Abstract
Background
The goals of neoadjuvant systemic therapy (NST) are to reduce tumor volume and to provide a prognostic indicator in assessing treatment response. Digital breast tomosynthesis (DBT) was developed and has increased interest in clinical settings due to its higher sensitivity for breast cancer detection compared to full-field digital mammography (FFDM).
Purpose
To evaluate the accuracy of DBT in assessing response to NST compared to FFDM, ultrasound (US), and magnetic resonance imaging (MRI) in breast cancer patients.
Material and Methods
In this retrospective study, 95 stages II–III breast cancer patients undergoing NST and subsequent surgeries were enrolled. After NST, the longest diameter of residual tumor measured by DBT, FFDM, US, and MRI was compared with pathology. Agreements and correlations of tumor size were assessed, and the diagnostic performance for predicting pathologic complete response (pCR) was evaluated.
Results
Mean residual tumor size after NST was 19.9 mm for DBT, 18.7 mm for FFDM, 16.0 mm for US, and 18.4 mm for MRI, compared with 17.9 mm on pathology. DBT and MRI correlated better with pathology than that of FFDM and US. The ICC values were 0.85, 0.87, 0.74, and 0.77, respectively. Twenty-five patients (26.3%) achieved pCR after NST. For predicting pCR, area under the receiver operating characteristic (ROC) curve for DBT, FFDM, US, and MRI were 0.79, 0.66, 0.68, and 0.77, respectively.
Conclusion
DBT has good correlation with histopathology for measuring residual tumor size after NST. DBT was comparable to MRI in assessing tumor response after completion of NST.
Keywords
Introduction
Neoadjuvant systemic therapy (NST) has evolved as a well-established treatment for locally advanced breast cancers and increasingly being used for early breast cancer.1,2 It may decrease the extent of the tumor, thus increasing the chances of successful breast-conservation surgery (BCS) and provide prognostic information to evaluate treatment response. 3 NST has proved to be equivalent to postoperative chemotherapy in terms of disease-free and overall survival, and pathological complete response (pCR) has been increasingly observed after NST for invasive breast cancer.4–8 Identification of pCR at the end of NST prior to surgery may influence surgical decision-making regarding both the breast and axilla; therefore, confident identification of pCR may lead to less radical breast surgery and sentinel node biopsy rather than axillary clearance in women with pre-treatment positive nodes. 9
In patients receiving NST, the residual tumor size is assessed prior to surgical treatment to determine the efficacy of the treatment, whether the patient is eligible for surgery, and the appropriate surgical technique to be applied. 10 The assessment of the response to NST usually relies on a combination of clinical examination and imaging. For image evaluation of the extent of the residual tumor after NST, full-field digital mammography (FFDM), breast ultrasound (US), and magnetic resonance imaging (MRI) are commonly used. To date, contrast-enhanced breast MRI has been considered the most effective imaging modality for assessing the tumor response to NST because the enhancement patterns can detect tumor angiogenesis, the accompanying changes in tumor microcirculation, and increased permeability of the newly formed vessels.11–17 Despite the high accuracy of MR imaging, false positive and false negative results still occur.10,14–16 Underestimation of residual tumor size may lead to involved surgical margins and repeat surgery, whereas overestimation may lead to overly radical surgery (including mastectomy when BCS may have been possible), and poorer cosmetic, and psychosocial outcomes. 18 Moreover, although mammographically identified residual microcalcifications do not always correlate with residual tumor burden, MR imaging has a limited ability to accurately evaluate the extent of malignant microcalcifications that require complete excision. 19
Recently, digital breast tomosynthesis (DBT) was developed and has sparked increased interest in clinical settings due to its higher sensitivity for breast cancer detection compared to other imaging methods.20–23 One of the main issues with FFDM is that it causes obscuring of tumor outline due to overlying surrounding breast tissue; DBT overcomes this major problem. 24 However, few studies have assessed the diagnostic performance of DBT in preoperative staging after NST in comparison to traditional imaging methods such as FFDM, US, or MRI.24,25
The purpose of this study, therefore, was to evaluate the accuracy of DBT in assessing residual tumor and predicting pathologic complete response (pCR) compared to FFDM, MRI, and US after completion of NST in breast cancer patients.
Materials and methods
Study design and patient selection
A review of hospital records was conducted to identify patients who had biopsy-confirmed, locally advanced (stage II or III), invasive breast cancers who underwent NST and surgical treatment at our hospital between January 2017 and March 2019. Patients who had undergone preoperative imaging evaluation by breast DBT, FFDM, US, and MRI at the completion of NST were enrolled in the present study. DBT was routinely used for post-NST evaluation at our hospital. This retrospective study was approved by our institutional ethics committee and was conducted in accordance with the Declaration of Helsinki (2000). Informed consent was waived because of the retrospective nature of the study.
Imaging techniques
DBT and FFDM images were sequentially acquired in one session with single breast positioning and compression per view, while mediolateral oblique and cranio-caudal views were acquired as 25 projections over an angle of 50° (Mammomat Inspiration, Siemens, Erlangen, Germany). An anode/filter combination W/Rh was used at a tube voltage identical to that used for FFDM, along with automated exposure control and iterative reconstruction technique. The DBT slice images were reconstructed as 1 mm slice images and a high in-plane resolution of 0.085 mm × 0.085 mm.
US was performed using a 12 MHz probe and the Noblus ultrasound scanner (Hitachi, Ltd, Japan). The US images were obtained in both the sagittal and transverse planes. Three measurements were obtained from the tumor in the sagittal, transverse, and anteroposterior planes.
MRI was performed with a 3.0-T scanner (Achieva Philips Healthcare Best, Netherlands) using a dedicated breast array coil. The dynamic breast examination was performed before and after intravenous contrast material injection (Magnevist, Bayer Yakuhin, Japan) through the antecubital vein with a dose of 0.1 mmol/kg followed by flush-out with 20 ml of saline solution using a power injector. Axial T1-weighted non-contrast MR images and the following three post-contrast dynamic sequences (first contrasted acquisition was performed 20 seconds after injection and the last acquisition was performed 6 minutes after injection) were obtained. Post-processing manipulation included the production of standard subtraction, reverse subtraction and maximum intensity projection and multiplanar reconstruction images.
Imaging interpretation
Images were evaluated by two board-certified radiologists (R.M. and H.T., with 15 years and 8 years of experience, respectively) in consensus. A dedicated workstation (WE View, Hitachi, Ltd, Japan) was used for interpretation of MRI and US studies. DBT and FFDM were interpreted on a digital mammography workstation (Plissimo MG, Panasonic, Japan) equipped with a set of 5-MP monochrome LCD monitors (MFGD5621HD, 2048 × 2560 pixels, 21.3-inch display; BARCO, Torhout, Belgium). The presence of any abnormality that might indicate potential residual cancer was assessed and the longest diameter of the lesion was measured.
Histopathological evaluation
Patients underwent BCS or mastectomy and surgery of the ipsilateral axilla (sentinel lymph node biopsy in the case of node-negative and axillary lymph node dissection in the case of N+) after completion of NST. Surgical specimens were evaluated by dedicated breast pathologists. Residual tumor size was assessed by gross and microscopic examination of the excised tumor specimens. In the case of multifocal breast cancer, the sum of the diameters for all target lesions was calculated. Tumors were cut sagittal into parallel slices and the slice with the cut surface exhibiting the longest dimension was used to measure the longest and perpendicular diameters of the tumor, considered to be the gold standard. Tumor regression grade was classified into four categories: Grade 0 (No Response) to Grade 3 (Complete Response) according to the Miller-Payne criteria. 26 A pathologic complete response (pCR) was defined as complete disappearance of invasive cancer in the breast irrespective of ductal carcinoma in situ or nodal involvement.
Histologic type, histologic grade, estrogen receptor (ER), progesterone receptor (PR), and the human epidermal growth factor receptor 2 (HER2) expression status were determined from histopathologic reports of core biopsies performed before NST. Tumors were classified into the following subtypes: Luminal, ER or PR positive; HER2-enriched, ER and PR negative and HER2 positive; or triple-negative type, that is, ER, PR, and HER2 negative.
Statistical analysis
Descriptive statistics were given as median (range) and mean ± SD. Categorical variables were stated as frequencies and percentages. Intraclass correlation coefficient (ICC) analyses were used to evaluate the agreement and discrepancies between the longest dimension measured by DBT, FFDM, US, and MRI and the longest dimension measured by pathology. Bland–Altman plots were used to visualize the differences between tumor size measured on DBT, FFDM, US, MRI, and histological size. For predicting pCR, diagnostic performances were calculated and the area under the curve (AUC) values were compared by receiver operating characteristic (ROC) curve analysis.
The Statistical Package for BellCurve for Excel version 3.20 (Social Survey Research Information Co., Ltd.; Tokyo, Japan) was used for statistical analysis. A p value < .05 was considered statistically significant.
Results
Clinical characteristics of study population.
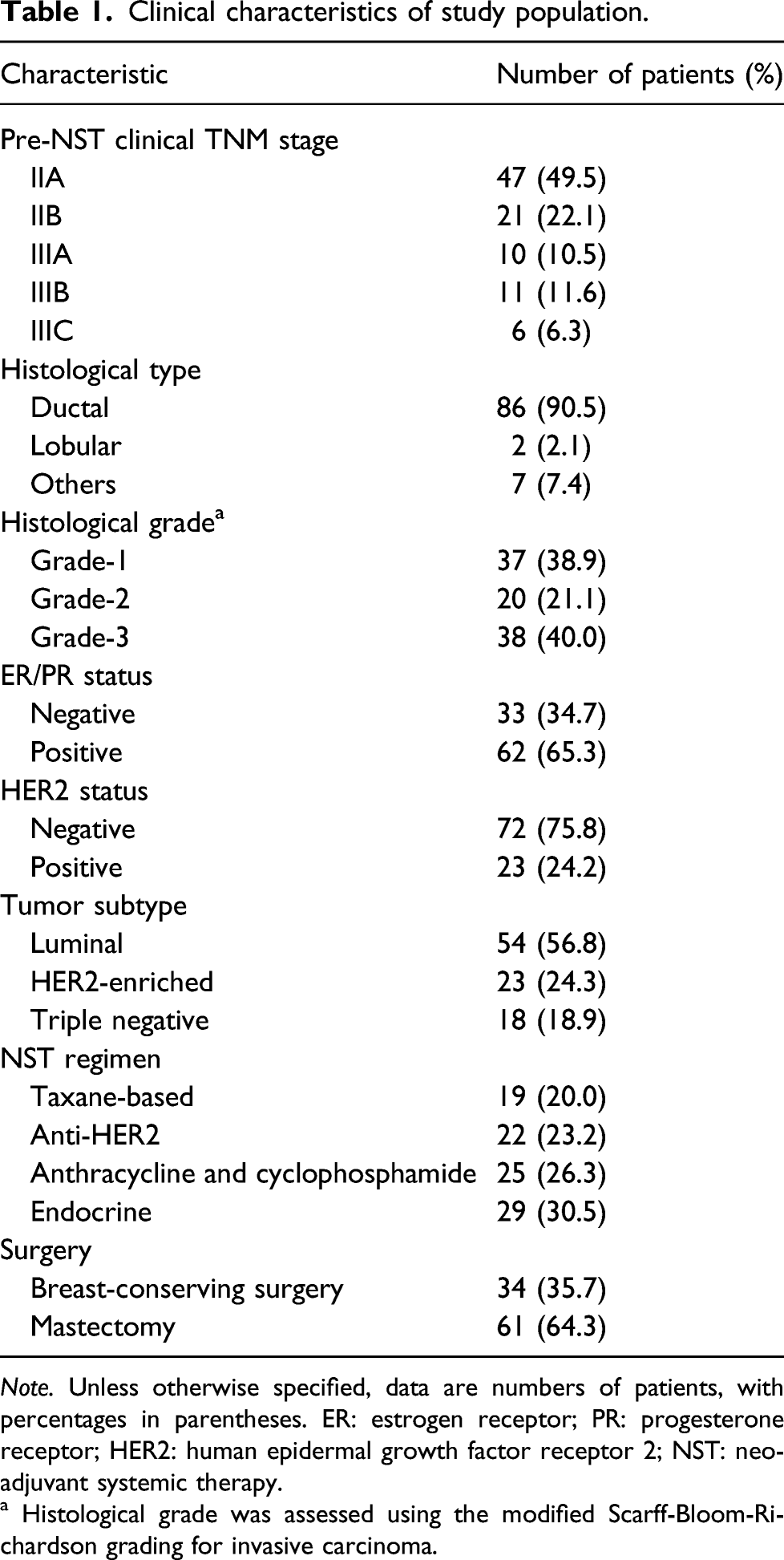
Note. Unless otherwise specified, data are numbers of patients, with percentages in parentheses. ER: estrogen receptor; PR: progesterone receptor; HER2: human epidermal growth factor receptor 2; NST: neoadjuvant systemic therapy.
a Histological grade was assessed using the modified Scarff-Bloom-Richardson grading for invasive carcinoma.
After NST, pathologic mean tumor size of the invasive tumors alone was 14.1 mm ± 15.3, and that of the total tumor including ductal carcinoma in situ (DCIS) component was 17.9 mm ± 17.7 at surgical histopathologic examination. Pathological responses of the lesions to NST were Grade 1 (n = 52), Grade 2 (n = 18), and Grade 3 (n = 25).
Pathologic mean residual tumor size (including the DCIS component) after NST was 17.9 mm (range of 0–114 mm) compared with 19.9 mm (0–115 mm) for DBT, 18.7 mm (0–115 mm) for FFDM, 16.0 mm (0–90 mm) for US, and 18.4 mm (0–90 mm) for MRI, respectively. The ICC values between measurements of each imaging modality and pathologic tumor size were 0.85 for DBT, 0.74 for FFDM, 0.77 for US, and 0.87 for MRI (Figure 1). When analyzed according to subtypes, the ICC values for the luminal subtype were 0.45 for DBT, 0.47 for FFDM, 0.85 for US, and 0.67 for MRI. The ICC values for the HER2-enriched subtype were 0.97 for DBT, 0.61 for FFDM, 0.69 for US, and 0.65 for MRI. The ICC values for the triple-negative subtype were 0.98 for DBT, 0.63 for FFDM, 0.53 for US, and 0.98 for MRI. The agreement of residual DBT abnormality and pathology was highest in HER2 and triple-negative subtype patients and lowest in the luminal subtype patients. The triple-negative subtype showed almost perfect reliability in the prediction of residual tumor size on both DBT and MRI. Scatter diagrams of correlation between residual tumor size by imaging and pathology. (a) Correlation between residual tumor size between digital breast tomosynthesis (DBT) and pathology. (b) Correlation between residual tumor size between full-field digital mammography (FFDM) and pathology. (c) Correlation between residual tumor size between ultrasound (US) and pathology. (d) Correlation between residual tumor size between magnetic resonance imaging (MRI) and pathology.
Discrepancies between DBT, MG, US, MRI size, and pathological size.
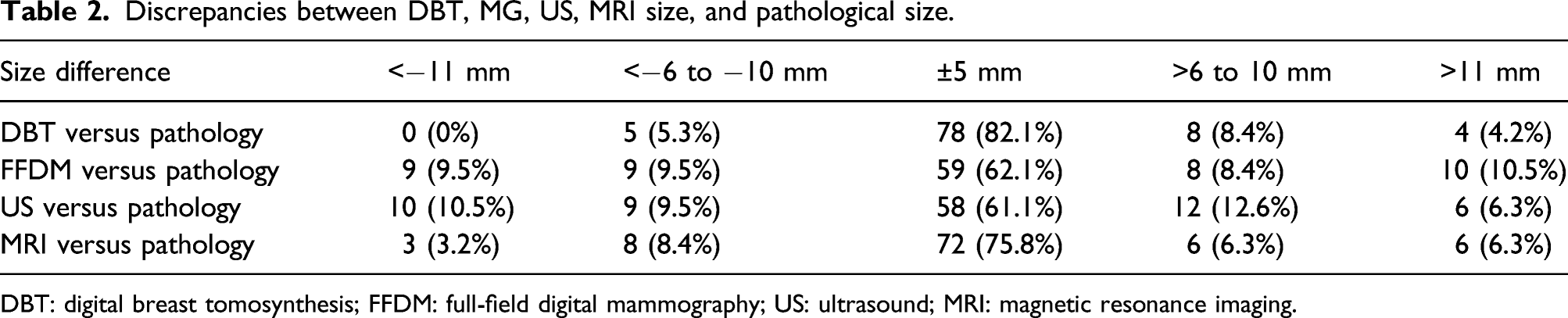
DBT: digital breast tomosynthesis; FFDM: full-field digital mammography; US: ultrasound; MRI: magnetic resonance imaging.

Fifty-year-old female patient with invasive ductal carcinoma (HER2 type) with pathologic complete response. (a) Initial DBT showed 31 mm sized lobulated mass with indistinct margin in left breast subareolar area. After NAC showed no residual mass. (b) Pre- and post-NAC FFDM. (c) pre- and post-NAC MRI showed mass with irregular margin, which was disappeared after NAC.

Seventy-year-old female patient with triple-negative tumor on her left breast. Final pathologic residual tumor size was 5 mm. (a) Initial DBT showed adjacent 22 mm and 15 mm sized irregular mass with indistinct margin left breast. DBT after NAC showed 5 mm mass. (b) Pre- and post-NAC FFDM. (c) Preoperative MRI after NAC showed non-mass enhancement measured as 18 mm.
Twenty-five of 95 (26.3%) patients achieved pCR. The AUC values for predicting pCR on DBT, FFDM, US, and MRI were 0.79 (95% CI, 0.66–0.88), 0.66 (95% CI, 0.54–0.79), 0.68 (95% CI, 0.56–0.80), and 0.77 (95% CI, 0.68–0.89), respectively (p < .05 for FFDM, p = .1352 for US, and p = .8068 for MRI, when DBT was used as the reference) (Figure 4). DBT and MRI have the best results overall. Area under the curves for prediction of pathologic complete response (pCR).
Discussion
In our study, we evaluated the diagnostic performance of DBT for detecting and measuring residual tumor in patients with breast cancer after NST. DBT and MRI were found to be more sensitive than FFDM and US in assessing residual tumor after NST. The correlation coefficient of DBT and MRI according to the pathology was found to be high. Among molecular subtypes of invasive breast cancers, HER2 and triple-negative subgroups showed the highest agreement between DBT and pathology. The triple-negative subgroup showed almost perfect agreement in the prediction of residual tumor extent on both DBT and MRI. Moreover, MRI and DBT outperform FFDM and US in the prediction of pCR.
DBT was recently developed and applied clinically; this method has shown advantages in the evaluation of mass, asymmetry, and architectural distortions. Moreover, the reproducible findings on DBT have made it a more reliable tool than other traditional imaging methods such as FFDM. DBT shows higher detection rate and diagnostic accuracy for both benign and malignant mass-like lesions, with better sensitivity and specificity and lower recall rates.20–23 However, there are few studies on the usefulness of DBT in assessment of breast cancer after NST.24,25 DBT has been reported to improve diagnostic accuracy after NST and the combination of DBT and other imaging modalities was shown to provide more accurate assessment of response to NST. 24 Park et al. reported that DBT and MRI better correlated with pathology than that on mammography and US. 25 These results are consistent with the findings of our study.
The expected post-NST changes on DBT or FFDM can include size of mass, density of mass, and extent of microcalcification. One of the biggest challenges in assessment of DBT after NST might be the extent of microcalcification. Several authors have correlated mammographic microcalcification with surgical specimen and concluded that residual microcalcification could be due to both residual tumor and treated cancer with necrotic material.27–29 Feliciano et al. evaluated whether the loss of MRI enhancement can be a predictor for tumor response after NST regardless of presence of residual microcalcification and concluded that complete excision of tumor bed calcification remained standard practice. 30 In our study, overestimation was observed in residual tumor size measured by DBT included luminal subtype manifest as microcalcification and small spiculated lesion, whereas underestimation was mainly observed by FFDM. This result may be due to the superiority of DBT over FFDM in assessment of tumor morphology including tumor margin and size.
A systematic review showed that MRI performance was superior to US, FFDM, and clinical examination in evaluating residual tumor after NST.31,32 In particular, MRI has the ability to differentiate between non-vascularized therapy-induced fibrosis and residual vital tumor. 33 However, MRI showed a tendency to overestimate pathologic size after NST.13,14 The accuracy of post-NST assessment differs based on tumor subtype. The size of ER/PR positive tumors was less accurately assessed than that of HER2-positive or triple-negative subtypes. 34 The main reason for discordance between MRI and pathological size is DCIS histology. The non-mass-like enhancement of DCIS on MRI may be the reason for the overestimation of the size of the invasive tumor. Moreover, the enhancement of a lesion on MRI depends on its neovascularization. This neovascularization with increased microvessel permeability is the prerequisite for contrast agent pooling in and around malignant lesions. 35
US has always been an attractive supplement to other imaging modalities in breast cancer patients. However, US underestimates tumor extent to a higher degree in our study. It is difficult to differentiate between a fibrotic change induced by neoplastic change and a reduction in the tumor by NST. US can result in over-/under-diagnosis of chemotherapy-induced fibrosis. In addition, it is difficult to measure the overview of a large mass lesion or multi-centric lesion, such as locally advanced tumors that could be treated by NST, by hand-held probe. The inherent pliability of breast tissue also means that tumor dimensions may vary depending on patient positioning: upright (DBT and FFDM), supine (US), and prone positions (MRI). Furthermore, the effects of NST may introduce greater bias in residual tumor measurement relative to the preoperative setting: reactive inflammation, fibrosis, or necrosis may be difficult to distinguish from residual tumor, and measurement errors may be additive when tumors regress as multiple scattered deposits. 36
The practical aspects of the imaging modalities have been shown to have clinical advantages with respect to patient preferences. Claustrophobia and time constraints are the two most common complaints for breast MRI. Patients who undergo DBT often express lower rates of anxiety and higher rates of comfort compared to MRI. Time savings can be substantial because MRI acquisition in most institutions lasts approximately 30–60 min, whereas DBT requires 7–10 min. Additionally, the potential for substantial cost-savings compared with MRI makes DBT an appealing option in a stringent healthcare economy.
Our study has several limitations. First, it was a single-center study and, therefore, also examined a small study population. Second, the study population included only two cases of invasive lobular carcinoma, which is known to be more difficult to evaluate on imaging and is usually less responsive to NST compared with IDC. Further studies with a larger number of patients are needed. Third, DBT, US, and MRI were not performed on the same day, although the interval between the different imaging modalities was not too long. Fourth, as in the clinical setting, tumors were only measured on one slice on DBT and MRI and in one plane by the pathologist. If the cutting plane was different from the DBT and MRI slice direction, the tumor diameter might differ. Finally, patients went through multiple chemotherapy regimens and different numbers and types of NST cycles. Because of the heterogeneous usage of the chemotherapeutic regimens, it was difficult to compare the results regarding the impact of the chemotherapy among the studies.
In conclusion, DBT showed good correlation and agreement with pathology for residual tumors after NST. DBT appeared to perform as well as MRI in assessing the presence of residual tumor. Our findings are encouraging since DBT might be useful for patients with contraindications to MRI and patients in regions with limited MRI availability.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Japan Society for the Promotion of Science; Grant-in-Aid for Scientific Research (JP17K10375).
