacturer’s instructions. We also measured levels of IL-4, IL-5, IFN-γ (eBioscience, Inc., CA, USA), and stromal cell-derived factor 1α (SDF-1α; RayBiotech, Inc., Norcross, GA, USA) in the MLN cell culture supernatant, as well as IL-4, IL-5, IFN-γ, and IL-10 (eBioscience, Inc., CA, USA) in the splenocyte culture supernatant using an ELISA kit, according to the manufacturer’s instructions.
Flow cytometry
We examined the expression of major histocompatibility complex (MHC) class II I-A/I-E, plasmacytoid dendritic cell antigen 1 (PDCA-1), CD86, and CD11c in the MLN cells (3–4 animals per group). For fluorescence-activated cell-sorting (FACS) analysis, we used the following monoclonal antibodies: I-A/I-E (2G9, Rat IgG2a κ FITC-conjugated; BD Biosciences, San Diego, CA, USA), PDCA-1 (JF05-1C2.4.1, rat IgG2b FITC-conjugated; Miltenyi Biotec GmbH, Bergisch Gladbach, Germany), CD86 (GL1, Rat IgG2a κ PE-conjugated; BD Biosciences, San Diego, CA, USA), and CD11c (HL3, Hamster IgG1 λ2, PE-conjugated; BD Biosciences, San Diego, CA, USA). The cells were incubated with each antibody for 30 min on ice, and fluorescence was measured on a FACSCalibur (Becton, Dickinson and Company, Franklin Lakes, NJ, USA), as previously reported.21,22
Statistical analysis
Data are expressed as the mean ± standard error (SE) of N observations. We tested the significance of differences between the groups using two-way analysis of variance followed by a Tukey–Kramer test or non-parametric Kruskal–Wallis analysis followed by Steel’s multiple comparison test (Excel Statistics 2010, Social Survey Research Information Co., Ltd., Tokyo, Japan). For the experiments limited to OVA administration, differences between the OVA + BPA groups and the OVA group were evaluated using one-way analysis of variance followed by a Dunnett’s test or non-parametric Kruskal–Wallis analysis followed by Steel’s multiple comparison test.
Results
Cellular profile in the BAL fluid
The differential cell counts in the BAL fluid are presented in Table 1. The BPA-M group had a significantly higher number of macrophages than the Vehicle group (
Cellular profile in the BAL fluid following exposure to BPA in a mouse model of allergic airway inflammation.
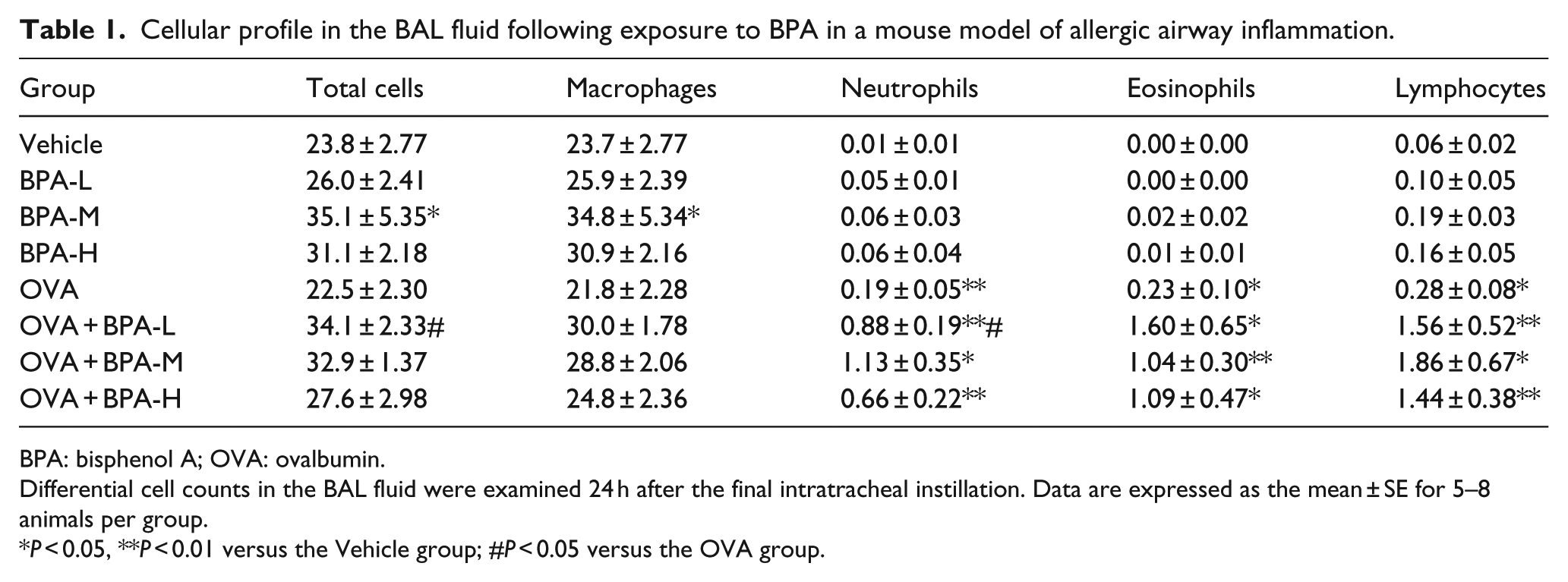
BPA: bisphenol A; OVA: ovalbumin.
Differential cell counts in the BAL fluid were examined 24 h after the final intratracheal instillation. Data are expressed as the mean ± SE for 5–8 animals per group.
Protein expression of cytokines and chemokines in the lungs
OVA administration alone significantly increased the expression of Th2 cytokines (i.e. IL-5, IL-13, and IL-33) and chemokines (i.e. MCP-1 and eotaxin) in the lungs compared to that following vehicle administration (IL-5, IL-13, MCP-1, and eotaxin,
Protein expression of Th1/Th2 cytokines in the lungs following exposure to BPA in a mouse model of allergic airway inflammation.

IL: interleukin; BPA: bisphenol A; OVA: ovalbumin.
The protein expression of Th1/Th2 cytokines in the supernatants of homogenized lungs were measured by ELISA 24 h after the final intratracheal instillation. Data are expressed as the means ± SE for 5–8 animals per group.
Protein expression of chemokines in the lungs following exposure to BPA in a mouse model of allergic airway inflammation.

BPA: bisphenol A; MIP-1α: macrophage-inflammatory protein 1-apha; KC: keratinocyte chemoattractant; MCP-1: monocyte chemoattractant protein-1; MCP-3: monocyte-chemotactic protein 3; OVA: ovalbumin.
Protein expression of chemokines in the supernatants of homogenized lungs were measured by ELISA 24 h after the final intratracheal instillation. Data are expressed as the means ± SE for 5–8 animals per group.
Histopathological findings in the lungs
No remarkable differences regarding pathological alterations in the lungs were observed in the Vehicle groups. OVA administration resulted in an increase in eosinophil and lymphocyte accumulation in the peribronchial and perivascular regions (OVA + BPA-L, OVA + BPA-M, and OVA + BPA-H groups,

Histological findings in the lungs. Histological changes in the lungs were investigated 24 h after the final intratracheal instillation. Representative photomicrographs of the lung sections and quantitative analyses of H&E staining (a) and PAS staining (b). The infiltration of eosinophils and lymphocytes into the peribronchial and perivascular regions was evaluated using H&E staining. Goblet cell differentiation and mucus hyperproduction in the bronchial epithelium was evaluated using PAS staining. Data are expressed as the means ± SE of four animals per group. *
OVA-specific Ig antibody production in the serum
The OVA groups had significantly higher levels of OVA-specific IgE (OVA group,

Production of OVA-specific Ig antibodies following exposure to BPA in a mouse model of allergic airway inflammation. OVA-specific IgE (a), OVA-specific IgG1 (b), and OVA-specific IgG2a (c) in the serum were measured by ELISA 24 h after the final intratracheal instillation. Data are expressed as the means ± SE for 5–8 animals per group. *
Level of anti-inflammatory mediators
The serum corticosterone levels were significantly lower in the OVA + BPA-L group compared to the OVA group (

Changes in the level of anti-inflammatory mediators following exposure to BPA in a mouse model of allergic airway inflammation. (a) Serum corticosterone. (b)
MLN cell phenotype and function
All the OVA groups, except the OVA + BPA-H group, had significantly higher total MLN cell numbers compared to those in the Vehicle group (

The number and activation of MLN cells following exposure to BPA in a mouse model of allergic airway inflammation. MLN cells were prepared 24 h after the final intratracheal instillation and the total cell number was counted. The expression of cell surface molecules and cytokine production in MLN cells from the OVA groups were performed by FACS and ELISA, respectively. The cytokine content in the culture supernatants was measured after the 66-h culture in the presence or absence of OVA. (a) Total cell number. (b) The percentage of CD86+ cells, MHC class II+ cells, and MHC class II+ CD86+ cells. (c) The percentage of CD11c+ PDCA-1+ cells (pDCs) and CD11c+ PDCA-1–cells (cDCs). (d) IL-4, (e) IL-5, (f) IFN-γ, and (g) SDF-1α. (a) Data are expressed as the means ± SE for 5–8 animals per group. (b–g) Data are expressed as the means ± SE for 3–4 animals per group. **
Spleen function
BPA exposure decreased the number of splenocytes in the groups that received OVA (OVA + BPA-M group vs OVA group,

Change in the spleen function following exposure to BPA in a mouse model of allergic airway inflammation. Splenocytes were prepared from the OVA groups 24 h after the final intratracheal instillation and total cell number was counted. The splenocytes were cultured for 64 h with or without OVA and then cell proliferation and cytokine production were measured by ELISA. (a) Total cell number, (b) cell proliferation, (c) IL-4, (d) IFN-γ, and (e) IL-10. Data are expressed as the means ± SE of four animals per group. #
Discussion
We investigated whether intratracheal exposure to low-dose BPA during the juvenile period of development affects allergic airway inflammation and immune cell function in mice. We found that BPA enhanced OVA-induced lung inflammation, as well as the Th2 response in the MLN; however, BPA exposure alone did not induce these inflammatory responses. The enhancement of allergic inflammation was greater following the lower doses of BPA compared to the higher dose, possibly due to former disrupting biological processes, including the immune system.
Previous experimental studies using animal models have reported that perinatal exposure to BPA promotes the development of allergic airway inflammation in pups.8–10 Although few animal models have documented BPA-mediated aggravation of airway allergy in adults, He et al. 23 demonstrated that the oral administration of high-dose BPA promotes OVA-induced lung eosinophilia.
This study is the first report to demonstrate that the intratracheal exposure to low-dose BPA during the juvenile period of development can disrupt the immune response and enhance allergic airway inflammation in an animal model. BPA exposure was found to enhance the level of OVA-induced infiltration of inflammatory cells (Table 1), the protein expression of Th2 cytokines and chemokines (Tables 2 and 3), and histopathological changes (Figure 1). Moreover, these effects were more prominent with OVA + BPA-L and/or OVA + BPA-M. The lung histology revealed that OVA-induced increases in inflammatory cell accumulation and goblet cell differentiation were enhanced following BPA exposure, which was particularly observed in the OVA + BPA-M group (Figure 1(a) and (b)). In the OVA-specific Ig antibody production, there were large differences according to each animal. This variation may be caused by the specificity/sensitivity of the ELISA kit and by interference from components in serum. Although it did not reach statistical significance, BPA exposure enhanced OVA-induced Ig production more than fourfold (Figure 2(a)–(c)). In particular, the levels of OVA-specific IgE were approximately sevenfold higher in the OVA + BPA-L and OVA + BPA-M groups compared to OVA alone (Figure 2(a)).
Th2 cytokines and chemokines play a key role in the development of allergic airway inflammation. 24 We demonstrated significant increases in IL-33 and RANTES in the lung tissue of the OVA + BPA-L and OVA + BPA-M groups (Tables 2 and 3). IL-33 is a member of the IL-1 cytokine family, has been shown to be a crucial regulator of mast cell function, 25 and can induce eosinophilic inflammation and airway hyper-responsiveness. 26 RANTES is a chemoattractant for T-cells, eosinophils, and basophils that has also been shown to induce eosinophilic inflammation and airway hyper-responsiveness. 27 Therefore, a low-dose BPA-mediated increase in the expression of Th2-prormoting cytokines and chemokines (e.g. IL-33 and RANTES) may aggravate OVA-induced allergic inflammation and airway hyper-responsiveness. We also undertook a preliminary examination of respiratory function using unrestrained whole-body plethysmography, which demonstrated that BPA exposure tends to induce an increase in breathing rate and the bronchoconstriction indicators PenH (enhanced pause) and pause without a methacholine challenge (data not shown).
As a next step, we examined the phenotypes and function of immune cells in the MLN. OVA + BPA-L and OVA + BPA-M induced the activation of antigen-presenting cells, including an increase in MHC class II and CD86 expression, as well as the cDC subset (Figure 4(a)–(c)). DCs are major antigen-presenting cells within the lungs and contribute to the development of asthma. It has been shown that OVA-loaded bone marrow-derived cDCs can induce allergic airway inflammation and hyper-responsiveness, whereas CD8α+β− and CD8α+β+ pDCs primarily induce regulatory T-cells that prevent this induction.
28
OVA + BPA-L resulted in significantly higher percentages of cDCs in the MLN compared to OVA alone (Figure 4(c)) due to the promotion of SDF-1α, a stromal-derived CXC chemokine that is a ligand for CXCR4. The expression of CXCR4 is low in immature DCs and subsequently upregulated during maturation.
29
Therefore, the observed increase in SDF-1α production (Figure 4(g)) may contribute to an increased activation of DCs in the MLN following BPA exposure. OVA + BPA also promoted the production of the Th1 cytokine IFN-γ in MLN cells re-stimulated with OVA
In the analysis of the splenocytes in the OVA groups, BPA exposure dose-dependently suppressed OVA-stimulating cell proliferation and Th1/Th2 cytokine production (Figure 5(b)–(d)), as well as increase the level of anti-inflammatory IL-10 production (Figure 5(e)). For instance, IL-10-producing regulatory B-cells can suppress allergic inflammation via regulatory T-cells. 30 BPA may induce IL-10-mediated suppressing function of spleen, which may be one explanation for the attenuation of allergic airway inflammation following a high-dose BPA exposure.
It has been also shown that the anti-inflammatory activity of glucocorticoids and estrogen receptor beta (ERβ),31–33 as well as the presence of mitochondrial glucocorticoids and ERβ in lung tissue cells is reduced in asthma.
32
Therefore, we examined these mediators and found that OVA + BPA-L significantly decreased the levels of serum corticosterone, a glucocorticoid, compared to OVA alone (Figure 3(a)). BPA-L alone also decreased the levels of serum corticosterone (24 ng/mL) compared to the Vehicle (105 ng/mL); however, the difference did not achieve statistical significance. These results indicate that a low dose of BPA can induce a suppressive effect on serum corticosterone levels. The gene expression of the glucocorticoid receptor in the lung was not altered by BPA exposure (data not shown). Regarding the estrogen-related nuclear receptors, OVA-induced suppression of
Consequently, the findings of this study indicate that a low dose of BPA aggravates the OVA-induced allergic reaction and a high dose of BPA may modulate the inflammation by induction of an anti-inflammatory response. In other words, low-dose BPA may promote allergic inflammation and high-dose BPA may induce immune suppression. In the promotion of allergic inflammation, our study suggests that BPA shows inverted U-shaped dose response that is a characteristic of endocrine-disrupting chemicals.
Numerous studies have demonstrated an association between the exposure to environmental estrogen and the development of asthma; moreover, environmental estrogens also modify each step of the allergic sensitization process.
6
In addition, ER expression is found on immunoregulatory cells including airway epithelial cells and immune cells.6,34 In the immune cells, BPA promotes DC differentiation
This study investigated the effects of intratracheal exposure to BPA to examine the local effects in airway allergy. We plan to investigate the effects of low-dose BPA in the diet during the juvenile period of development in a subsequent study. Since different duration and routes of chemical exposure may induce variable biological effects, it is important to use a range of experimental conditions considering environmental exposure. We are also currently investigating the effects of BPA exposure on the brain and nervous system in this murine model and intend to examine biological crosstalk and interactions in future studies to determine the mechanisms of BPA on allergy development.
In conclusion, this study demonstrated that intratracheal exposure to BPA during the juvenile period of development enhanced allergic airway inflammation and modified the expression of inflammatory and anti-inflammatory mediators in a murine model. These results indicate that low-dose BPA through the intratracheal route may aggravate allergic inflammation by enhancing Th2 responses via a disruption of the immune system.
Supplementary Material
Supplementary Material, Supplemental_Figure_S1 – International Journal of Immunopathology and Pharmacology
Supplementary Material, Supplemental_Figure_S1 for International Journal of Immunopathology and Pharmacology by Eiko Koike, Rie Yanagisawa, Tin-Tin Win-Shwe and Hirohisa Takano in International Journal of Immunopathology and Pharmacology
Footnotes
Acknowledgements
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Environment Research and Technology Development Fund of the Ministry of the Environment, Japan (5S2-12) and by the Advanced Research Programs, the National Institute for Environmental Studies, Japan (1115AA082).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
