Abstract
Metaplastic breast cancer (MBC) is a rare and heterogeneous malignancy characterized by the presence of both epithelial and mesenchymal elements. Among its variants, MBC with osteoclastic giant cells is extremely uncommon and presents diagnostic challenges because of its diverse histopathological and imaging features. We report the imaging features of a patient with pathologically proven MBC with osteoclastic giant cells as follows: a high-density mass with microcalcification was revealed on mammography; and a microlobulated, mixed cystic and solid mass with posterior acoustic enhancement was revealed on ultrasound. MRI revealed a round mass with circumscribed margins, rim enhancement and high T1-weighted signal intensity due to internal haemorrhage, heterogeneous T2-weighted high-signal intensity and initial fast enhancement and rapid washout kinetics that was classified as BI-RADS 5. Left mastectomy was performed, and the final diagnosis was consistent with that of MBC.
Introduction
Metaplastic breast cancer (MBC) is a rare and aggressive form of breast cancer that represents less than 1% of all invasive breast carcinomas. 1 According to the WHO classification, MBC can be subclassified into several types on the basis of predominant histologic differentiation, including low-grade adenosquamous, squamous cell, spindle cell, and mixed metaplastic carcinomas and metaplastic carcinomas with mesenchymal differentiation, among others. 2 Variants of MBC containing osteoblastic giant cells (OGCs) are extremely rare and represent an estimated proportion of 0.003–0.12% of all breast cancers. 3 These multinucleated giant cells are typically reactive histiocytic cells and are not malignant, but their presence contributes to the morphological complexity and diagnostic difficulty. Moreover, tumours of this variant often display high-grade features and are frequently negative for hormone receptors and HER2 expression, aligning them with the triple-negative breast cancer (TNBC) molecular subtype, which is associated with poor prognosis and limited treatment options. 4 Importantly, this rare but invasive disease exhibits diverse imaging characteristics. 5 To our knowledge, the radiological findings and pathological features of MBC with OGCs have been described in only a few reports.3,6,7 Understanding its clinical and imaging manifestations, especially MRI manifestations, can help breast radiologists make early diagnoses and provide timely treatment. Here, we report a rare case of MBC with OGCs to highlight its imaging features and elaborate in detail the diagnostic challenges that such cases may pose to physicians.
Case report
A 51-year-old woman presented with a progressively enlarging mass in her left breast that she had first incidentally 6 months prior. The mass was initially small and occasionally painful and was treated empirically with oral antibiotics for presumed inflammation. However, over time the lesion gradually increased in size, prompting further evaluation. This case report was approved by WeiFang Traditional Chinese Hospital (Approval No.: 2025YX291).
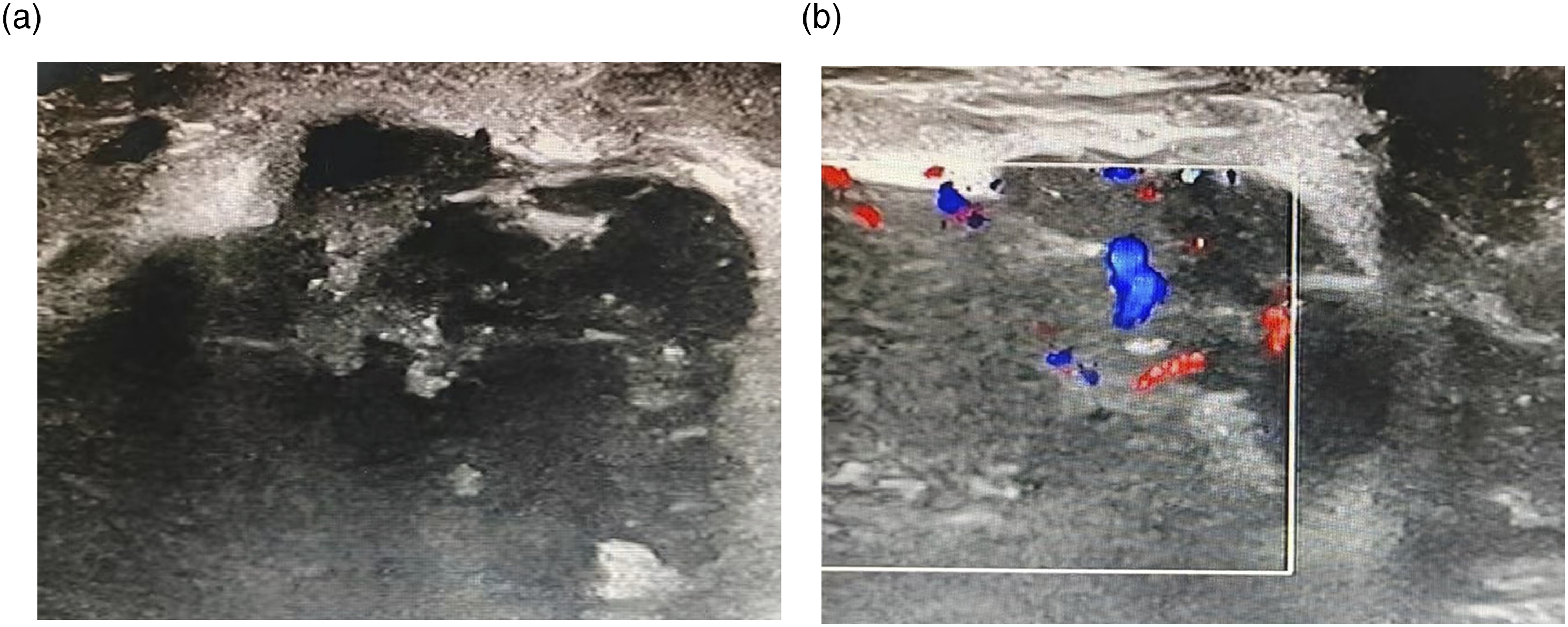
Mammography revealed a round, well-defined high-density mass located in the upper inner quadrant of the left breast, accompanied by amorphous microcalcifications (Figure 1). Ultrasonography revealed a round, mixed, cystic-solid mass measuring approximately 8 cm × 8 cm, with microlobulated margins. Colour Doppler imaging demonstrated peripheral punctate vascular flow (Figure 2). MRI revealed a round cystic-solid lesion at the 10 o’clock position in the left breast. On T1-weighted images, the lesion exhibited partial high-signal intensity, suggesting intralesional haemorrhage. On T2-weighted images, the majority of the lesions showed high-signal intensity. The solid component was eccentrically distributed along the periphery of the mass and appeared round in shape. The solid part of the lesion showed an elevated signal with a low apparent diffusion coefficient (0.987 × 10-3 mm2/s) on diffusion-weighted imaging (DWI). Dynamic contrast-enhanced MRI (DCE-MRI) revealed rim enhancement with initial rapid enhancement and rapid washout kinetics, which was categorized as Breast Imaging Reporting and Data System category 5 (BI-RADS-5) (Figure 3). Mammograms of the left breast in MLO (a) and CC (b) view showing a high-density mass with microcalcification in the inner upper quadrant, with a clear margin. Ultrasound of the left breast showing a large heterogenous lesion with cystic areas within. Doppler colour shows the internal blood vessels (a) and (b). Magnetic resonance imaging of lesions of the left breast (arrows). (a) Lesions with haemorrhage intensity on T1-weighted images. (b) High-signal intensity on T2-weighted images. (c) and (d) On DWI, the mass shows diffusion restriction. (e) and (f) Dynamic contrast-enhanced imaging with time signal intensity curves for washout pattern.

The lesion was categorized as highly suspicious for malignancy, and a core biopsy was performed. Histopathology revealed atypical hyperplasia of the epithelium and a cystic structure of the stratified epithelium, and numerous multinucleated osteoclast-like giant cells could be seen within the cyst. Immunohistochemical (IHC) analysis revealed its expression status as calponin (+), CK (+), CK5/6 (+), CK7 (+), ER (−), Ki67 (30%), P63 (+) and vimentin (+). A left mastectomy was subsequently performed on the patient. Histopathological examination revealed a metaplastic breast cancer with osteoclastic giant cells. Macroscopically, the tumour was 7.5 cm × 6.5 cm × 5.5 cm in size. Areas of necrosis were identified within the tumour. Under a microscope, the tumour was observed to be composed of spindle-shaped tumour cells. Locally, a large number of osteoclast-like multinucleated giant cells could be seen, but the nuclei of the multinucleated giant cells did not have atypia. Breast ductal epithelial hyperplasia and ductal carcinoma in situ (DCIS) could be observed in the peripheral part of the tumour (Figure 4). The patient subsequently received adjuvant chemotherapy. She was free of recurrence during 6 months of follow-up. Microscopic findings showing pleomorphic epithelioid, and spindle-cells admixed with numerous osteoclastic-like giant cell (a). Spindle cells are adjacent to focal necrotic ductal carcinoma in situ (b) (arrows).
Discussion
Breast cancer is most commonly found in the glandular epithelium. However, when the glandular epithelium differentiates into nonglandular mesenchymal tissue, the tumour may undergo biochemical changes. MBC accounts for less than 5% of all breast cancer cases. 8 In 1990, Wargotz and Norris first defined MBC with osteoclastic giant cells (OGCs) as a distinct breast cancer subtype on the basis of 29 cases. Histologically, it features a sarcoma or spindle cell background, mixed with invasive ductal or intraductal carcinoma, and a cell matrix rich in OGCs. 9 MBC can present with different histological and morphological characteristics. MBC with OGCs, presented in this case, is a rare type.
Although the mass in this patient was round and had a clear boundary, it is important to note that microcalcification was revealed by the initial mammography. Twadell et al. described an 82-year-old female patient whose mammogram showed an irregular high-density mass with pleomorphic calcifications, and ultrasound revealed a hypoechoic mass with increased tissue stiffness. 3 Chhieng et al. specifically studied 32 cases of metaplastic carcinomas containing heterologous osteochondral components and reported that calcification and necrotic areas were very common. 10 While it is not typical for MBC tumours to present as calcified masses, this may reflect the degree of tumour ossification, as pathology later revealed the production of a bone-like matrix in this type of tumour. Because osteoid formation can be considered as a calcified lesion on radiological images, it is still very important to consider MBC in the differential diagnosis.
The presence of necrosis and cystic changes, as observed in our case, is particularly characteristic of MBC. The histopathological factors associated with high intensity on T2-weighted images include a high proportion of cells rich in cytoplasm, haemorrhage, necrosis, cystic degeneration, mucous matrix and intratumoral haemorrhagic changes. 11 Tokuda et al. specifically studied spindle cell carcinoma of the breast (a subtype of MBC) and reported that 70% of cases demonstrated high-signal intensity on T2-weighted images, which correlates pathologically with coagulative necrosis and cystic degeneration. 12 Kim et al. described a case of MBC with chondroid differentiation that exhibited heterogeneous high-signal intensity on T2-weighted images, corresponding to a chondromyxoid matrix and cystic components pathologically. 6 Since MBC is a highly malignant tumour, its rapid growth can easily exceed the blood supply capacity, thereby causing extensive internal necrosis within the tumour. In this case, the lesion was partially cystic and showed high-signal intensity on T1-weighted images, suggesting the presence of haemorrhage within the lesion. Upon postoperative histopathological analysis, haemorrhage was identified within the mass. These findings are in line with those of a case report of MBC with chondroid differentiation, which also highlighted eccentric solid components, rim enhancement and haemorrhagic signals on T1-weighted images. 6 Osteoclast-like giant cells usually appear in areas with a stronger inflammatory response, and inflammation itself may lead to increased vascular permeability or vascular damage, thereby increasing the possibility of haemorrhage, which might be the reason for the tumour haemorrhage.
Differential diagnosis is necessary to differentiate MBC from several malignant and benign breast lesions. (1) Invasive ductal carcinomas (IDCs): The characteristic imaging features of IDCs include an irregular shape and spiny margin, pleomorphic calcification and a posterior acoustic shadow. These characteristics are not common in MBC. 13 Compared with IDC, MBC has a larger tumour size, less lymph node involvement and a higher tumour grade and is negative for hormone receptors expression. 1 (2) Fibroadenomas: Fibroadenomas are a benign fibroepithelial tumours composed of mesenchymal and epithelial components. On mammography, fibroadenomas present as well-defined round or oval masses, often with popcorn-shaped calcification features. 14 The lesion shows a high-signal intensity on T2-weighted images. The enhancement is mostly uniform, and haemorrhage is uncommon. 15 (3) Phyllodes tumours: On mammography, a phyllodes tumour is a large, well-circumscribed isodense mass with rare calcification. 14 The rapidly growing phyllodes can also show high-signal intensity on T2-weighted images, accompanied by internal cystic spaces and haemorrhage. 16 (4) Papillomas: Intraductal papillomas are usually located centrally (within 3 cm of the nipple). On MRI, they present as solid or mixed cystic/solid masses with various kinetic enhancement patterns, but they can also present as linear nonglandular enhancement. Associated duct dilatation is often visible. 17
In conclusion, MBC with OGCs is a rare subtype of breast malignancy with nonspecific imaging features. Its characteristics include rapid growth, a low axillary lymph nodes involvement rate and high local and distant recurrence rates. Owing to its poor response to systemic treatment, the prognosis of most MBC is poor. On mammography and ultrasound, MBC is characterized by circumscribed margins, round shape and posterior acoustic enhancement. T2-weighted high-signal intensity combined with haemorrhage, early enhancement and a rapid washout kinetics in a peripheral rim and nonenhancing internal components on MRI are helpful in differentiating it from other breast malignancies. Therefore, multimodal imaging studies combining immunohistochemical and pathological studies are necessary for the diagnosis of MBC. Further studies may be needed to develop targeted treatment options to improve clinical outcomes.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: yu hai tong; ZR202103060229.
