Abstract
Asthma is a prevalent non-communicable disease that affects both children and adults, therefore it is important to constantly research new potential drugs that inhibit molecular targets involved in the pathogenesis of the asthmatic disease. In this experimental model, we induced chronic allergic airway inflammation by 28 days sensitization with ovalbumin (OVA). The oral administration of one or 10 mg/kg GSK429286 A lasted for two final weeks in OVA-sensitized animals. Firstly, we performed an in vivo analysis by measuring the number of coughs after citric acid inhalation and additionally measured the changes in specific airway resistance (sRaw) after inhalation of bronchoconstrictor histamine to show the effect of GSK429286 A on airway hyperactivity. Secondly, we focused on the anti-inflammatory effect of GSK429286 A by measuring the levels of inflammatory cytokines, namely IL-2, IL-4, IL-5, and IL-13 in the lung homogenates. Finally, analyze the markers of remodeling: collagen III and V, transforming growth factor betta 1 (TGF-β1), and smooth muscle actin (SMA) in lung homogenates, and in sections from lungs. Our results showed the significant effect of GSK429286 A in reducing sRaw, and a number of coughs during in vivo analysis. In addition, administration of GSK429286 A showed a reduction in the levels of inflammatory cytokines, IL-2, IL-4, and IL-5, as well as a reduction in remodeling markers, such as collagen III and V, and TGF-β1. These findings indicate that oral administration of GSK429286 A, which inhibits Rho-kinase pathway, has a significant therapeutic effect on allergic asthma in guinea pigs.
Background
Allergic asthma is a chronic inflammatory disease characterized by airway hyperresponsiveness, inflammation and remodeling. ROCK inhibitors have now been shown to have the potential to alleviate these symptoms, although the specific effects of a new ROCK inhibitor, GSK429286 A, remain underexplored.
Objective
The aim of this study was to evaluate the therapeutic effects of a novel ROCK inhibitor, GSK429286 A, which exhibits a high affinity for both ROCK1 and ROCK2 isoforms, on allergic asthma in a guinea pig model, focusing on its effects on airway hyperresponsiveness, inflammation, and remodeling
Methods
To induce allergic asthma, guinea pigs were sensitized with ovalbumin for 28 days, and in the middle of sensitization they were treated with different doses of the RoCK inhibitor, GSK429286 A. The study evaluated the effect of the administered doses on the reduction of airway hyperresponsiveness, by measuring specific airway resistance (sRaw), and the number of coughs after citric acid inhalation. We also monitored the anti-inflammatory effect by measuring levels of inflammatory cytokines, IL-2, IL-4, IL-5, IL-13, and remodeling markers, such as collagen deposition, and goblet cell hyperplasia. In addition, we monitored the possible anti-remodeling effect of GSK429286 A by histopathological examination.
Results
The ROCK inhibitor, GSK429286 A, showed an effect on suppressing airway hyperresponsiveness by reducing sRaw and the number of coughs in treated guinea pigs compared to controls. Our investigated drug suppressed the release of key mediators of inflammation, including IL-2, IL-4, and IL-5, thus demonstrating the effect of this ROCK inhibitor on the suppression of inflammation in the airways. Finally, GSK429286 A reduced markers of airway remodeling such as collagen deposition and goblet cell hyperplasia.
Conclusion
GSK429286 A, an inhibitor of the ROCK pathway, exhibits significant anti-inflammatory and antiremodeling effects in a guinea pig model of allergic asthma. Indeed, we demonstrate its effect on suppressing airway hyperreactivity and reducing cough frequency. These findings suggest that GSK429286 A may be a promising therapeutic agent for allergic asthma, although further studies are needed to investigate its long-term efficacy, underlying mechanisms, and optimal dosing strategy.
Keywords
Introduction
Bronchial asthma is a chronic inflammatory respiratory disease, with a rising prevalence, affecting approximately 300 million people worldwide. 1 Typical features of asthma are airway hyperresponsiveness, airway obstruction, increased mucus production, and increased inflammatory and remodeling markers. 2 All these factors together lead to the development of symptoms like wheezing, coughing, and shortness of breath, which reduces the quality of the patient’s life. Currently, pharmacotherapy of asthma is insufficient because there is not existing universal drug with bronchodilator, anti-inflammatory and anti-remodeling effect. 3 Therefore, it is necessary to investigate new drugs for asthma treatment, and our attention was drawn to Rho-associated kinases.
Rho-associated kinases are also called Rho-associated coiled-coil forming kinases (ROCK/Rho). They are a part of the serine-threonine specific protein kinases family AGC, which is named after protein kinases A, G, and C. Two isoforms of Rho-associated kinases have been discovered, namely ROCK1, and ROCK2. 4 Their activation is mediated by the bonding with GTP-RhoA, subsequent initiation of molecular cascades leads to the inhibition of the airway smooth muscle (ASM) relaxation and causes airway hyperresponsiveness. 5 Research indicates, that ROCK inhibitors could cause a reduction in airway smooth muscle constriction in vivo 6 and also in vitro.7,8 Indeed, an increase in ROCK expression was demonstrated in the lungs of the experimental animal model of allergic inflammation. There is evidence of the anti-inflammatory effect of ROCK inhibitors through affecting inflammatory cells, such as eosinophils, 9 macrophages, 10 mast cells 11 and T cells, 12 which participate in the pathogenesis of asthma. ROCK inhibitors have been shown to have the anti-inflammatory effect in mouse models of allergic airway inflammation,13,14 and in guinea pig model. 15
In addition, pharmacological inhibition of the ROCK pathway has previously been demonstrated to suppress lung fibrosis and airway remodeling in a mouse,16,17 guinea pig, 18 and rat 19 model of allergic asthma and lung fibrosis. Also, in our previous study, we demonstrated an anti-remodeling effect of ROCK inhibitor hydroxyfasudil, by decreasing collagen type III, V, transforming growth factor-beta1 (TGF-β1) and epidermal growth factor receptor in an experimental model of allergic asthma in guinea pigs. 20 Furthermore, the anti-inflammatory effect, anti-remodeling effect, and decreased airway hyperresponsiveness were demonstrated in ROCK-knockout mice sensitized by ovalbumin (OVA). 21
To verify the effects of ROCK inhibitors, this research examined the anti-asthmatic activity of a new ROCK inhibitor, GSK429286 A, in an experimental model of allergic asthma in guinea pigs. The anti-asthmatic activity was investigated by measuring airway hyperresponsiveness in vivo
Material and methods
Animals
The approved animal breeding facility Envigo (Netherlands) provided male Dunkin-Hartley guinea pigs, that were used to develop the model of allergic asthma. The weight of guinea pigs was from 200 to 250 g. The standard laboratory conditions for animals were kept: the air temperature 22 ± 2°C, relative humidity from 50% to 60%, 12 h light mode day and night, continual access to clean water and feed ad libitum, and 7 days quarantine after arrival.
Chemicals
Tested Rho-kinase inhibitor GSK429286 A (Rho-inh.) was obtained from MedChemExpress (MedChemExpress, New Jersey, USA). Aluminium hydroxide (Al (OH3) serves as an adjuvant for ovalbumin sensitization (Centralchem, Bratislava, Slovakia). Histamine (histamine dihydrochloride), citric acid, and ovalbumin from chicken egg albumin grade V were obtained from Sigma Aldrich Chemicals (US). All chemicals for nebulization were dissolved in 0.9% saline.
Experimental design and treatment protocol
The allergen, in our case ovalbumin, served to induce chronic inflammation in the lungs. The anti-asthmatic effect of GSK429286 A, the Rho-kinase inhibitor, was studied. The anti-asthmatic activity of GSK429286 A was compared with OVA-sensitized animals.
The guinea pigs were randomly divided into the following groups: (1) Healthy control group (OVA -): eight animals administered 0.9% saline for 28 days. (2) Positive control group (OVA +): eight animals sensitized with ovalbumin (OVA) for 28 days. (3) First therapeutic group (GSK429286A):– eight animals sensitized with OVA for 28 days and treated daily with GSK429286A (1 mg/kg, per os) for the last 14 days. (4) Second therapeutic group (GSK429286 A): eight animals sensitized with OVA for 28 days and treated daily with GSK429286A (10 mg/kg, per os) for the last 14 days.
The animals were humanely killed 1 day after the last OVA inhalation and treatment. Immediately, after being sacrificed the lung samples were washed in DPBS and stored at −80°C.
Protocol for the development of allergic asthma in Guinea pigs
The protocol for the development of allergic inflammation in the airways of guinea pigs was described in previous publication,
22
and is shown in Figure 1. The 28-day experimental protocol for allergic airway inflammation in guinea pigs. OVA was administered subcutaneously (s.c.), intraperitoneally (i.p.), and by inhalation (inh.). For intraperitoneal and subcutaneous application 0.5 mL/animal of the solution consisting 5 mg of OVA and 100 mg Al (OH)3 in 10 ml of 0.9% saline (OVA solution) was used. OVA solution was applied i.p. on the 1st and 4th day, s.c. on the 1st and 8th day, and by inh. in days 12 and 15 (2.0 mg/mL OVA); days 18 and 20 (5.0 mg/mL OVA); days 23, 25 and 27 (10.0 mg/mL OVA). The inhalation with OVA lasted for 3 min and was mediated by a wholebody double-chamber plethysmograph HSE type 855 (Hugo Sachs Elektronik, Hugstetten, Germany) using a Pari Jet Nebuliser (Paul Ritzau, PariWerk-GmbH, Germany) with output 5 L/s, and particle mass median diameter 1.2 μm. OVA – group represents healthy animals, whose were treated without OVA just with saline at the same doses.
Airway reactivity under in vivo conditions
For the measurement of sRaw, which demonstrates in vivo changes in the smooth muscle contractility the double-chamber plethysmograph HSE type 855 (Hugo Sachs Elektronik, Hugstetten, Germany) was used. The conscious guinea pigs were inhaled for 30 s with histamine (0.1 mM) using a Pari Jet Nebuliser (Paul Ritzau, PariWerk-GmbH, Germany) with output 5 L/s, and particle mass median diameter 1.2 μm. After each inhalation, the pressure changes in the air between the two body-plethysmograph chambers serve for the calculation of the average in vivo sRaw values using HSE Pulmodyn Pennock plethysmograph software (HSE type 855, Hugo Sachs Elektronik, Hugstetten, Germany). 23
The number of coughs under in vivo conditions
The double-chamber plethysmograph HSE type 855 (Hugo Sachs Elektronik, Hugstetten, Germany) was used to measure the number of coughs under in vivo conditions. The conscious animals were irritated with 0.3 M citric acid aerosol for 3 min. The citric acid aerosol was generated by a Pari Jet Nebuliser (Paul Ritzau, PariWerk-GmbH, Germany) with an output of 5 L/s, and particle mass median diameter 1.2 μm. To determine the count of coughs while inhaling citric acid aerosol, the number of peaks in the basic expiratory airflow was employed. The number of peaks was mediated through a pneumotachograph, which was connected to the nasal chamber of the body plethysmograph. Coughing in guinea pigs is also accompanied by animal movement. For this reason, the number of coughs from the pneumotachograph was confirmed by the observation of two experienced observers.
ELISA kits for TGF-β1; collagen III, V; SMA, NF-κB in lung tissue homogenates
Total protein concentrations for TGF-β1; collagen type III, and V; and for NF-κB in the homogenates from lung tissue were determined using the DCA protein assay (Bio-Rad, Hercules, California, US). The absorbance of the homogenates was measured spectrophotometrically at 750 nm by using a Varioskan Flash Spectral Scanning Multimode Reader (Thermo Fisher Scientific, Waltham, Massachusetts, US). Final total protein concentration in the homogenates was calculated according to the calibration curve, where bovine serum albumin (μg/mL) was used as a standard.
The level of monitored proteins was measured by commercially available ELISA kits, namely: Guinea pig Transforming Growth Factor Beta, Guinea pig Collagen Type III, Guinea pig Collagen Type V, Guinea pig Smooth muscle actin, Guinea pig nuclear factor kappa b (NF-κB), (MyBioSorce, San Diego, USA). All analyses were performed according to the manufacturer´s instructions.
The homogenates for TGF-β1, collagen type III and V; SMA were prepared by weighing 100 mg from the lung tissue. The tissue was washed in ice-cold 0.01 M phosphate buffer (PBS) with pH = 7.0–7.2 and immediately homogenized with 1000 μL of lysing solution (Tissue Extraction Reagent I, Invitrogen, Thermo Fisher Scientific, Massachusetts, US) with protease inhibitors (Protease Inhibitor Cocktail, Sigma Aldrich, US) using Laboratory Homogenizer Stuart SHM2/EURO (Bibby Scientific, UK). The homogenates were centrifuged at 7500 G for 10 min at 4°C, the supernatant was taken up and stored in sterile tubes at −80°C. The homogenate for NF-κB was prepared by mixing washed 500 mg lung tissue with 500 µl of PBS. The prepared homogenates were subjected to two freeze-thaw cycles and were rapidly centrifuged at 1500 G for 15 min at room temperature. The created supernatants were collected, aliquoted and stored in tubes at −80°C.
The created absorbances were measured spectrophotometrically at 450 nm on the Varioskan Flash Spectral Scanning Multimode Reader (Thermo Fisher Scientific, Waltham, Massachusetts, US). The final concentrations were analyzed by calibration curve with the software SkanIt Software for Varioskan Flash version 2.4.5 (Thermo Fisher Scientific, Waltham, Massachusetts, US).
Bio-Plex analysis of inflammatory cytokines
The homogenates from lung tissue were prepared according to the company´s protocol by using a commercial lysate kit (Bio-Plex Cell Lysis Kit, Bio-Rad, Hercules, California, US). Subsequently, the homogenates were centrifuged at 4500G for 4min at 4°C on MICRO 220R Centrifuge (Hettich GmbH, Tuttlingen, Germany). Created supernatants were used for qualitative and quantitative analysis of inflammatory cytokines (IL-2, IL-4, IL-5, IL-13) by Bio-Plex 200 Protein Array System and Bio-Plex ProTM Human Cytokine Th1/Th2 Assay (Bio-Rad, Hercules, California, US). All steps of the assay were performed according to the manufacturer´s instructions. The Bio-Plex Manager 6.0 software (Bio-Rad, Hercules, California, US) was used to evaluate the obtained data. The concentration of obtained inflammatory cytokines is expressed in pg/ml.
Immunohistochemical staining of the lungs from guinea pigs
The left lungs were washed with DPBS and fixed with formalin solution for 48 h. The Sirius red staining was used for the presence of collagen in bronchioles, antibodies against SMA were used for the detection of smooth muscle mass in the bronchioles, and antibodies against MUC5AC were used for the presence of mucus cells in the walls of bronchioles. The fixed tissue was dehydrated through ascending grades of ethanol baths, impregnated with paraffin and cut into 6 μm thick sections for the Picrosirius red staining and into 4 μm for detection of SMA and MUC5AC in the bronchioles. All mentioned chemicals were performed from Sigma Aldrich (US).
Detection of the amount of collagen in bronchioles by Sirius red: Paraffin embedded sections were deparaffinized by xylene and descending ethanol baths and distilled water. Deparaffinized slides were incubated with Weigert’s hematoxylin for 8 min and then removed by water for 10 min. Then, the slides were stained in Picrosirius red solution (Sigma Aldrich, US) for 1 h. After 1h, the slides were washed twice with 1% acetic acid solution, and dehydrated in pure ethanol and xylene. In the last step, dehydrated slides were mounted with Entellan (Millipore Sigma, Bur-lington, MA, USA). The results of the staining were collagen fibers visible with red colour, cell nuclei with black colour, and background with yellow colour. The Olympus BX43 microscope (Olympus, Tokyo, Japan) was used for visualization of the slides, and Zen software (Carl Zeiss, Germany) for the semi-quantification of collagen fibers and for measurement of airway wall thickness. 24 The signal from collagen from negative control served as a 100% and all groups were compared to negative control.
Detection of smooth muscle mass by SMA antibodies and mucus mass by MUC5AC antibodies: the paraffin embedded sections were deparaffinized, revitalized, and rehydrated. After these steps, the slides were incubated with 3% peroxide solution for 10 min at room temperature to block endogenous peroxidases, and then washed with 50 mM Tris solution supplemented with 0.1% TRITON-X 100 (Sigma) (TBS-T). The primary rabbit antibodies against SMA (Abcam, UK), and primary mouse antibodies against MUC5AC (Abcam, UK) were diluted 1:250 in TBS-T and applied individually to the sections for 2 h at room temperature. The slides were then washed and incubated for 10 min with LSAB2 System-HRP (Dako, Denmark). The slides were then washed with TBS-T and incubated for 5 min with DAB substrate (Dako, Denmark), counterstained with Mayer´s hematoxylin (Bio-optica, Italy) and mounted with Entellan medium (Merck, USA).
The sections were viewed with an Olympus BX43 microscope (Olympus, Japan) equipped with a photo camera Canon E0S 2000D. We used the Quick Photo Micro program, version 3.2 (Olympus) to image capture the sections and measure the thickness of the smooth muscle layer in the bronchiolar wall (dark brown cytoplasm of SMA-positive smooth muscle cells). 25 Quantitative analysis of MUC5AC expression was evaluated using the public domain Image J program (National Institute of Health, Bethesda, MD, USA) using the cell counter function. A total of 168 fields of view were analyzed. The numbers of MUC5AC positive goblet cells in experimental groups were counted in the 93 and 75 sections, respectively. To measure the area occupied by MUC5AC-positive cells in the defined region of interest, goblet cells in the bronchioli of lung parenchyma (dark brown secretory granules in the cytoplasm of MUC5AC- positive cells) were selected (sampling grid size: 0.06 x 0.06 cm). 26 All histopathological evaluations were performed by an independent histologist in a blinded way.
Ciliary beat frequency analysis by high-speed video-microscopy
Our diagnostic approach includes a brush biopsy technique in in vitro conditions. Cytology brush rotated within the trachea is used to obtain a sample of the tracheal cilia found here. Once the sample is collected, the brush is placed into a basal medium (RPMI-1640 medium). In the next step, the biological material is transported from cilia suspension on the microscopic slide for preparing specimens to examine ciliary beat frequency (CBF). The stable temperature (37°C) of both cilia suspension and microscopic slides is maintained using TS-100C, Thermo-Shaker for microtubes and PCR plates (SIA Biosan, Latvia).
The aim of this method is to make approximately up to 10 5-s-long video-records of the same microscopic preparation making them at 1-min intervals for which structural integrity of the epithelium is required.
High-speed imaging with high-speed video camera (Basler A504kc; Basler AG, Germany) connected to an inverted phase contrast microscope (Zeiss Axio Vert. A1; Carl Zeiss AG, Göttingen, Germany) and computer is used for the evaluation of specimens microscopically using a 40x objective lens providing a total magnification of 400x.
Ciliary Analysis software (LabVIEW™) constantly used by us for high-speed video microscopy analysis helps with image processing and detects the region of interests (ROIs) near the surrounding of beating cilia based on the differences in grey-level intensity. The intensity variance curve of selected suitable ROIs recording in time is then processed by the fast Fourier transformation (FFT) algorithm for automated and real-time analysis of movement kinetics. The output of a Fourier Transform is a frequency spectrum of ciliary beating in selected ROIs which corresponds to each intensity variance curve. 27
Statistical analysis
The presented data are in the form of means ± SEMs. One-way Anova analysis using GraphPad Prism 9.3.1 software (GraphPad, USA) was performed to determine statistical significance. Statistical significance was considered if the
Results
The measurement of specific airway resistance (sRaw) after histamine inhalation
The value changes in sRaw were measured after inhalation of 0.1 mM histamine during 30 s in guinea pigs under in vivo conditions on the third and fourth week of OVA-sensitization (Figure 2). The administration of 10 mg/kg GSK429286 A (Figure 2, Rho-inh. 10) for last 14 days of OVA sensitization decrease significantly the values of the sRaw against the animals sensitized only with OVA (Figure 2, OVA +) in the fourth week of experiment. However, the changes in sRaw were not significantly decreased in sensitized animals with saline (Figure 2, OVA -) compared to OVA + group. Effect of GSK429286 A on specific airway resistance (sRAW) in vivo conditions in experimental model of allergic asthma in guinea pigs. The measurement of the change’s values in sRaw after inhalation of 0.1 mM histamine for 30s after 3 weeks and 4 weeks inhalation of OVA. OVA – group represents guinea pigs sensitized with saline for 28 days and OVA + with ovalbumin. Administration of inhibitor of Rho-kinase, GSK429286 A, in concentration of 1 mg/kg (Rho-inh. 1) and 10 mg/kg (Rho-inh. 10) per os was carried out on the 15th day of sensitization. Data are shown as average ± SEM; *p<0,0.05 OVA + versus tested substance.
The number of cough reflex responses in vivo
To determine the number of coughs, the conscious animals were inhaled with 0.3 M citric acid for 3 min. The highest number of coughs was monitored in the animals sensitized with OVA (Figure 3(a), OVA +). Fourteen-day treatment with 10 mg/kg GSK429286 A (Figure 3(a), Rho-inh. 10) in OVA-sensitized animals resulted in a significant reduction in cough effort versus OVA + group, but the lower concentration of 1 mg/kg of GSK429286 A (Figure 3(a), Rho-inh. 1) had no effect on the number of coughs on 28 day of the experiment. Effect of GSK429286 A on cough reflex response (A) and on ciliary beating frequency (CBF, B) in experimental model of allergic asthma in guinea pigs. The number of coughs after 3 min inhalation of 0.3 M citric acid solution was measured after 3 weeks and 4 weeks inhalation of OVA (A). Ciliary beating frequency was measured immediately after killing of animals (B). OVA – group represents guinea pigs sensitized with saline for 28 days and OVA + with ovalbumin. Administration of inhibitor of Rho-kinase, GSK429286 A, in concentration of 1 mg/kg (Rho-inh. 1) and 10 mg/kg (Rho-inh. 10) per os was carried out on the 15th day of sensitization. Data are shown as average ± SEM; *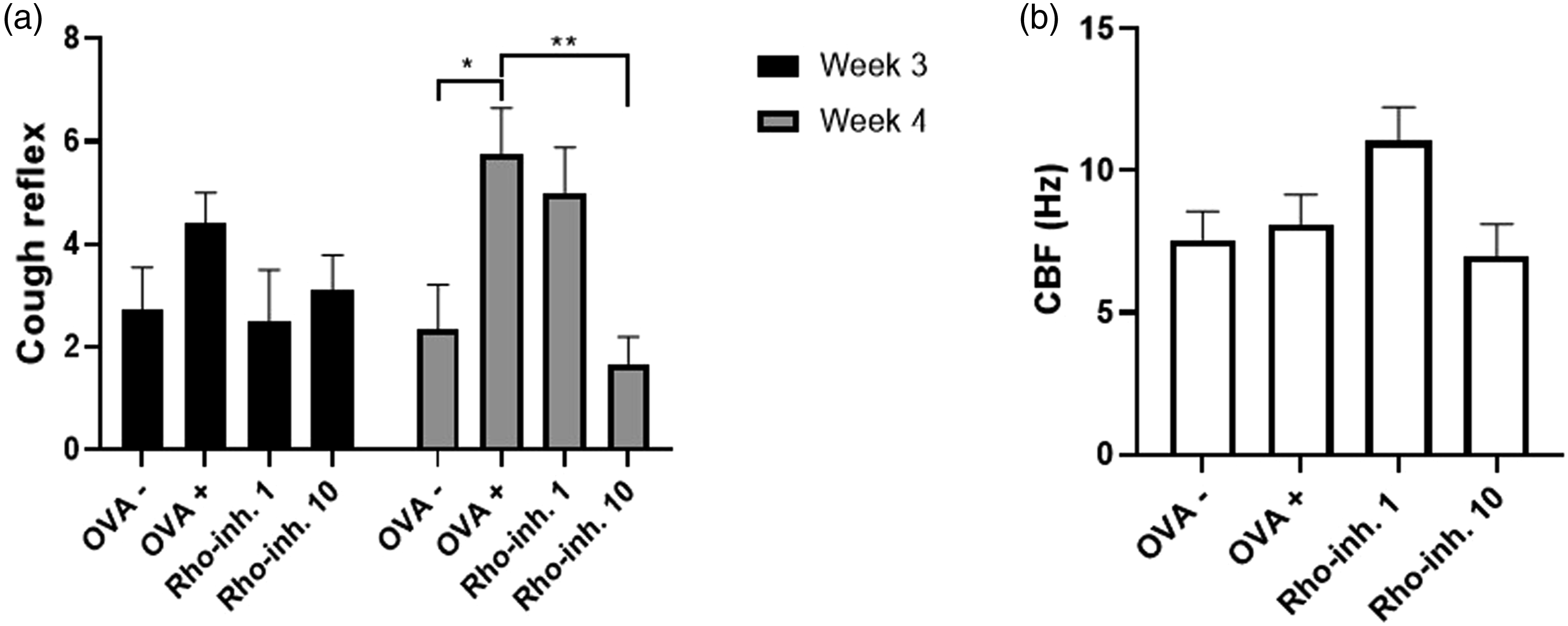
The measurement of ciliary beating frequency in vitro
The ability of effective ciliary movement was evaluated by monitoring the changes in CBF under in vitro conditions (Figure 3(b)). CBF was slightly increased after the administration of 1 mg/kg GSK429286 A (Figure 3(b), Rho-inh. 1) in OVA-sensitized animals, but on the other hand this effect was not observed in animals treated with administration of higher concentration GSK429286 A (Figure 3(b), Rho-inh. 10) compared to animals sensitized with saline (Figure 3(b), OVA -) or with ovalbumin (Figure 3(B), OVA +).
Nuclear factor kappa B (NF-κB) concentration in lung tissue
To demonstrate the effect of GSK429286 A on the expression of transcription factors we measured the concentration of NF-κB in the homogenates from the lungs. The obtained data indicate an increased concentration of NF-κB in OVA-sensitized animals for 28 days (Figure 4, OVA +). In the comparison with other groups, namely: healthy animals (Figure 4, OVA -), and 28 days OVA-sensitized animals with administration of GSK429286 A with concentration of 1 mg/kg (Figure 4, Rho-inh. 1) and with 10 mg/kg (Figure 4, Rho-inh. 10) for the last 14-days of sensitization, the level of NF-κB was significantly increased. The concentration of nuclear factor kappa b (NF-κB) in lung tissue homogenates. The concentration of transcription factor NF-κB was measured by ELISA and subsequently quantified for the appropriate amount of proteins. OVA – group represents unsensitized guinea pigs treated with saline for 28 days and OVA + with ovalbumin. Administration of inhibitor of Rho-kinase, GSK429286 A, in concentration of 1 mg/kg (Rho-inh. 1) and 10 mg/kg (Rho-inh. 10) per os was carried out on the 15th day of sensitization. Data are shown as average ± SEM; *
Cytokine concentration in lung homogenates
To analyze the inflammatory cytokines in lung tissue we measured the level of Th2-cytokines, specifically IL-4, IL-5, IL-13 and, in addition, the level of IL-2. The results showed a significant increase in IL-2 (Figure 5(a), OVA +), IL-4 (Figure 5(b), OVA +), and IL-5 (Figure 5(c), OVA +) in samples from OVA-sensitized animals compared to samples from healthy animals (Figure 5(a)–(c), OVA -). The administration of 10 mg/kg GSK429286 A for the last 14 days of sensitization significantly decreased the level of cytokines, in this case IL-2 (Figure 5(a), Rho-inh. 10), IL-4 (Figure 5(b), Rho-inh. 10), and IL-5 (Figure 5(c), Rho-inh. 10) compared to Ova-sensitized animals. Additionally, we did not observe significant changes in levels of IL-13 in lung tissue from OVA-sensitized animals compared to the other groups (Figure 5(d)). The concentration of cytokines, namely IL-2, IL-4, IL-5 and IL-13 in lung tissue homogenates. The concentration of cytokines was measured by ELISA and subsequently quantified for the appropriate amount of proteins. OVA – group represents unsensitized guinea pigs treated with saline for 28 days and OVA + with ovalbumin. Administration of inhibitor of Rho-kinase, GSK429286 A, in concentration of 1 mg/kg (Rho-inh. 1) and 10 mg/kg (Rho-inh. 10) per os was carried out on the 15th day of sensitization. Data are shown as average ± SEM; *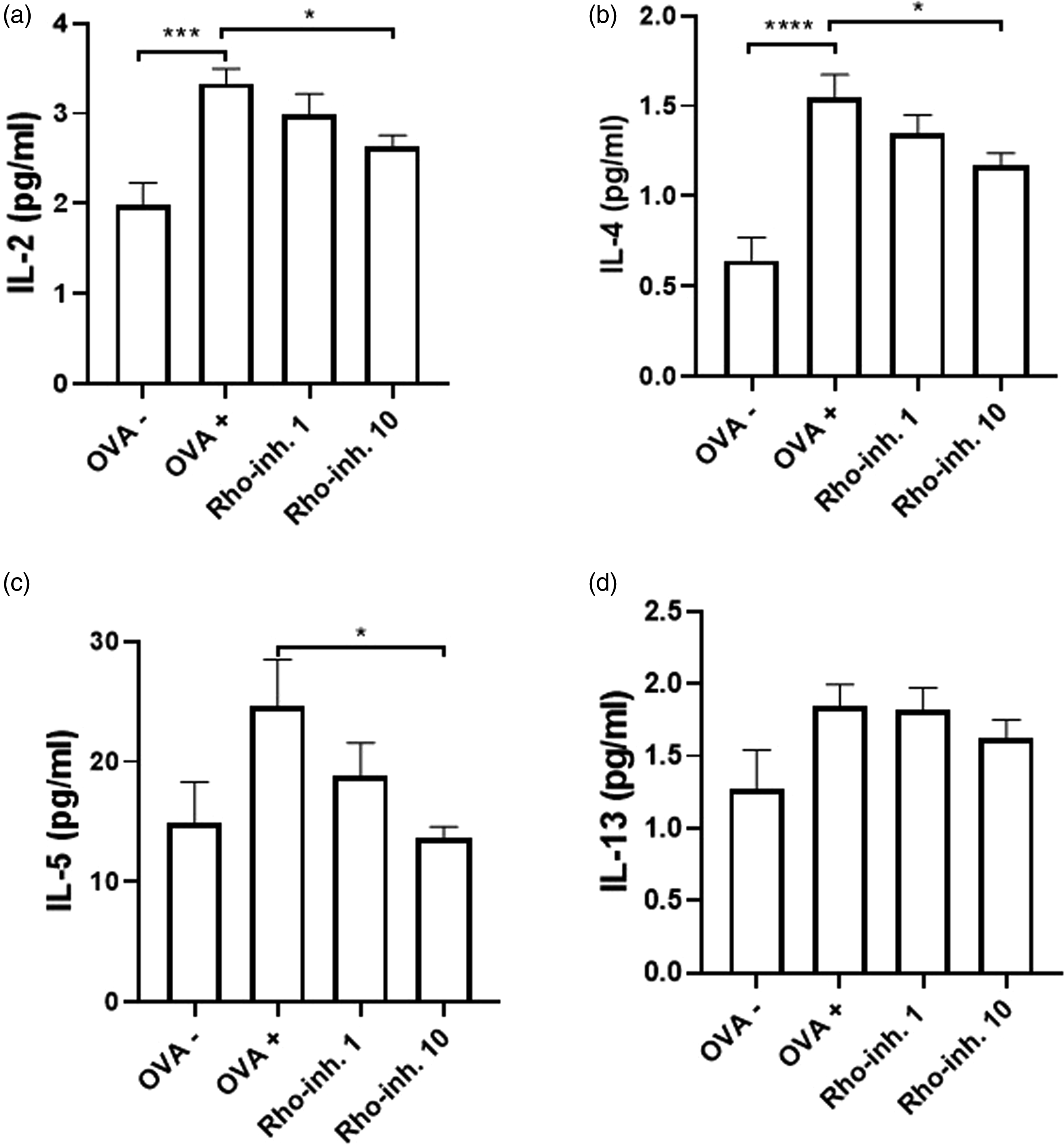
Immunohistochemical detection of collagen I and III, SMA, and MUC5AC in the lung sections
Immunohistochemical detection of collagen I and III accumulation in lung bronchioles was performed by Picrosirius red staining (Figure 6, Sirius red staining; Supplement 1). The highest amount of collagen I and III was visualized (Figure 6, OVA +) and calculated (110%, Figure 6(a), OVA+) in sections from the lungs of OVA-sensitized animals. The amount of collagen in lung sections from animals treated with saline (100%, Figure 6(a), OVA -) and animals with administration of 10 mg/kg GSK429286 A (100,9%, Figure 6(a), Rho-inh. 10) was significantly decreased compared to OVA + group. On the other hand, there were no significant changes in smooth muscle mass (Figure 6(b); Figure 6, SMA; Supplement 2), probably due to short-term OVA sensibilization. The number of positive mucus cells (Figure 6, MUC5AC; Supplement 3) was calculated by public domain Image J (Figure 6(c)). The amount of mucus in the bronchioles of saline-treated animals (Figure 6, OVA -) was significantly reduced compared to OVA + group (Figure 6(c)), but administration of 10 mg/kg GSK42926 A on day 14 of sensibilization had no effect on the amount of mucus compared with the OVA - group. Immunohistochemical determination of the collagen I and III by Picrosirius red staining, amount of smooth muscle mass by antibodies against SMA, and amount of mucus by antibodies against MUC5AC in the lung section. The Picrosirius red staining, immunohistochemistry of SMA and MUC5AC was made on the lung section from guinea pigs sensibilized by saline (OVA -), ovalbumin (OVA +), and sensitized by ovalbumin and on the 15th day of sensitization the administration of 10 mg/kg GSK429286 A (Rho-inh. 10) per os during 28 days of experiment. The amount of collagen in bronchioles was measured semi-quantitatively by using ZEN software. The signal from collagen from negative control (OVA-) serves as a 100% and all groups are compared to negative control (A). The amount of smooth muscle mass was measured by wall thickness in the bronchioles using Quick Photo Micro program (B), and the Image J program was used to determine mucus in the bronchioles (C). Data are shown as average ± SEM; **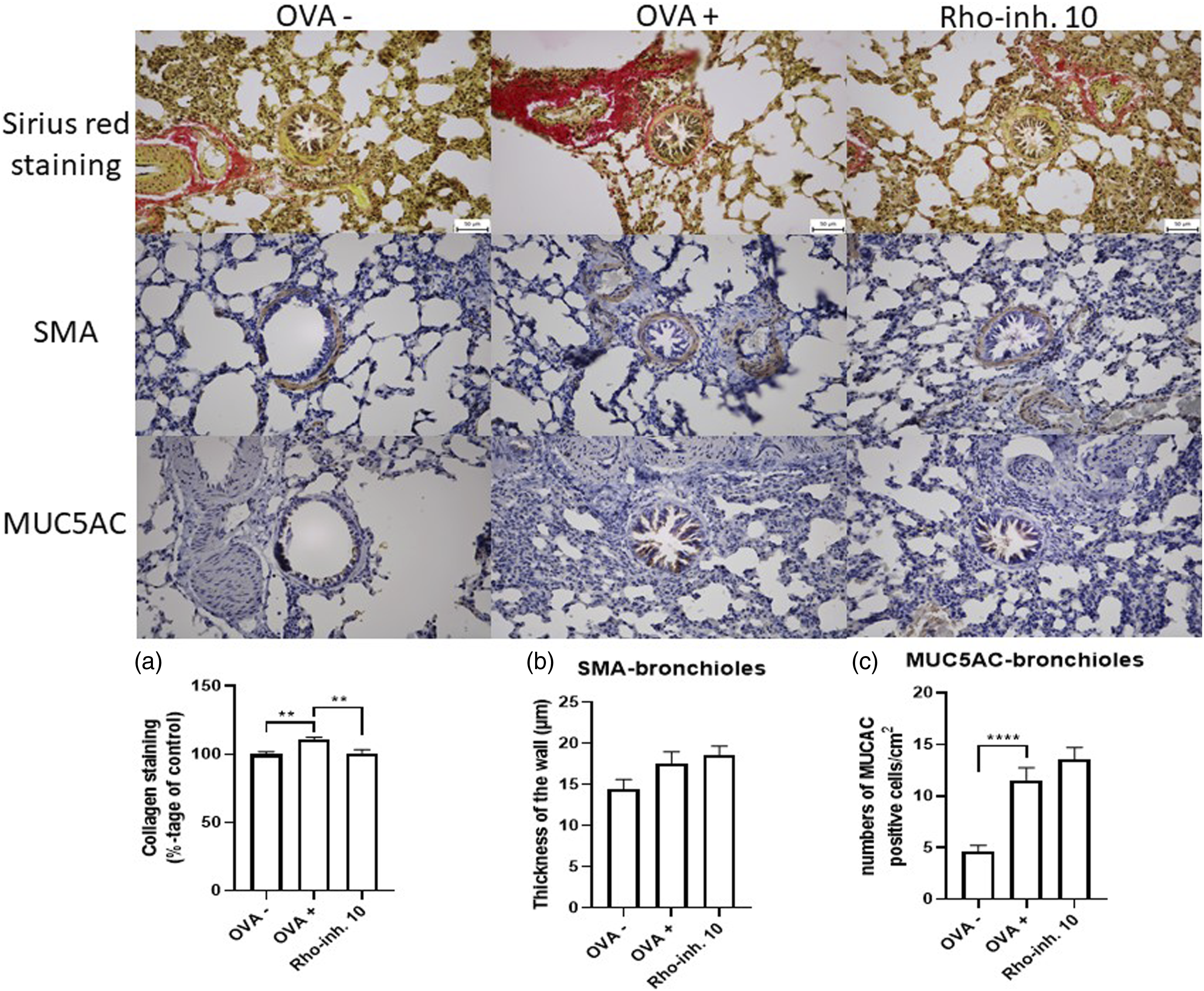
Determination of remodeling factors (collagen III and V, TGF-β, and SMA) in lung homogenates
To investigate the anti-remodeling effect of GSK429286 A we measured collagen type III and type V, TGF-β1, and SMA in the homogenates from the lungs tissue. Our results confirmed the activated remodeling processes in the lungs in the animals sensitized with ovalbumin for 28 days, which was demonstrated by increased levels of collagen type III (Figure 7(a), OVA +), collagen type V (Figure 7(b), OVA +), TGF-β1 (Figure 7(c), OVA +), and SMA (Figure 7(d), OVA +). Significant reduction of remodeling proteins: collagen type III and type V, and TGF-β1 was demonstrated in 28 days OVA-sensitized animals with the administration of GSK429286 A in a dose of 1 mg/kg (Rho-inh. 1) and 10 mg/kg (Rho-inh. 10) compared with OVA + group. On the other hand, significant changes of levels in SMA were not proven probably due to short-term sensibilization by OVA. The changes of levels of collagen III (COL 3, A), collagen V (COL 5, B), transforming growth factor beta 1 (TGF-β1, C), and smooth muscle actin (SMA, D) in the lung homogenates. The concentration of COL one and 3, TGF-β1, and SMA proteins was measured by ELISA method and subsequently quantified for the appropriate amount of proteins. OVA – group represents unsensitized guinea pigs treated with saline for 28 days and OVA + with ovalbumin. Administration of inhibitor of Rho-kinase, GSK429286 A, in concentration of 1 mg/kg (Rho-inh. 1) and 10 mg/kg (Rho-inh. 10) per os was carried out on the 15th day of sensitization. Data are shown as average ± SEM; *
Discussion
Many studies have posited that inhibitors of Rho-kinase have possible therapeutic potential in the reduction of airway hyperresponsiveness, anti-inflammatory and anti-remodeling activity in inflammatory conditions, such as allergic asthma.5,10,17 Indeed, there is evidence, that Rho-kinase pathway is associated with the pathophysiology of asthma. 5 Our study focused on the effects of the new Rho inhibitor GSK429286 A in an experimental model of allergic asthma in guinea pigs. The obtained results showed that GSK429286 A has anti-inflammatory effect (decreased levels of IL-2, IL-4, and IL-5); an effect on airway remodeling (reduction in TGF-β1, collagen type III and V); and an effect on airway hyperreactivity (reduction of sRaw after inhalation of bronchoconstrictor histamine, and the reduction of number of coughs after citric acid inhalation).
Allergic asthma is associated with hypersensitivity and hyperreactivity in the airways after bronchoconstriction stimuli. This is caused by multiple factors, such as penetration of inflammatory cells such as eosinophils into airways, 28 and with higher levels of inflammatory cytokines, growth factors, or chemokines in allergic inflammation. 29 We proved that administration of 10 mg/kg of GSK429286 A reduces the hyperreactivity of the airways to histamine after 14-days of therapy. This effect could be caused separately or through the linked effects of the following mechanisms: anti-inflammatory effect (decreased level of IL-2, 4, 5); reduction of growth factors and transcription factors (NF-κB); and by the possible effect of Rho-kinase inhibitor on smooth muscle. Previous studies have shown that inhibition of Rho-kinase activity could have an impact on smooth muscle relaxation in two ways. Firstly, through a calcium-dependent pathway by inhibition of phosphorylation of CPI-17, and subsequently inhibiting myosin light chain (MLC) phosphorylation leading to smooth muscle relaxation. 30 Secondly, through a calcium-independent pathway, which is mediated by molecular mechanisms leading to dephosphorylation of MLC and subsequent relaxation. 31 In addition, our previous study showed a similar effect on airway reactivity by using another inhibitor of Rho: hydroxyfasudil. 20
Allergic asthma is associated with higher infiltration, cumulation and synthesis of immune cells, such as T-helper (Th) cells, eosinophils, neutrophils, and mast cells in the airways. This immune response leads to a higher synthesis of specific cytokines, such as IL-4, 5, 9, 13, GM-CSF, IFN-γ, and TNF-α, which is followed by releasing these cytokines to the environment of inflammation. 32 However, at the beginning of the immune response, the allergen activated immune cells and also the transcriptions factors, specifically NF-κB. Activated NF-κB moves from cytosol to the cell nucleus, where it plays a crucial role in the transcription of genes involved in inflammation, such as cytokines. 33 One of these cytokines is also IL-2, whose function is regulating the growth, differentiation, and activity of Th-cells. 34 There is also a shred evidence, that inhibition of IL-2 could influence differentiation and proliferation of Th-2 cells, which may lead to decreased synthesis of cytokines like IL-4, 5, 9, 13 and GM-CSF. 35 In addition, this long-lasting increased inflammation in airways may lead to remodeling. 36
In this research, we used the ovalbumin-induced model of allergic asthma in guinea pigs, which is related to eosinophil type of asthma, also called Th-2 type of asthma. 37 We demonstrated, that 14 days of oral application of 10 mg/kg GSK429286 A have a significant anti-inflammatory effect in experimental allergic airway inflammation. This has been proven by the decreased levels of transcriptional factor NF-κB, IL-2, and Th2-cytokines, such as IL-4 and IL-5, in the lung tissue. We did not observe this effect on decreased levels of cytokines associated with Th-1 cells.
IL-4 and IL-13 are closely related cytokines, that mediate their effect by stimulation of complex receptor systems consisting of type I and type II receptors. While type I receptors, found on hematopoietic cells, can only bind IL-4, type II receptors, present on structural cells of the airways, respond to both cytokines. 38 We assume that even though the level of IL-13 remains in our research unaltered, its effects can be compensated by the excessive production of IL-4, which leads to the dual stimulation of both type I and type II receptors. Furthermore, it was reported that elevated IL-13 levels were detected only in 50 % of asthmatic patients, regardless of disease severity. 39 Experimental animal studies demonstrated that neutralization of IL-13 reduced airway hyperreactivity and mucus production. 40 However selective anti-IL-13 antibody, tralokinumab, did not change the rate of asthmatic exacerbation in a double-blind, placebo-controlled study. 41 On the other hand, Dupilumab, an antibody targeting common subunit (IL-4Rα) of both type I and type II receptors, reduced asthma exacerbations and improved respiratory symptoms in asthmatic patients. 42
Asthma-associated airway remodeling is manifested by changes in composition, organization, and content of the cellular constituents and molecular mechanisms in the airway wall, such as goblet cell metaplasia, subepithelial fibrosis, increased ASM mass, angiogenesis and others. 43 It is assumed, that subepithelial fibrosis is mainly caused by TGF-β1, which concentration is higher in patients with asthma. TGF-β is mainly produced by epithelial cells, macrophages, fibroblasts, and in the case of asthmatic airways by eosinophils. 44 Indeed, subepithelial fibrosis was caused by the intra-tracheal addition of TGF-β1 in the murine model of asthma. 45 Airway remodeling and fibrosis development are stimulated by TGF-β1 by boosting the synthesis of extracellular matrix proteins (ECM), such as collagen type I, III, and V, and by increasing the proliferation of ASM. 46 Furthermore, the application of antibodies against TGF-β1 in the murine model of asthma reduces ASM proliferation, levels of ECM proteins, and production of mucus. 47
Recent experimental studies have shown that the Rho-kinase pathway is involved in pulmonary remodeling and inhibition of both isoforms of ROCK, namely ROCK1 and ROCK2, suppressed TGF-β-induced remodeling in lungs. 48 Our results confirmed these findings in the allergen-sensitized animals with the administration of both concentrations of GSK429286 A, inhibitor of ROCK, for 14 days by reduction of the level of TGF-β1 in the lung homogenates. Similar results were obtained in our previous study by the administration of hydroxyfasudil, inhibitor of ROCK. 20
We mentioned, that TGF-β1 is connected with the synthesis of ECM proteins, such as collagen type I, III, and V, in asthmatic airways. The function of collagen type I and III in healthy people is to maintain the structural framework in the interstitium, bronchi, and alveolar wall. 49 On the other hand, increased deposition of ECM proteins in the airways has been described in asthmatic patients, which could affect the contractile properties of ASM cells and may contribute to remodeling changes. 50 Furthermore, collagen type V is a less abundant collagen of ECM proteins in airways, but its increased levels are associated with asthma. 51 In our experimental model of allergic asthma in guinea pigs, we found increased levels of collagen type III, and also V in the homogenates from lungs of OVA-sensitized animals and their decrease was observed after 14 days of administration of both concentrations of GSK429286 A. These results are in concordance with the results from, 48 who found that inhibition of both ROCK isoforms prevented TGF-β1 stimulation of collagen production.
The airway defense mechanisms, such as cough reflex and mucociliary clearance, are increased in patients with asthma, and may be stimulated by higher levels of inflammatory mediators, airway remodeling, and other mechanisms. 52 These factors lead to neuronal changes, known as the neuronal plasticity, in airways. There is evidence, that exposure to ovalbumin could increase the neuronal response in guinea pigs. 53 In our experiment, the cough reflex in guinea pigs was induced by inhalation of an aerosol of citric acid, which is a chemical irritant of the C-fibers in afferent neurons in airways. The higher cough response was measured in OVA-sensitized animals, and a decline to healthy control values was recorded after 14 days of administration of higher concentration GSK429286 A. This effect of GSK429286 A could be caused by an indirect anti-inflammatory effect or by possible partial renovation of neurons induced by Rho-kinase inhibition. 54
The decisive defense of the airways is not only the cough response, but also the mucociliary clearance. Mucociliary clearance acts in addition to the cough reflex to remove foreign particles, pathogens, toxins and viscous mucus from the airways. Its efficacy is determined by ciliary beating and the physical properties of mucus and is particularly important in conditions of cough reflex suppression. Substances that have both antitussive and cilia-inhibiting effects are prone to airway obstruction, with the risk of airflow limitation, local atelectasis, decline in lung function and secondary bacterial infections. This was the main reason why we analyzed the dominant parameter of ciliary beating, CBF, despite the fact that ROCK1 and ROCK2 are only involved in the process of ciliogenesis, whereas other kinases play a role in ciliary function. 55 This correlates well with the results of our study, where neither dose of Rho-kinase inhibitor had a modulatory effect on CBF.
In summary, although this study provides valuable insights into the Rho-kinase inhibitor, GSK429286 A, as a potential therapeutic agent for allergic asthma, there are still some limitations. First of all, our chosen animal model of allergic asthma does not fully reflect the clinical features of allergic asthma in humans. Secondly, further elucidation of the underlying molecular mechanisms, efficacy, and safety of the drug we have studied is needed. Indeed, the inclusion of a broader range of doses, extension of study duration, and consideration of combination therapy will increase the comprehensiveness and clinical translational relevance of the findings. Addressing these limitations in future research will be essential before Rho-kinase inhibitors can be introduced into practice.
Conclusions
Our results confirmed, the promising potential of Rho-kinase inhibitor, GSK429286 A, as a therapeutic agent for allergic airway inflammation. The administration of 10 mg/kg of GSK429286 A for the final 2 weeks in guinea pigs sensitized with ovalbumine, showed positive results across all key parameters associated with asthma, such as decreased bronchoconstriction, decreased inflammation markers, and decreased markers of remodelation. These results revealed a promising effect of Rho-kinase inhibitor, GSK429286 A, in an induced experimental model of allergic asthma. Nonetheless, further research, including proving the effect of other substances, that are considered as an inhibitor of Rho-kinase, and also clinical studies in human populations are needed.
Supplemental Material
Supplemental Material - Anti-remodelling effect of non-selective ROCK inhibitor, GSK429286A, in experimentally induced allergic airway inflammation
Supplemental Material for Anti-remodelling effect of non-selective ROCK inhibitor, GSK429286A, in experimentally induced allergic airway inflammation by Eduard Gondáš, Jozef Mažerik, Matúš Dohál, Soňa Bálentová, Michal Pokusa, Daniela Vargová, Lukáš Smieško, Martina Šutovská, Marta Jošková and Soňa Fraňová in International Journal of Immunopathology and Pharmacology
Footnotes
Acknowledgments
The authors are very grateful to MRs Katarína Jesenská, Margéta Kondeková, and Agáta Rešetárová for their technical support in this project.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Slovak Research and Development Agency [Grant No. APVV-19-0033, and APVV-23-0261] and the Slovak Scientific Grant Agency [Grant VEGA 1/0314/21 and VEGA 1/0042/24].
Ethical statement
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
Data availability statement
The data supporting this study’s findings are available on request from the corresponding author.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
