Abstract
Background
Management of rectal cancer requires accurate staging and treatment. Neoadjuvant chemoradiotherapy offers tumour size reduction and mitigation of the risk of local relapse. Patients with complete response to neoadjuvant treatment can be enclosed in watchful waiting (WW). Recent studies have explored magnetic resonance imaging (MRI) T1 relaxation time (T1RT) as a predictive biomarker for treatment response in rectal cancer. Preliminary findings indicate that lower T1RT correlates with pathologic complete response. However, inclusion of patients in WW remains unexplored.
Purpose
This prospective study aims to investigate T1RT 6 weeks after neoadjuvant treatment and the ability to determine complete response.
Material and methods
MRI scans are conducted on a 1.5 T MRI-unit. T1RT is measured at time of diagnosis and 6 weeks after neoadjuvant treatment. Experienced radiologists analyse T1RT using specialised software. Treatment decisions are made in multidisciplinary team conferences based on tumour staging. Endpoints include tumour visibility on MRI and endoscopy, along with histopathological analysis of surgical specimens. Statistical methods include t test and receiver operating characteristic curves. Sample size calculations showed we must enrol 76 participants to achieve a statistical power of 80% with an α = 0.05.
Results
Data analysis begins in winter 2025. Results are planned to be submitted in spring 2026.
Conclusion
The implications of this study extend to the potential refinement of treatment strategies, offering patients the prospect of improved outcomes and the potential avoidance of surgery-associated risks. We expect to find a lower relaxation time in fibrotic tissue compared to non-responsive cancerous tissue after 6 weeks.
Keywords
Introduction
Determination of the cancer stage is essential for choosing the treatment of rectal cancer. 1 Neoadjuvant chemoradiotherapy (CRT) has demonstrated efficacy notably in tumour size reduction and mitigating the risk of local relapse.2,3 Some patients may achieve a clinical complete response (cCR) to this neoadjuvant CRT. Patients who achieve cCR may go into watchful waiting (WW), which is a follow-up regimen without surgery. Instead of surgery, this group is followed closely with magnetic resonance imaging (MRI), computed tomography (CT), digital exploration, and endoscopy. Every patient diagnosed with rectal cancer will have an MRI scan to assess the stage of their disease, and this has resulted in improved treatment and survival rate. 4 MRI is the preferred imaging modality for staging and restaging rectal cancer. 5
Apparent diffusion coefficient (ADC) values of pretreatment MRI have been significantly associated with a pathological complete response (pCR). 6 Yet, Schurink et al. reported that ADC has limited predictive value for response to pretreatment CRT 7 ; therefore, more precise tools are still needed.
MRI relaxation time is the time it takes for the protons to return to their equilibrium state after a radiofrequency pulse in the MRI scanner. There are two main types of relaxation time in MRI 8 : T1 (spin-lattice or longitudinal relaxation time) and T2 (spin-spin or transverse relaxation time). Initially, the protons are perturbed by an external radiofrequency pulse, which flips their spins away from alignment. The pulse passes, and the protons regain their original alignment with the magnetic field. 8 Different tissues have different relaxation times. These differences contribute to the contrast seen in MRI images, allowing for the differentiation of various structures and abnormalities.8,9
MRI T1 relaxation time (T1RT) is used diagnostically in multiple organs. T1RT can be used for distinguishing normal myocardium from ischemic disease, 10 for early evaluation of fibrosis following pancreatitis, 11 and diagnostics of breast tumours. 12 T1RT can discern healthy tissue from tumorous and different types of tumours in the rectum,9,13 yet articles regarding T1RT in relation to rectal cancer are scarce. Before CRT became a standard, a study showed a correlation between shorter T1RT in fibrosis compared to local relapse of adenocarcinoma. 14
We conducted a systematic review on the use of MRI T1RT for assessing treatment outcome for rectal cancer patients undergoing neoadjuvant treatment. We searched databases PubMed, Embase, and MEDLINE (Ovid) on September 23, 2024. The search strings consisted of ‘rectum’, ‘neoplasm’, and ‘T1 relaxation time’ (or terms equal to these). Search strings are given in Appendix A.
Two reviewers (MRB and SRR) independently screened and excluded irrelevant studies (Figure 1). Any disagreements during exclusion were resolved by consensus. Inclusion criteria for the full-text review required studies to investigate the use of T1 mapping in rectal cancer patients undergoing neoadjuvant CRT. Exclusion criteria were (1) non-English publications and (2) non-original research articles (including case reports, commentaries, conference abstracts, editorials, letters, and reviews). Flowchart illustrating the process of screening and exclusion during the systematic review.
After exclusion, we had only one study left for inclusion. Lian et al. 15 hypothesised that T1RT values at the primary MRI scan was correlated with response type. They found that, for patients who received neoadjuvant treatment, primary T1RT was significantly lower in tumours who obtained pCR, and in tumours which were downstaged in T-category. These first results are promising, but participants were not included in the watchful waiting (WW) regimen, and they used an MRI scanner from a different vendor. 15
Our aim is to improve diagnostics of rectal cancer including neoadjuvant treatment response evaluation. The objective of this prospective study is to evaluate the diagnostic accuracy of MRI T1RT at 6 weeks after neoadjuvant treatment in detecting fibrosis as an indicator of complete response to neoadjuvant therapy in patients with rectal cancer. This includes participants in WW. Furthermore, we want to investigate if the MRI T1RT is correlated to the tumour volume and ADC. Finally, we will assess reproducibility by investigating the inter- and intraobserver variability.
Material and methods
Study design
This is a prospective, single centre study. All participants undergo standardised MRI scans on a 1.5 T MRI-unit (Koninklijke Philips N.V., Amsterdam, the Netherlands). The T1 maps are processed using IntelliSpace software (Koninklijke Philips N.V., Amsterdam, The Netherlands), which allows quantitative measurement of T1RT [ms].
MRI scans are performed exclusively at University Hospital of Southern Denmark, Vejle, Denmark. Inclusion of participants began in May 2023 and is expected to conclude by the summer of 2025. To reach the desired power for this study, the aim is to enrol 76 participants.
Patient and public involvement
The chairman of the patient relatives council for the hospital has been involved in the preparation of the protocol.
The trial is published on ClinicalTrials.gov under ID NCT05876026. All outcomes, including positive, negative, and inconclusive will be submitted for publication in international peer-reviewed scientific journals.
Participants
Inclusion criteria in this trial are: (1) age older than 18 years, (2) inclusion in the WW regimen in accordance with national guidelines, and (3) rectal cancer undergoing CRT as the initial treatment. This includes patients with a clinical tumour category (cT) of 1–3 and advanced T3 (cd) disease and T4.
Exclusion criteria are contraindications for MRI such as marked claustrophobia, MRI incompatible implants, or previous radiotherapy to the pelvis.
Experienced gastrointestinal radiologists (MRB and SRR) will draw the regions of interest (ROI) and report the exams. Both radiologists have participated in the MRI rectal cancer workshop organised by the European Society of Gastrointestinal and Abdominal Radiology, and the senior radiologist has 20 years of experience in staging of rectal cancer.
The exams are performed on the already planned MRI scans in the participant’s programme in a blinded fashion, see below. The first T1RT is measured on the primary staging MRI. The second measurement is done on the first re-staging MRI scan 6 weeks after CRT (Figure 2). Flowchart of study design. MRI, magnetic resonance imaging; TNM, tumour, node, metastasis; MDT, multidisciplinary team.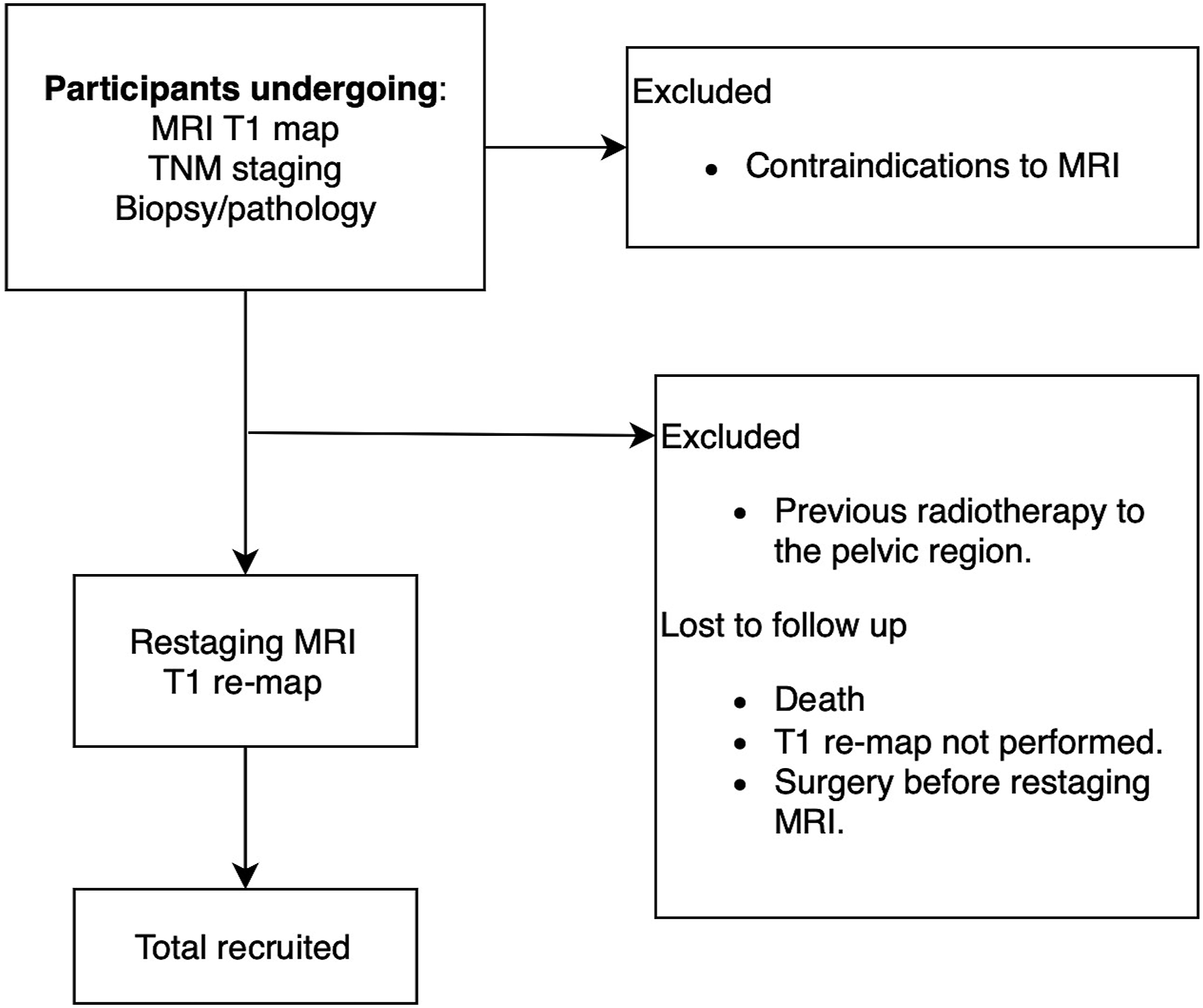
Treatment
Each participant is discussed at a multidisciplinary team conference (MDT). Experienced doctors from oncology, radiology, surgery, and pathology discuss and decide the optimal treatment options that should be offered to the patient. Treatment options are based on the guidelines by the Danish Colorectal Cancer Group (DCCG).
Neoadjuvant treatment regimen.

Neoadjuvant treatment regimen. mrEMVI+, extra mural vascular invasion on MRI; MRF, mesorectal fascia; SCRT, short course radiotherapy; TNT, total neoadjuvant therapy; CRT, chemoradiotherapy; PS, performance status.
For patients with mismatch repair defect rectal cancer, some will be selected for a neoadjuvant immunotherapy.
Patients with tumours >10 cm from the anal verge or above the peritoneal reflection can be offered neoadjuvant treatment if (1) they are at risk of not of achieving radical surgery or (2) have resectable tumours with mrEMVI+.
Treatment can consist of radio- and/or chemotherapy. T1RT is masked/blinded from the MDT during the experiment.
Patients selected for the WW regimen also follow the guidelines from DCCG. There are two ways to be considered for WW, these are primary and secondary organ preserving treatment. The primary organ preserving treatment can be considered for patients with low rectal tumours in category T1-3 M0. The secondary organ preserving treatment can be considered for patients who develop a cCR as assessed by both MRI and endoscopy.
Endpoint
The gold standard of pCR is no histopathologically detectable cancer in the surgical specimen; for patients not undergoing surgery, the gold standard for cCR is that the tumour is not visible on the 3-month follow-up MRI scan (including faded diffusion weighted image (DWI) signal).
After neoadjuvant treatment, MRI T1RT is measured again and compared with the gold standard. The response assessment should be based on all three modalities: endoscopy, T2W-MRI, and DWI. If any discrepancies should arise between modalities, the overall response assessment should rely more on endoscopy than on MRI. 17 A near-complete response evaluation/term is used in the first 6 months from the end of CRT.
The histopathological methods chosen are the postsurgical pathological tumour node (ypTN) tumour-node-stage version 8 and five point Mandard Tumour Regression Grade system. 18
MRI and T1 relaxation time measuring
Scan parameters of the rectal MRI examinations on 1.5 T MRI system.

MRI, magnetic resonance imaging. FOV, field of view. AP, anterior-posterior. RL, right-to-left. FH, foot-head. All T2-weighted images are made with MultiVane-XD. *Sequences axial to tumour axis.

54y female, with an annular tumour (marked with green) in the lower part of the rectum. The figure originates from the IntelliSpace software. (A1) displays the T1 weighted relaxation time sequence and (A2) on the right displays the derived T1 map in yellow and blue colours. This tumour has a relaxation time of 1356 milliseconds at time of staging. Bright yellow areas represent a higher relaxation time as per the colour scale on the far. (B1) and (B2) show the tumour with reduced thickness after neoadjuvant radio chemotherapy, 62 Gray/28 fractions concomitant with capecitabine. Relaxation time at this point is 1103 milliseconds.
On the T1 map, the ROI is drawn on the border of the tumour using a free-hand technique. The ROI is drawn after evaluating the tumour on the T2 weighted MRI sequences. Areas of artefacts are excluded. The mean values will be calculated for the relaxation time. In cases where the tumour spans multiple slices, ROIs are marked on each slice with visible tumour.
All T1RT measurements will be done blinded. The ROIs are drawn blinded to the histopathological properties of the tumour and choice of treatment and the 3-month follow-up MRI. At the second T1RT measuring, the radiologists are also blinded to the initial T1RT measurement. The same procedure is performed on the re-staging MRI examination.
Volume and diffusion restriction
Volume is measured by drawing around the periphery of the tumour on each image with visible tumour using free hand technique in RIPA SYD (Koninklijke Philips N.V., Amsterdam, the Netherlands). The software calculates the volume.
As all rectal cancers present diffusion restriction on MRI, the diffusion restriction properties are evaluated by the ADC. The b-values are 200-400-600-800-1000 as these values are standard in cancer evaluation in the department.
Sample size
The null hypothesis posits that T1RT does not have diagnostic value when determining fibrosis in a rectal cancer following neoadjuvant treatment.
According to sample size calculations, we must enrol 76 participants to achieve a statistical power of 80% with an α = 0.05. This is based on the assumption of a 10% complete response rate and accounting for 20% of missing data/loss to follow-up.
This sample size calculation is done on numbers from a recent study, which included 63 participants. 15
Statistics
The paired t test will be used for descriptive statistical analysis of MRI T1RT, comparing measurements at the time of diagnosis with those after neoadjuvant treatment during restaging.
Receiver operating characteristic (ROC) curves are used to evaluate the diagnostic performance of pre-and posttreatment T1 RT when detecting response to neoadjuvant treatment, using conventional MRI as the gold standard to define response. In addition, a T1RT cut-off value for complete response and or near complete response will be determined with the Youden Index. 19
Correlation coefficients will be used to display the possible correlation between T1RT and tumour volume and ADC, respectively.
The intraclass correlation coefficients (ICC) will be used for evaluating the inter- and intraobserver variability of T1RT measures.
Statistical analyses will be performed in Stata (version 18.0) software (StataCorp, Texas, USA). A p-value <.05 is considered statistically significant.
Ethics
The T1RT measurement will not pose any risk to the participants, as the measurement is done after the previously planned MRI scan has been completed. Furthermore, a power analysis has been conducted, and the time measurement during the MRI scan is performed in a blinded manner as described above. An MRI contrast agent is not used in the evaluation of rectal cancer. This project has been approved and accepted by The Regional Committees on Health Research Ethics for Southern Denmark. Number: S-20230004.
Data sharing is not applicable to this article as no datasets were generated or analysed.
Discussion
In this study, we want to investigate the diagnostic value of MRI T1RT prospectively when evaluating rectal cancer subsequent to neoadjuvant treatment.
If the T1RT measurement is reliable for predicting and monitoring treatment response in rectal cancer, patients with rectal cancer may benefit from more tailored and less invasive precise treatment options. This is particularly relevant in the context of neoadjuvant treatment, where accurate assessment of treatment response is crucial for guiding subsequent clinical management decisions. Some patients may avoid surgery with the risk of a permanent stoma.
T1RT can potentially be a new non-invasive imaging biomarker for evaluation of treatment response. MRI is the standard imaging procedure during diagnostic workup for rectal cancer; therefore, the only extra inconvenience for the patients is a slight prolongation of the scan time. No extra visits are required during this project.
One of the key strengths of utilising MRI T1RT lies in its ability to provide quantitative measurements, thereby enabling objective assessment of treatment response. Unlike conventional imaging techniques, which also rely on qualitative interpretations, T1RT will probably offer a more reproducible mean of evaluating therapeutic efficacy compared to ADC measuring. DWI is known to improve evaluation of treatment response in rectal cancer compared to traditional MRI. 20 Though it is well known that DWI is highly dependent on the imaging parameters as well as the magnet strength, furthermore, DWI sequences are potentially artefact-prone. 21 This quantitative approach not only enhances diagnostic accuracy, but also facilitates longitudinal monitoring of treatment response over time. A constraint of the novel T1RT method resides in the slice thickness; however, it is anticipated that this parameter may be reduced in the future.
In the future, investigating the interplay between radiomics and T1RT could further enhance our ability to evaluate and predict treatment response in rectal cancer.
In conclusion, we expect to find a lower relaxation time in fibrotic tissue compared to non-responsive cancerous tissue. This could have the potential to change clinical practice.
Footnotes
Author contributions
Mathias Rosenfeldt Byriel, Torben Frøstrup Hansen, Hans Rahr, Lars Henrik Jensen, Signe Timm, Jan Lindebjerg, Mazen Schnefeldt, and Søren Rafael Rafaelsen: Substantial contribution, drafting, final approval, and agreement to be accountable.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Appendix
Search strings
PubMed
((“rectum”[MeSH Terms] OR “rectum”[All Fields] OR “rectal”[All Fields] OR “colorectal”[All Fields])
AND (“neoplasms”[MeSH Terms] OR “cancer”[All Fields] OR “tumor”[All Fields] OR “adenocarcinoma”[MeSH Terms] OR “carcinoma”[MeSH Terms] OR “carcinoid tumor”[MeSH Terms] OR “malignant”[All Fields] OR “malignancy”[All Fields] OR “polyps”[MeSH Terms] OR “adenoma”[MeSH Terms] OR “benign”[All Fields]))
AND (“T1 mapping”[MeSH Terms] OR “T1 mapping”[All Fields] OR “T1 relaxation time”[All Fields] OR “T1 relaxation”[All Fields] OR “T1 times”[All Fields] OR “T1 relaxation times”[All Fields] OR “longitudinal relaxation time”[All Fields])
Embase
((‘rectum’/exp OR rectum:ti,ab OR rectal:ti,ab) AND (cancer:ti,ab OR ‘adenocarcinoma’/exp OR malignant:ti,ab OR malignancy:ti,ab) AND (‘T1 mapping’:ti,ab OR ‘T1 relaxation time’:ti,ab OR ‘T1 relaxation’:ti,ab OR ‘longitudinal relaxation time’:ti,ab))
Medline (Ovid)
((“rectum”/exp OR rectum:tw OR rectal:tw) AND (cancer:tw OR “adenocarcinoma”/exp OR malignant:tw OR malignancy:tw) AND (“T1 mapping”: tw OR “T1 relaxation time”:tw OR “T1 relaxation”:tw OR “longitudinal relaxation time”:tw))
