Abstract
Introduction
Intraductal papillary mucinous neoplasm (IPMN) is a type of pancreatic cystic neoplasms (PCNs), 1 and is also a precursor lesion of pancreatic cancer. IPMNs exhibit a broad spectrum of neoplastic transformation ranging from low-grade dysplasia (LGD) to high-grade dysplasia (HGD) and invasive carcinoma (IC).2–4 The natural history of potential malignant transformation remains uncertain and therefore the clinical management of patients with IPMNs is controversial.5–7
With the widely use and improvement of high-resolution imaging examinations, the number of patients with IPMNs has increased significantly. Hence, it is important to identify the surgical indications accurately in the management of IPMN patients. Based on radiology and/or histology studies, IPMNs can be classified into three morphological types, main duct type IPMNs (MD-IPMNs), branch duct type (BD-IPMNs), and mixed type IPMNs (MT-IPMNs). 8 The probability of HGD/IC in the MD-IPMNs or MT-IPMNs is 38%–68%, and the malignant risk for patients with BD-IPMNs may range from 12% to 47%. 9 Therefore, MD-IPMNs and MT-IPMNs are usually regarded as absolute indications for surgery in several guidelines.8, 10 However, preoperative imaging examinations maybe overdiagnose MD-IPMNs and MT-IPMNs, which may result in unnecessary surgical risk. 11 In addition, the current guidelines, such as the 2018 European Evidence-based Guidelines on Pancreatic Cystic Neoplasms, 10 the 2017 International Association of Pancreatology (IAP) guidelines, 8 the 2015 American Gastroenterology Association (AGA) guidelines, 12 and the 2018 American College of Gastroenterology (ACG) guidelines, 13 agreed that the risk of malignancy should be balanced against the life-expectancy and comorbidity. But the recommended IPMNs surgical indications are still different. 14 How to apply existing guidelines to improve the accuracy of IPMNs diagnosis in clinical practice remains to be further studied.
Main pancreatic duct (MPD) diameter is one of the critical indicators for surgical resection. However, the threshold value of 1.0 cm is challenged by several studies. Previous research showed that MPD in European and American populations suggests a much smaller threshold.15–17 Due to European, American, and Asian populations may have an ethnical heterogeneity, whether these data apply to Asian populations remains unclear. To the best of our knowledge, there are no relevant studies on Asian populations. Hence, in the present study, we explored the related factors of HGD/IC-IPMNs based on populations in southeastern China and compared the efficiency of current mainstream guidelines in diagnosing HGD/IC-IPMNs. Moreover, we calculated the threshold of MPD in identifying malignant IPMNs and constructed a regression coefficient-based predictive nomogram model.
Material and Methods
Patients
The study was approved by the Institutional Review Board of Affiliated Hospital of Nanjing University of Chinese Medicine (2017NL-137-05), and the requirement for informed consent was waived due to its retrospective nature. The reporting of this study conforms to STROBE guidelines. 18 This retrospective study included 151 patients with pathologically proven IPMNs who underwent surgery between 2017 and 2020. The inclusion and exclusion criteria of IPMN patients were as follows. We included patients who had confirmed histological diagnosis of IPMNs and completed preoperative magnetic resonance imaging (MRI) examination. We excluded patients who (1) had medical history of other tumor, (2) had received any treatment (chemotherapy, radiotherapy, chemoradiotherapy) before surgical resection. This study has de-identified all patient details.
Data Collection
Demographic information, clinical features, laboratory testing, and preoperative radiological characteristics were collected at the time of diagnosis. Demographic information included gender, age; clinical features included pancreatitis and symptoms (jaundice and abdominal pain). Laboratory testing included the serum levels of carbohydrate antigen 19-9 (CA 19-9) and carcinoembryonic antigen (CEA).
MRI Protocol
All patients underwent preoperative MR imaging examinations (Signa Excite HDx 3.0T, GE Medical Systems, USA). MRI sequences included T1-weighted images [repetition time/ echo time (TR/TE) = 180/2 ms; section thickness, 5 mm; spacing, 1 mm], T2-weighted images [TR/TE = 6000/90 ms; section thickness, 5 mm; spacing, 1 mm], 2D magnetic resonance cholangiopancreatography (2D-MRCP)[TR/TE = 1460/1220 ms; section thickness, 40 mm; field of view (FOV), 340*340 mm], 3D-MRCP [TR/TE = 4615/700 ms; section thickness, 3 mm; FOV, 400*400 mm]. For contrast enhanced T1-weighted images, intravenous Gd-DTPA (Bayer-Schering, Germany) of 0.1 mmol/kg was administered at a rate of 2 mL/s.
Radiological Analysis
Radiological characteristics included types of IPMN, pancreatic duct diameter (MPD), cyst size, tumor location, and the presence or absence of mural nodule (>5.0 mm). MD-IPMN was defined as dilation of the pancreatic main duct diameter with a diameter greater than 5 mm. BD-IPMN was defined as a cystic lesion with a radiographically identifiable branch-duct connection to the main duct. MT-IPMN was defined as a cystic lesion having radiographic characteristics of both main and branch-duct IPMN. The results of preoperative imaging for the extent of involvement of the pancreatic ductal system were compared with surgical histopathology.
Histological Examinations
For pathological evaluations, IPMNs were categorized as low-grade dysplasia (LGD), high-grade dysplasia (HGD), and invasive carcinoma (IC). HGD and IC were classified as high-risk diseases, and LGD was classified as low-risk diseases, according to the World Health Organization classification system published in 2019.
Statistical Analysis
Univariate and multivariate analysis were performed to predict the risk factors associated with high-risk IPMNs. For the differences between low-risk and high-risk IPMNs, categorical variables were compared using the χ2 test or Fisher's exact test, and quantitative variables were compared using Student's t-test or Mann-Whitney U test. The multivariate logistic regression analyses were used to identify the independent predictive factors for high-risk IPMNs. The predictors with a P value < .05 (mural nodules, MPD diameter, CA19-9) and some important clinical factors (gender, age, cyst size) were included in the final model. The receiver operating characteristic (ROC) curves were performed to determine the cutoff levels for the MPD diameter and evaluate the diagnostic ability of the predict factors. Then based on the optimal cutoff levels, a categorical MPD diameter was included in the logistic models again. Spearman correlation analysis was performed to test the association between significant predictors in the logistic model. Finally, a nomogram was constructed to represent the multivariable model, and measured by C-index and calibration plots. Statistical significance was defined as P < .05. All statistical analyses were performed using SPSS 26.0 software, GraphPad Prism 9.0.0 software, or R 4.0.2 software.
Results
Patient Characteristics
The clinical characteristics of all 151 patients with IPMNs are shown in Table 1. Preoperative imaging classified the IPMNs as MD-/MT-IPMNs in 78/151 (51.7%), as BD-IPMNs in 73/151 (48.3%). Low-risk IPMNs accounted for 104/151 (68.9%), and high-risk IPMNs accounted for 47/151 (31.1%). Compared with patients with BD-IPMNs, patients with MD-/MT-IPMNs were more likely to have high-risk diseases (48.7% vs 12.3%, P < .001).
Demographic and Clinical Variables, Diagnostic Work-Up, and Baseline Features of IPMN Population.
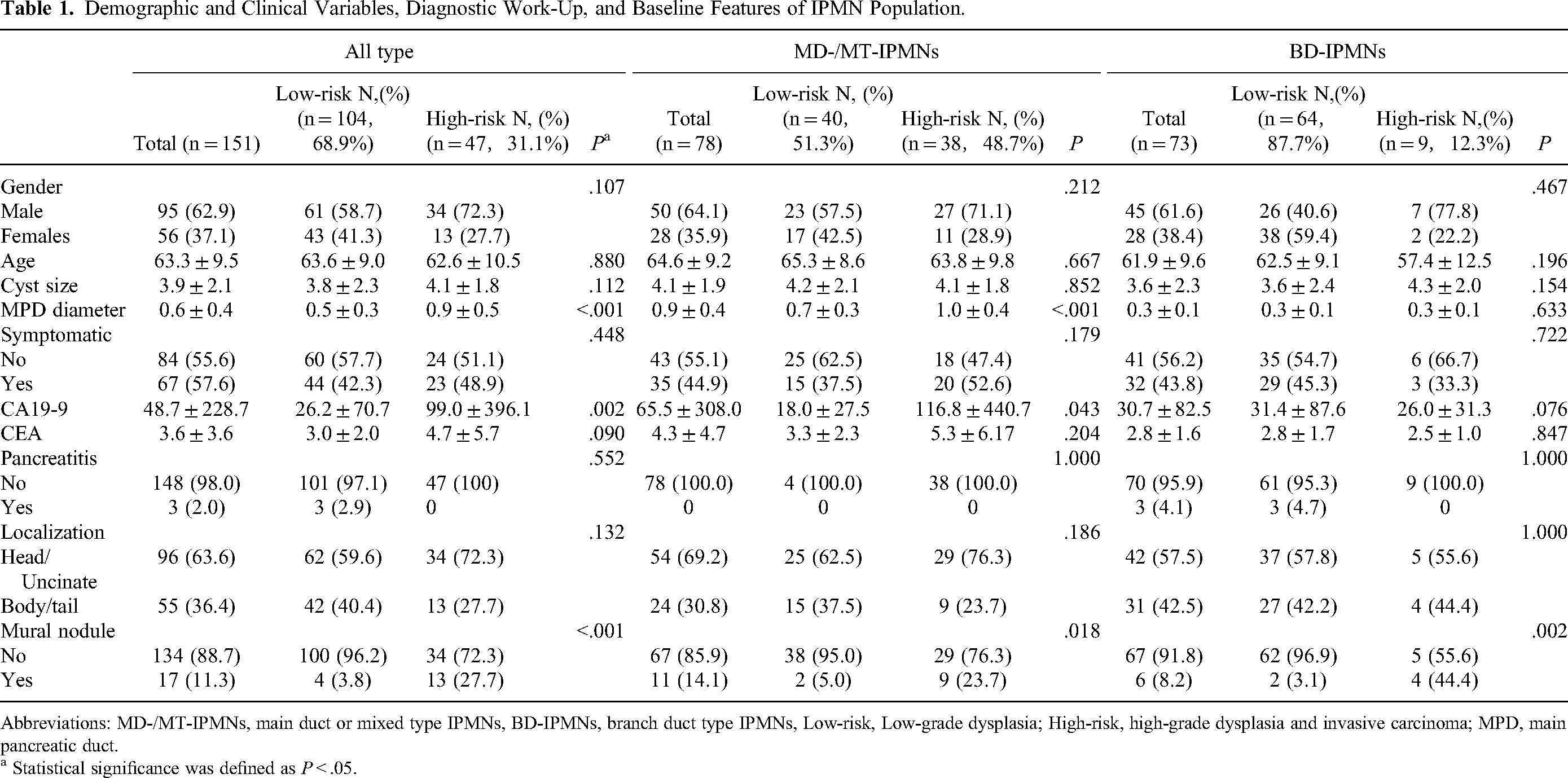
Abbreviations: MD-/MT-IPMNs, main duct or mixed type IPMNs, BD-IPMNs, branch duct type IPMNs, Low-risk, Low-grade dysplasia; High-risk, high-grade dysplasia and invasive carcinoma; MPD, main pancreatic duct.
a Statistical significance was defined as P < .05.
Univariate analysis results showed that MPD diameter, elevated serum CA19-9 levels, and the presence of mural nodule were associated with high-risk IPMNs in both MD-/MT-IPMN subsets and all type IPMN subsets. However, the mural nodule was the only significant predictor for high-risk IPMNs in BD-IPMN subset.
Risk Factors Associated High-Risk IPMNs in Univariate and Multivariate Analysis
Based on above significant variables, the multivariate logistic regression models were constructed, and the cyst size, gender, and age were also included in the models. As shown in Table 2, for all type IPMNs, both MPD diameter (Odd ratio (OR), 24.44; 95% confidence interval (CI), 6.10–97.97; P < .001) and the mural nodule (OR, 10.97; 95% CI, 2.64–45.54; P = .001) remained as the independent associated factors with high-risk IPMNs. For MD-/MT-IPMNs, MPD diameter (OR, 15.07; 95% CI, 2.018–112.491; P = .008) was the only independent associated factor for high-risk IPMNs. For BD-IPMNs, the mural nodule (OR, 33.59; 95% CI, 3.66–308.26; P = .002) was the only independent associated factor for high-risk IPMNs.
Association Between Potential Risk Factors of High-Risk IPMN Were Estimated Using Multinomial Logistic Regression.

Abbreviations: OR, Odds Ratio; 95% CI, 95% Confidence Interval.
a Treated as a continuous variable.
Cutoff Values for MPD Diameter in Predicting High-Risk IPMNs
As shown in Figure 1, for all type IPMNs, the mean diameter of the MPD was 0.5 ± 0.3 cm in patients with low-risk IPMNs and 0.9 ± 0.5 cm in patients with high-risk IPMNs (P < .001). For MD-/MT-IPMNs, the mean diameter of the MPD was 0.74 ± 0.3 cm in patients with low-risk IPMNs and 1.04 ± 0.4 cm in patients with high-risk IPMNs (P < .01). In contrast to the above-mentioned subsets, the MPD diameter was no difference between low-risk and high-risk disease for patients with BD-IPMNs (0.32 ± 0.15 cm vs 0.32 ± 0.11 cm, P = .633).

The scatter plots of MPD diameter in the different subsets of IPMNs. (A) The MPD diameter of low-risk and high-risk groups in all types of IPMN. (B) The MPD diameter of low-risk and high-risk groups in MD-/MTIPMN. (C) The MPD diameter of low-risk and high-risk groups in BD-IPMNs. **P < .01, ****P < .0001.
As shown in Figure 2, ROC curves were used to determine the cutoff values of the MPD diameter in predicting high-risk IPMNs. For all type IPMNs, the cutoff value of the MPD diameter was 0.77 cm, with an area under the curve (AUC) of 0.775, a sensitivity of 61.7%, and a specificity of 87.3%. For MD-/MT-IPMNs, the cutoff value of the MPD diameter was 0.82 cm, with an AUC of 0.737, a sensitivity of 68.4%, and a specificity of 79.5%. Subsequently, we assessed the diagnostic capacities of MPD diameters as the categorical variables using cutoff values recommended by guidelines (>1.0 cm). For all type IPMNs and MD-/MT-IPMNs, the AUC values of MPD diameter >1.00 cm were 0.657 (sensitivity, 38.3%; specificity, 93.1%) and 0.656 (sensitivity, 68.7%; specificity, 62.5%), respectively. The AUC values of MPD diameter >0.50 cm were 0.687 (sensitivity, 76.6%; specificity, 60.7%), and 0.500, respectively.

Performance of the risk factors in predicting the high-risk IPMNs using ROC analysis. (A) Diagnostic values of MPD diameter (continuous data) for all type IPMNs and MD-/MT-IPMNs; (B) Diagnostic values of MPD diameter with different cutoff values (1.0 cm, 0.5 cm, and 0.77 cm) for high-risk lesions in all type IPMNs; (C) Diagnostic values of MPD diameter with different cutoff values (1.0 cm, 0.5 cm, and 0.82 cm) for high-risk lesions in MD-/MT-IPMNs; (D) Diagnostic values of the MPD diameter, mural nodule, and the combination model for high-risk lesions in all type IPMNs.
For all type IPMNs, the AUC value of MPD diameter >0.77 cm was increased to 0.746. For the subset of MD-/MT-IPMNs, the AUC value of MPD diameter >0.82 cm was increased to 0.742. Therefore, compared with the cutoff values of 0.5 and 1.0 cm suggested by the guidelines, the predictive performance for the high-risk IPMNs would be improved with cutoff values of 0.77 cm in all type IPMNs, and cutoff values of 0.82 cm in MD-/MT-IPMNs.
Construction of a Predictive Nomogram for High-Risk IPMNs
Then based on the optimal cutoff values, we focused on the value of MPD diameter as the categorical variables in the differentiation between the low-risk and high-risk IPMNs. As shown in Table 3, besides the mural nodule and MPD diameter, gender, age, location of tumor, and cyst size were also included in a logistic model. For MD-/MT-IPMNs, MPD diameter >0.82 cm (OR, 7.47; 95% CI, 2.32–24.07; P = .001) was still the independent high-risk predictor. For all type IPMNs, multivariate analysis demonstrated that the mural nodule (OR, 12.98; 95% CI, 3.18–52.97; P = .000) and MPD diameter >0.77 cm (OR, 12.67; 95% CI, 4.80-33.48; P < .001) remained the independent predictors. Besides, there was no significant correlation between mural node and MPD diameter (P = .08, r = 0.144). Therefore, we further constructed a combined model (mural nodule and MPD diameter >0.77 cm) to predict high-risk IPMNs. The combined model showed a better predictive performance than mural node or MPD diameter alone (AUC = 0.803 vs 0.619, 0.746), and another combined model (mural nodule and MPD diameter >1.0 cm) with an AUC = 0.722.
Logistic Regression Analysis of Possible Predictors of Malignancy in Subsets of All Type IPMNs and MD-/MT-IPMNs.

a MPD is a categorical variable (cutoff = 0.77 cm).
b MPD is a categorical variable (cutoff = 0.82 cm).
As shown in Figure 3, based on the independent risk factors obtained from logistic analysis (the mural nodule and MPD diameter >0.77 cm), a nomogram was created to predict high-risk disease in all type IPMNs. The C-index of the nomogram was 0.803, and calibration plots showed a good level of agreement with the predicted risk and actual risk.

Clinical nomograms for predicting high-risk lesions in all type IPMNs. (A) The model includes two variables in the nomogram: the mural nodule and MPD diameter (cutoff = 0.77 cm). (B) Calibration curve of the nomogram for predicting high-risk lesions in all type IPMNs.
Discussion
The management of IPMNs remains a challenge because the risk of malignancy is difficult to predict. 19 In recent years, with a growing number of patients diagnosed with IPMNs, the amount of IPMN-related pancreatic surgery has also increased. However, an unnecessary pancreatic surgery for LGD IPMNs will cause a series of problem. In contrast, without early detection and curative resection of lesions with HGD and/or IC might increase pancreatic cancer mortality. Therefore, in the last two decades, several guidelines for managing IPMNs or cystic neoplasms of the pancreas have already been proposed. But none of them reached a consensus because our knowledge of its natural history is not perfect yet. 20
In the present study, we analyzed a cohort of 151 patients with resected IPMNs confirmed by final histology. We found that patients with MD-/MT-IPMNs (48.7%) were more likely to have high-risk IPMNs than BD-IPMNs (12.3%). Our results were similar to the previous studies.9, 21, 22 Hence, surgical resection is recommended for MD-/MT-IPMNs because of their high malignant potential.8, 23, 24 However, the accurate diagnosis of MD-/MT-IPMNs remained a clinical challenge. It was suggested that about 20% of surgical removals of the IPMNs due to presumed MD-/MT-IPMNs were finally identified as BD-IPMNs, 25 which may make patients undergo unnecessary surgical procedures risks.
Our data confirmed several risk factors for HGD/IC IPMNs described in the current guidelines. MPD diameter, elevated serum CA19-9 level, and presence of mural nodule were the most important predictor for high-risk IPMNs in both all type IPMNs and MD-/MT-IPMNs subsets. Furthermore, in the multivariate analysis, we found that MPD diameter and existing mural nodule were the independent predictors for high-risk IPMNs in all type IPMNs. CA19-9 was a significant predictor in univariate analysis, but in multivariate analysis, CA19-9 was no longer significant. When gender, age, tumor location, and cyst size were all included in the multivariable logistic regression analysis, we found that MPD diameter and mural nodule were still stable predictors for malignant IPMNs. Mural nodule was defined as any enhancing solid component protuberance within the cyst or dilated MPD. 26 In this study, mural nodule was associated with a 12.98-fold risk of malignant IPMNs.
Main pancreatic duct diameter is one critical factor for malignant IPMNs. However, the threshold value of 1.0 cm is challenged. Although several clinical guidelines on IPMNs are available, there are still many controversial issues, especially on MPD diameter regarding surgical indication. 2018 European and 2017 IAP guidelines suggested surgical resection for IPMNs with dilation of MPD exceeding 1.0 cm, while 2015 AGA and 2018 ACG guidelines recommended an MPD diameter of 0. 5 cm. Masaki et al. 27 revealed an MPD diameter of ≥ 1.2 cm to be a significant predictive factor for malignancy. However, only 29 patients with MD-IPMNs were recruited for their study. Crippa et al. 15 found that 0. 9 and 0.7 cm were the best cutoff to identify malignant IPMNs located in the head or body-tail of the pancreas, respectively. Del Chiaro et al. 16 found that a cutoff of 0.5 to 0.7 cm MPD diameter was the best predictor to discriminate between malignant and benign IPMNs. Ateeb et al 17 suggested using a cutoff between 0.6 and 0.99 cm to detect precursor lesions that are suitable for surgery. We further determined the cutoff value of MPD diameter that can distinguish high-risk IPMNs from low-risk IPMNs in our population. We found that the cutoff value of MPD diameter was 0.77 cm for all type IPMN subsets. Our data supported that the threshold value of MPD diameter should be reconsidered. Of note, most previous studies have little concern about the diagnostic values of the MPD diameter = 1 cm or MPD diameter = 0.5 cm, which was recommended by the guidelines as the surgical indication. Hence, we compared the different cutoff values of MPD diameter to assess the diagnostic efficiency. We found that using a cutoff value of 5 mm has a higher sensitivity (76.6%) but lower specificity (60.7%) compared to using a cutoff value of 0.77 cm (sensitivity 61.7%, and specificity 87.5%, respectively). This will result in unnecessary surgery for many patients who only need surveillance. The treatment for IPMNs is surgical resection and sometimes followed by complications. 3 On the other hand, using a cutoff value of 1.0 cm have a higher specificity (93.1%) but lower sensitivity (38.3%), which might decrease overall survival. Based on our cohort study, the cutoff value of 0.77 cm is a more suitable value to distinguish high-risk IPMNs from low-risk IPMNs.
Finally, we construct a regression coefficient-based nomogram to predict the high-risk IPMNs for all types of IPMN. In the last few years, several models have been proposed to overcome univariate analysis limitations. In 2020, Jeong et al. 28 developed a nomogram that contains enhancing mural nodule ≥5 mm, serum level of CA 19-9, MPD diameter, and acute pancreatitis, which could help predict the malignant potential of IPMNs (AUC 0.955). However, the results of imaging features were based on revised international guidelines (2017 IAP guidelines). In 2021, Xu et al. 29 selected six variables (mural nodule, MPD diameter, abrupt change in caliber of MPD with distal pancreatic atrophy, cyst size, thickened enhancing cyst wall, and lymphadenopathy) to construct a prediction nomogram, and the AUC was 0.92 in the training set and was 0.87 in the validation set. However, the nomogram was not validated by external centers. In 2018, Attiyeh et al. 5 presented two independently validated nomograms (main duct and branch duct) for predicting high-risk IPMNs and displayed strong objective power. However, the accurate distinguishing MD-/MT-IPMNs from BD-IPMNs before surgery is also a challenge. In this study, our nomogram contained fewer variables but performed well in the prediction of high-risk IPMNs (C-index 0.803). One critical difference between our study and previous studies is that the cut-off value of 0.77 cm for MPD diameter not 1.0 cm or 0.5 cm. Moreover, the two variables included in the nomogram were all the independent predictors, which differed from most previous studies.
Several limitations in the present study should be acknowledged. Due to its superior spatial resolution, endoscopic ultrasonography (EUS) is an effective modality for the diagnosis of pancreatic lesions.30, 31 Some studies have reported that EUS had significant advantages over MRI and CT in detecting intracyst microstructures of pancreatic cystic neoplasms, such as septa and nodules.32–35 Unfortunately, the EUS is not routinely performed in our institution. So it is noteworthy that in this study, all our data measurements come from MRI examinations only. Whether our predictive model can be applied to EUS needs further study. Secondly, this cohort selected a group of patients who only underwent surgery without follow-up. To validate this result, comparative studies with surgically treated IPMN cases after follow-up exhibiting high-risk stigma are needed. Thirdly, although IPMN is the most common PCN, its overall incidence is still low, so our cohort only included 151 patients without calculation and justification of the sample size selected. Finally, the nomogram was constructed by our single-center data and was not validated by an external agency. Further studies should focus on a multicenter cohort of patients to obtain extensive evidence-based data to verify and improve this prediction nomogram.
Conclusions
We obtained the cut-off value of MPD diameter in all type IPMNs (0.77 cm) and MPD-involved IPMNs (0.82 cm) in predicting high-risk lesions. The presence of MPD dilation ≥0.77 cm and/or existing mural nodule showed good performance in predicting high-risk IPMNs. On the other hand, careful follow-up rather than immediate surgery should be applied to patients without mural nodule and with an MPD dilation <0.77 cm. 0.77 cm may be an acceptable cut-off value for MPD diameter in predicting malignant IPMNs.
Footnotes
Acknowledgments
We will thank the patients for their great help in this report. Huifeng Zhang and Yingying Cao contributed equally to this work. Xiao Chen and Zhongqiu Wang are co-correspondence authors for this study.
Authors’ Contributions
(I) Conception and design: Huifeng Zhang, Yingying Cao, Xiao Chen; (II) Administrative support: Zhongqiu Wang; (III) Collection and assembly of data: Kai Guo, Shuai Ren, Tingting Lin; (IV) Data analysis and interpretation: Kai Guo,Yaohui Wang, Yaping Zhang; (V) Manuscript writing: Huifeng Zhang, Yingying Cao. (VI) Manuscript revising: Xiao Chen, Zhongqiu Wang.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
The conduct of the study complied with the Declaration of Helsinki and was approved by the Institutional Review Board of Affiliated Hospital of Nanjing University of Chinese Medicine (2017NL-137-05). It's a project with waived informed consent.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Natural Science Foundation of China (Grant No. 82171925, 82202135), Foundation of Excellent Young Doctor of Jiangsu Province Hospital of Chinese Medicine (Grant No. 2023QB0112), Developing Program for High-level Academic Talent in Jiangsu Hospital of Chinese Medicine (Grant No. y2021rc03), and Innovative Development Foundation of Department in Jiangsu Hospital of Chinese Medicine (Grant No. Y2021CX19).
