Abstract
Osteosarcomas predominantly manifest in the long bones of the extremities, with rare occurrences in the skull. A case involving of a 53-year-old female who presented to the authors’ hospital for examination due to dizziness was incidentally found to have an occipital bone mass, which was initially diagnosed as a benign tumor and did not receive sufficient attention. Two years later, owing to tumor enlargement, the patient underwent further evaluation at the same institution, which revealed evidence of occipital bone destruction. Pathological analysis confirmed the diagnosis of osteosarcoma. The patient underwent surgical resection followed by radiotherapy. Despite its infrequency and uncharacteristic initial presentation, skull osteosarcomas should not be overlooked.
Keywords
Introduction
Osteosarcoma occurs mainly in the long bones and is more common in younger age groups; the second peak is in patients >60 years of age. 1 Osteosarcomas of the head and neck are rare, accounting for approximately 6%–10% of cases.2,3 Of these, <2% occur in the skull.4,5 Herein, we describe in detail a case of occipital osteosarcoma, including its clinical history, imaging findings, treatment, and prognosis. Additionally, a brief review of the relevant literature is also presented.
Case report
A 53-year-old female presented to the authors’ hospital in 2019 with dizziness. Head computed tomography (CT) revealed a bony mass in the left occipital region (Figure 1(a) and (b)). The mass was slightly uneven in density with a narrow base attached to the left occipital bone. The tumor was diagnosed as benign based on its morphological features and the integrity of the adjacent bones; as such, the lesion was not considered to be serious. The patient had chronic hypertension, for which control was not ideal for a prolonged period. Cerebrovascular disease was diagnosed after excluding other diseases, and the physician prescribed medications to control high blood pressure and improve circulation. Over the next 2 years, the patient experienced intermittent dizziness, which improved after taking medication. In 2021, the patient’s dizziness worsened and was accompanied by numbness of the left fingers, which was difficult to relieve with drug treatment. In addition, she felt that the mass in her left occipital region continued to grow and returned to the authors’ hospital. CT revealed a tumor with bone destruction, which was significantly larger than 2 years previously (Figure 1(c) and (d)). Magnetic resonance imaging (MRI) revealed a soft tissue mass in the left occipital region with heterogeneous signal intensity (Figure 2). The diffusion-weighted imaging (DWI) sequence revealed partial diffusion restriction, and the apparent diffusion coefficient (ADC) map revealed a significantly low signal. The enhanced scan revealed noticeable heterogeneous enhancement. Based on the clinical manifestations and imaging findings, a malignant tumor was considered, followed by chest and abdominal examinations to confirm whether metastasis had occurred. Chest CT revealed small pulmonary nodules, each measuring <3 mm, with no signs of metastasis. The patient underwent surgery, followed by radiotherapy. No signs of recurrence were detected during the two follow-up appointments in 2022 and 2023 (Figure 3). CT images before surgery. a and b (2019) revealed a tumor in the left occipital region without any bone destruction; the mass was attached to the left occipital bone with a narrow base (arrow). c and d (2021) revealed the tumor was larger than it was two years ago and with bone destruction (arrows). Axial MRI images before surgery (T1WI, T2WI, DWI, and enhanced T1WI images), a–d (2021) revealed a soft tissue mass in the left occipital region (arrows in (a and b)). DWI sequence showed partial diffusion restriction (arrows in (c)), and ADC map showed low signal (arrows in (d)). The tumor was heterogeneous and significantly enhanced. The left transverse sinus was involved (arrow in (e)). Axial MRI images (enhanced T1WI images) after surgery in 2022 (a) and 2023 (b) revealed that the left occipital bone was interrupted, the left occipital soft tissue mass excised, and no definite abnormal enhancement was observed (arrows).


Intraoperatively, the tumor was found to be located in the left occipital area with bone destruction. The tumor was tough and adhered to the dural membranes. Incisional biopsy revealed that most of the tumor cells were fusiform and epithelioid, with moderate to severe atypia. Mitotic images were easily visible and there was an abundance of fissure-like blood vessels in the interstitium; an osteoid matrix was observed in some areas. Additionally, binuclear cells with large nuclei and atypia were observed (Figure 4). There was also evidence of direct production by osteosarcomas is the presence of osteoids and an unmineralized extracellular matrix.
6
Osteosarcoma cells can express many different types of antigens but lack diagnostic-specific antigens. Combined with the immunohistochemistry, SATB2 was positively expressed. Although it has low specificity, it supported the diagnosis of osteosarcoma.
7
Pathological examination showed calcium-salt deposited bone (arrow heads in (a)) and immature bone (arrows in (a)), and spindle-shaped cells exhibit significant atypia (arrows in (b)).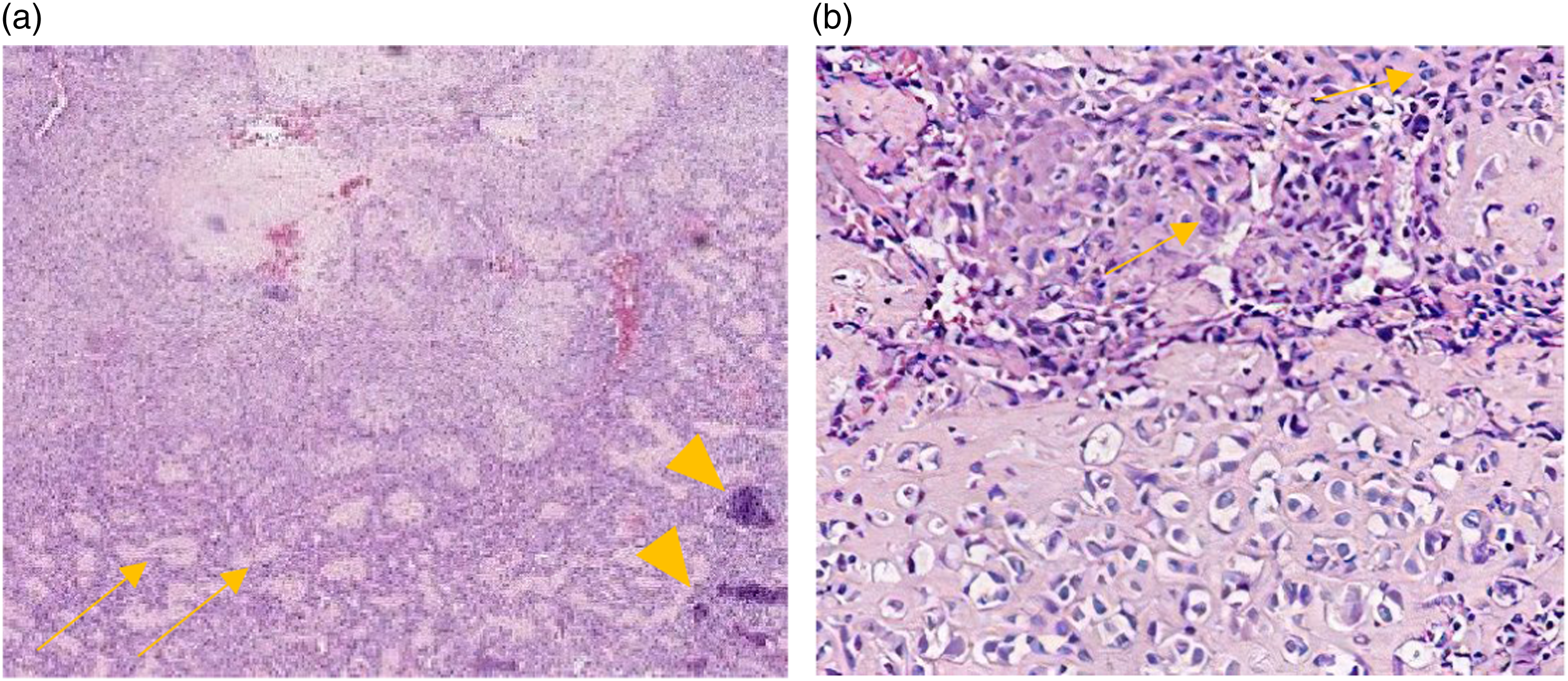
Discussion
The present report describes the clinical and imaging findings in a patient with skull osteosarcoma―a relatively common tumor―present in an uncommon location. Primary skull osteosarcoma mainly occurs in adolescents and individuals >50 years of age, 4 similar to osteosarcoma of the long bones. Luo et al. 8 reviewed 37 patients with head and neck osteosarcoma and found that skull osteosarcoma mainly occurred in the craniofacial bones, especially in the upper jaw, and rarely in the skull. In addition, virtually all reported cases in the skull involve bone destruction, and early manifestations are extremely rare. The clinical presentation depends on the location of the tumor, with most patients presenting with mass lesions sometimes accompanied by pain, whereas others may experience symptoms such as headache, exophthalmos, and visual impairment.5,9
Many researchers believe that skull osteosarcomas are associated with radiation exposure and trauma. Of 12 patients with calvarium and skull base osteosarcomas in a study by Luo et al. 10 six had a history of radiotherapy for nasopharyngeal cancer. Ramezanpour et al. 11 reported a case of skull osteosarcoma with multiple previous head traumas. Ronette et al. 12 reported a case with a history of head trauma 3 years before the discovery of the tumor; however, whether this was related to the development of the tumor is unknown. In our case, the patient had no relevant medical history. The etiology of skull osteosarcoma is unknown, and radiation or trauma may increase risk.
CT has important advantages for the evaluation of bone conditions. Skull osteosarcoma is similar to osteosarcoma of the extremities and can be classified into osteogenic, osteolytic, and mixed types. 10 The tumor margins are always irregular. Osteosarcoma is highly suggestive when osteosclerosis co-exists with bone destruction.8,10 Aggressive reactions revealed on CT include uncommon spiculated (sunburst or hair-on-end), disorganized, laminated, or Codman triangular reaction patterns, which are uncommon. 10 In this case, we observed a soft tissue mass with irregular bone destruction and fragmentation on CT images in 2021. Studies addressing the early manifestations of osteosarcoma are limited. In the present case, CT revealed a narrow basal bony mass with uneven density. Initially, it was misdiagnosed as a benign tumor and an osteoma was considered. However, there were many doubts, such as the density of the bony mass in this patient is not as high and dense as that of osteoma, and osteoma presents mostly as a broad basal attachment. However, the absence of soft tissue mass, well-defined bony mass, or obvious bone destruction at the time misled the diagnosis.
MRI is better than CT for revealing soft-tissue infiltration of the tumor. Osteosarcoma exhibits heterogeneous or low-signal intensity on T1-weighted images, and high, low, or heterogeneous signal intensity on T2-weighted images. 10 The composition of osteosarcoma is complex and may include hemorrhage, necrosis, or an osteoid matrix, 10 with no specific features on plain MRI. 8 DWI is superior in differentiating between benign and malignant tumors. Mansout et al. 13 reported that a mean apparent diffusion coefficient (ADC) value of 0.84 × 10-3 mm2/s was the best cut-off for distinguishing between malignant and benign bony lesions, for which sensitivity, specificity, and accuracy were 88.89%, 88.57%, and 88.71, respectively. Enhanced MRI can be used to assess soft-tissue involvement, such as the brain parenchyma, cerebral arteries or sinuses, bone, bone marrow, and skull base. 14 In the present case, the DWI sequence demonstrated a speckled high signal, the enhanced sequence exhibited obvious uneven enhancement, and the left transverse sinus was involved. These findings were suggestive of malignancy. However, it can still be confused with other cranial malignancies that need to be confirmed pathologically.
For skull osteosarcoma, achieving complete resection with adequate margins is reported to be the most critical factor for long-term survival15,16 and is key to the success of the surgery. 17 In patients with poor prognostic indicators, incomplete clearance, and advanced stages, additional radiotherapy is necessary. 15 However, for negative margins, due to the risk for radiation toxicity, there is much debate regarding whether adjuvant radiotherapy is required.16,18 Chemotherapy has already demonstrated a significant effect on extremity osteosarcoma; however, in skull osteosarcoma, it remains controversial.16,17
In conclusion, osteosarcomas are rarely observed in the skull. It can be challenging to make an accurate diagnosis as to whether bone destruction has occurred. CT and MRI are essential for preoperative diagnosis. Owing to individual differences, multimodal treatment plans must be developed according to histological subtype for individual diagnoses.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
