Abstract
Intraductal papillary mucinous neoplasms (IPMN) of the pancreas have the potential for malignant progression into adenocarcinoma. Colloid or mucinous non-cystic carcinoma of the pancreas is an uncommon variant neoplasm that can arise within an intestinal type IPMN and have a relatively improved prognosis but may mimic the more lethal tubular or ductal adenocarcinoma. Colloid carcinoma is an infiltrating ductal epithelial neoplasm containing primarily extracellular stromal mucin pools and scant amount of centrally floating neoplastic cells. While several reports have evaluated the unique pathologic and immunohistochemical profile of colloid carcinomas, there has been limited radiologic–pathologic correlation in the literature. We report a case of an 83-year-old female who presented for evaluation of slowly progressive abdominal pain and was found to have colloid carcinoma arising from an IPMN. This is one of the first reports to correlate the multimodality radiology including cinematic rendering (CR) and histopathology features associated with this tumor. An enhanced understanding of the correlation between imaging appearance and specific histopathologic findings may aid in the early recognition and treatment of this rare neoplasm. Emphasis is placed on CR as this may help guide surgical management.
Introduction
Intraductal papillary mucinous neoplasms (IPMN) are well described pancreatic lesions accounting for 3–5% of all pancreatic tumors. 1 IPMNs are classified into four histopathological types: gastric, intestinal, pancreatobiliary, and oncocytic. 2 When invasive, IPMNs are distinguished as either colloid (mucinous non-cystic) carcinoma or conventional tubular/ductal carcinoma. Colloid carcinoma arises from intestinal-type IPMN and represents 25% of all invasive IPMNs, while tubular/ductal adenocarcinoma arises from pancreatobiliary type IPMN.3–5 These two tumors have similar appearances and clinical presentations.6–8 Colloid carcinoma is differentiated by its less aggressive features and superior long-term prognosis compared to that of conventional ductal carcinoma, with reports of 5-year survival of 40–60% versus 10–15%, respectively.3,5–12 The WHO classification defines both tumors as malignant as they have invasive potential, however, their differing prognoses and metastatic tendencies make it important to carefully diagnose and monitor colloid carcinoma. 2
While several reports have evaluated the unique pathologic and immunohistochemical profile of colloid carcinomas, there has been limited radiologic pathologic correlation of colloid (mucinous non-cystic) adenocarcinoma in the literature particularly with reference to cinematic rendering (CR). We present a case of an 83-year-old female who presented for evaluation of slowly progressive intermittent abdominal pain and was found to have pancreatic colloid carcinoma. To our knowledge, this is one of the first reports to evaluate the radiologic features associated with this tumor in conjunction with pathology. We also explore how CR of computed tomography (CT) images may help in surgical planning and risk stratification.
The pancreas protocol CT is often considered the preoperative gold standard imaging modality to assess for vascular involvement and whether complete tumor resection can be achieved. A standard pancreas protocol CT includes multi-phase dynamic post contrast CT imaging using thin slice thickness, and in some instances standard 3D reconstructions. However, 3D cinematic rendered (CR) images are not routinely obtained. Previous authors have shown significant interobserver variability in determining relevant vascular involvement across different radiologists. Additionally, the diagnostic accuracy of preoperative pancreas protocol CT in predicting pancreatic tumor vascular involvement as compared to surgical findings only ranged from 73 to 83%.13–15 Such uncertainty could lead to unnecessary surgeries and unexpected intraoperative findings.
CR 3D evaluation of tumor vessel interfaces allows for real-time, dynamic assessment where the user can fully manipulate the rendering and view 3D perspectives that would best reveal critical tumor involvement. 13 Light and shadowing are presented in a photorealistic manner which more closely resembles the intraoperative appearance of structures compared to conventional radiologic images and could potentially lead to more accurate preoperative diagnosis. An enhanced understanding of the correlation between imaging including CR and specific histopathologic findings may aid clinicians in earlier recognition and improved risk stratification of this neoplasm.
Case report
An 83-year-old female with chronic obstructive pulmonary disease, hypertension, hyperlipidemia, and insulin-dependent diabetes mellitus presented with a 3-year history of slowly progressive intermittent abdominal pain, nausea, and vomiting and was found to have an 11 × 3 cm cystic pancreatic mass on imaging. Relevant surgical history included remote cholecystectomy and right breast mastectomy. Physical exam was notable for a soft, non-tender abdomen without palpable mass. Serum laboratory testing revealed normal values for CEA 2.7 ng/mL (normal <5.2 ng/mL) and CA19-9 26.8 U/ml (normal <35 U/ml).
Radiologic imaging ultimately yielded a suspicious complex cystic mass involving the body and tail of the pancreas for which surgical resection was warranted. The patient underwent distal pancreatectomy and splenectomy with curative intent. The histopathology was compatible with colloid adenocarcinoma arising within an IPMN and histopathologic analysis suggested the tumor was completely confined to the pancreas and completely excised. The patient recovered well from the surgery and was doing well at her last noted post-operative visit 6 months post operation.
Radiologic features
Computed tomography of the abdomen and pelvis performed during the workup showed a complex cystic mass in the pancreatic body and tail with dilation of the main pancreatic duct. Peripheral internal solid components were suggested (Figure 1). No vascular invasion or distant metastases were noted. Findings were suspicious for main duct IPMN and MRI was obtained for further evaluation. (a) Noncontrast axial CT image through the pancreas demonstrates a hypodense mass expanding the pancreatic body and tail. Associated main pancreatic duct dilatation is noted. (b) Arterial phase post contrast CT demonstrates peripheral solid enhancing components within the pancreatic mass. No vascular invasion or distant metastases were noted. (c) Axial fused PET/CT shows hypermetabolic activity within the solid peripheral enhancing components of the pancreatic mass. SUV max of the solid components was 5.2. CT: Computed tomography; PET: Positron emission tomography.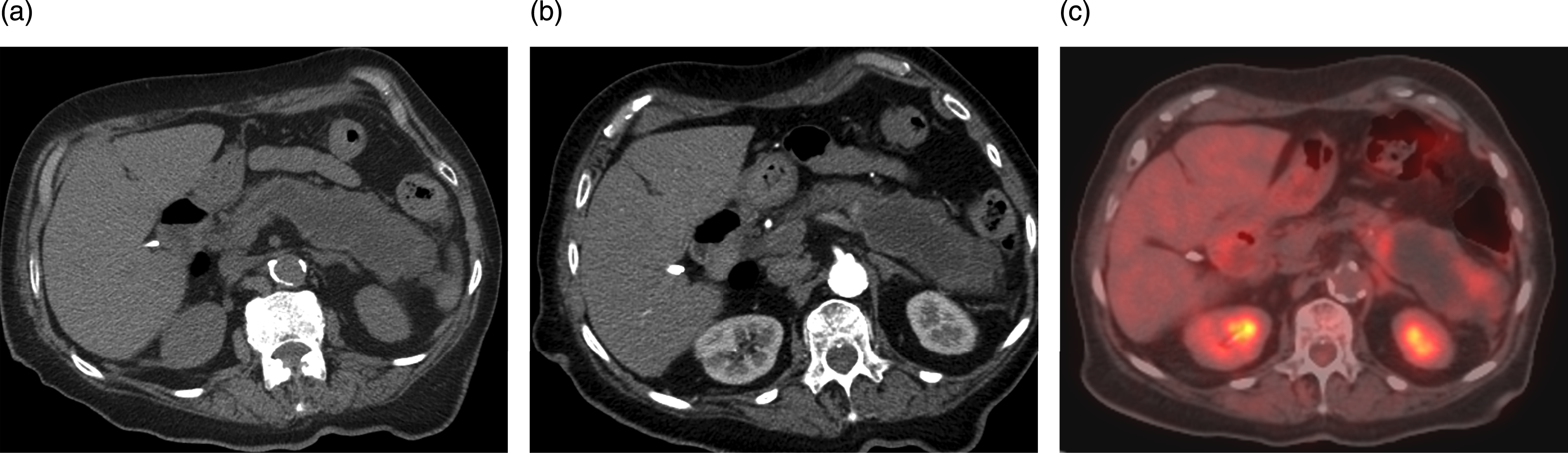
PET/CT showed increased metabolic activity within the solid peripheral components of the complex distal pancreatic mass. Maximum standard uptake value (SUV max) within the lesion was 5.2 (Figure 1).
MRI of the abdomen demonstrated a complex cystic mass involving the main pancreatic duct. Internal soft tissue nodular components showed post contrast enhancement and restricted diffusion (Figure 2). MRI better showed the expansile nature of the mass along with heterogeneous low T2 signal regions within the larger cystic appearing mass which is compatible with the diagnosis of colloid adenocarcinoma in the setting of IPMN. (a) Axial T2 weighted MR shows marked pancreatic ductal dilatation with a complex cystic mass with multiple poorly defined lower T2 signal filling defects. (b) Axial T1 weighted fat saturated non-contrast and (c) arterial phase post contrast show central low T1 signal with peripheral increased T1 corresponding to the solid peripheral components. (d) Coronal T2 weighted MR demonstrates the expanded nature of the pancreatic tail secondary to a high T2 signal mucinous lesion. (e) Axial DWI b value 1000 and (f) Axial ADC map demonstrate multi focal restricted diffusion within the pancreatic tail and body lesion. MR: Magnetic resonance; DWI: Diffusion weighted imaging; ADC: Apparent diffusion coefficient.
Endoscopic ultrasound (EUS) demonstrated a complex mixed cystic and solid mass involving the body and tail (Figure 3). The mucinous nature of internal components is reflected by internal echogenic components on EUS. (a) and (b) Endoscopic ultrasound images of the large pancreatic mass involving the body and tail. The complex cystic nature is appreciated with the overall hypoechoic appearance of the mass with multiple scattered hyperechoic foci corresponding to the mucinous nature of colloid adenocarcinoma.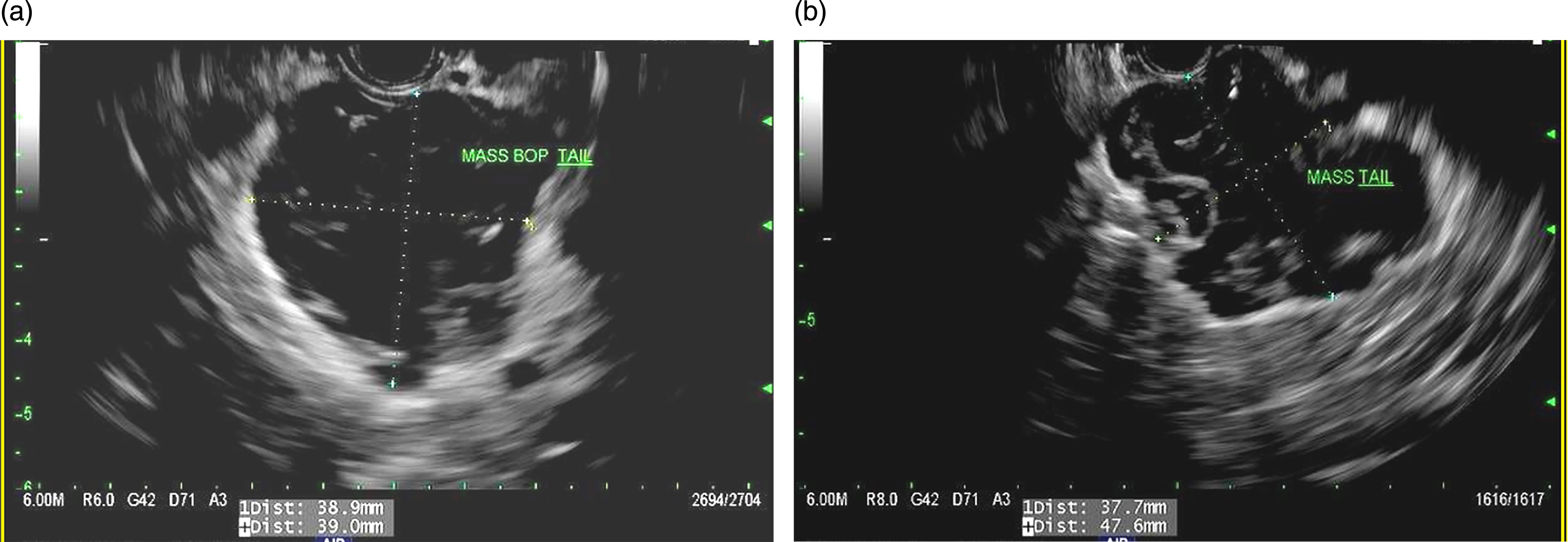
CR reconstructions were performed on CT venous phase images (Figure 4). In this case, CR allowed enhanced preoperative visualization of the peripancreatic vasculature to support the notion that no critical tumor vessel involvement was present. Additionally, CR improved the surgical confidence that no unexpected intraoperative findings such as altered vascular anatomy would be encountered. CR also helped to further delineate the complex cystic and solid configuration within the tumor itself which could help suggest colloid carcinoma over other more solid tumors. (a) Axial CT image (cinematic rendering) through the pancreatic tail mass. (Syngo Via, Siemens Healthcare, Erlangen, Germany) (b) Corresponding axial post contrast venous phase image through the pancreatic mass. (c) Coronal oblique cinematic rendered image through the pancreatic tail mass. Note the visualization of vasculature surrounding the pancreatic lesion. (d) Corresponding post contrast coronal reconstructed CT image. CT: Computed tomography.
Pathologic features
Gross examination revealed a 17.5 × 7.0 × 4.5 cm distal pancreas and spleen with a moderate amount of associated peripancreatic and splenic hilar adipose tissue. The pancreatic duct lumen was dilated up to 1.1 cm and visible at the resection margin extruding tan and mucoid material. The pancreas was sectioned along its long axis, revealing an 8.0 × 3.5 × 2.8 cm multilocular cystic lesion replacing the pancreatic duct and extending to the distal end of pancreas. Approximately 1 cm from the pancreatic margin of resection within the wall of the cyst was a 3.5 × 3.0 × 2.0 cm pink–white, fibrotic area with unevenly distributed smaller cysts. Microscopic evaluation revealed abundant mucin which was expanding an intestinal type IPMN with overt high-grade dysplasia. The lesion was focally lined by atypical cells and rare atypical cells were floating within the mucin. Focal fibrotic reaction was noted in the surrounding stroma (Figure 5). Mucinous (colloid) adenocarcinoma arising in an IPMN. (a) and (b) Low and high magnification of IPMN with pancreatic parenchyma alteration and high-grade morphology (arrows). (c) and (d) Colloid carcinoma and mucin dissecting fibrotic stroma, with detachment (arrow). (e) Colloid carcinoma abuts an artery (arrow); benign glands do not share this behavior. (f) Colloid carcinoma and mucin surrounds and abuts a nerve bundle (arrow). IPMN: Intraductal papillary mucinous neoplasms.
Immunostains for IgG and IgG4 performed on tissue block B6 show focal features of IgG4-related chronic pancreatitis. Immunostains for synaptophysin and chromogranin performed on tissue block B18 were negative. The immunohistologic features were consistent with colloid adenocarcinoma, stage PT2, PN0, arising in an IPMN.
Discussion
Pathologic and radiologic findings in pancreatic tumors.

Note: Reference table for histology, immunohistochemistry and radiology findings for differential considerations in pancreatic tumors commonly arising from IPMN. Adapted from reference (Bosman, 2010; Ren, 2010).
CT: computed tomography; IPMN: intraductal papillary mucinous neoplasm; MRI: magnetic resonance imaging.
Radiologically, colloid carcinoma appears as a poorly enhancing low attenuated mass on contrast-enhanced CT and very high signal intensity on T2-weighted MRI due to abundant extracellular mucinous components. On dynamic contrast-enhanced imaging, they show central poorly enhancing mucin pools with gradual peripheral and internal mesh-like enhancement of the intervening stroma causing a salt-and-pepper-like appearance.17–19 Conversely, tubular/ductal carcinomas appear as a hypodense mass with upstream dilatated biliary and pancreatic ducts, resulting in the “double-duct sign.” 2 MRI demonstrates enhancement with ductal obstruction but are often hypoattenuating in the pancreatic and portal venous phases secondary to fibroblastic proliferation and decreased vascularity. 20
Given the rarity of colloid carcinoma, there are no specific widely acknowledged preoperative workup guidelines currently available. The recommended diagnostic workup of colloid carcinoma is therefore the same for ductal adenocarcinoma, which includes imaging with CT or MRI and EUS.6–8 Colloid carcinomas are typically diagnosed after surgical resection, rather than during the preoperative workup.
Due to its low prevalence, treatment guidelines for colloid carcinoma mirror those for ductal adenocarcinoma. For resectable tumors, surgery is the recommended treatment, which can include pancreaticoduodenectomy, distal pancreatectomy, or total pancreatectomy based on location. Treatment with adjuvant therapy similar to ductal adenocarcinoma is recommended as comparable studies for colloid carcinoma specifically have not been performed.21,22
This is where CR may play a vital role in diagnostic workup and surgical management. Some authors have closely analyzed the preoperative CT features between colloid carcinoma and tubular carcinoma but did not correlate this with histopathologic findings. 23 Others have explored the utility of CR in improving pancreatic cancer surgical planning but mainly in regard to ductal adenocarcinoma and not colloid carcinoma. 24 As mentioned previously, CR is an emerging technology with roots in animation that uses multiple light sources to produce astoundingly life-like 3D images and exceptional visualization of nuanced anatomy, vascular supply, and surrounding structures. The settings can be manipulated to accentuate subtle differences in features such as internal architecture, enhancement, and vascular involvement.
Regarding surgical planning, CR is transpiring as a tool to determine resectability, assess arterial and portovenous involvement, identify anatomic variants, and detect occult metastases in other pathologies including pancreatic cancer, hepatocellular carcinoma, and intrahepatic cholangiocarcinoma, so it would not be a stretch to apply these same principles in discriminating colloid carcinoma and tubular carcinoma. 24 Our case showed a cystic mass in the pancreatic body and tail which communicated with a dilated pancreatic duct. It exhibited peripheral areas of mural nodularity consistent with the salt-and-pepper mesh-like appearance with some invasion of the peripancreatic fat. These imaging findings reliably reflect underlying histopathologic characteristics that can be accentuated with CR to aid in early recognition of these neoplasms.
Colloid carcinoma is pathologically defined as an infiltrating ductal epithelial neoplasm of pancreas characterized by extracellular stromal mucin pools in >50% of the neoplasm and scant centrally floating neoplastic cells.18,25 The lesion is usually >1 cm in diameter, arises from the main pancreatic duct or branch ducts, with varying degrees of duct dilatation.26,27 Colloid carcinoma cells exhibit inverse polarization, whereby basilar cells secrete mucin between the stroma and cells instead of toward the lumen, thereby separating the cell from the underlying stroma. The mucin surrounding the epithelial cells then serves as a physical barrier, allowing for predominantly expansile growth of the tumor rather than invasion of neoplastic cells.9,18,25,28 As presented in our report, this marked expansion of the lesion with presence of mucin pools pushing their way into the wall of the pancreatic duct corresponds with the heterogenous T2 weighted MRI appearance of the lesion.
Immunohistochemical profiles are often helpful in aiding the differentiation between colloid (mucinous non-cystic) and ductal carcinomas (Table 1). Immunohistochemical studies of colloid carcinoma, which is associated with intestinal-type IPMN, typically reveal diffuse expression of the surface glycoproteins CDX2 and MUC2 (features of intestinal differentiation). 11 While these were not available in our case, they would have been helpful in further confirming the diagnosis. In contrast, conventional ductal carcinoma, which is most often associated with pancreatobiliary type IPMN, exhibits the more aggressive MUC1 without MUC2 or CDX2 expression.5,11,12,29–31 MUC2 may exhibit tumor suppressor activity, which could explain the superior prognosis of colloid carcinoma and contribute to its indolent nature. 28
In conclusion, colloid carcinoma of the pancreas is a rare subtype of IPMN-associated epithelial tumor that necessitates further exploration. Colloid carcinoma of the pancreas presents radiologically as a poorly enhancing, low attenuated cystic mass with abundant mucin and peripheral and internal mesh-like enhancement of the intervening stroma. This is one of the first dedicated reports to evaluate the multimodality radiologic features with CR in conjunction with histopathology of this tumor type. Future studies involving CR of CT images would be helpful in defining its benefits in clinical and surgical management of these specific pancreatic cancers. An enhanced understanding of the clinical history, radiologic appearance, and histopathology can aid clinical teams in the early recognition of this rare neoplasm and distinguish it from the more lethal ductal adenocarcinoma.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by National Cancer Institute of the National Institutes of Health, No. R37CA229810.
