Abstract
Meckel’s cave or the trigeminal cistern is a subarachnoid space near the apex of the petrous portion of the temporal bone and contains cerebrospinal fluid and the Gasserian ganglion, which divides into the ophthalmic (V1), maxillary (V2), and mandibular (V3) nerves. Infectious, inflammatory, congenital, and neoplastic lesions can occur in Meckel’s cave. Leptomeningeal metastasis of glioblastoma (GBM), IDH-wildtype to Meckel’s cave is rare. We encountered a case of leptomeningeal metastasis of GBM to Meckel’s cave in an elderly female patient who presented with pain around her right eye. Magnetic resonance imaging revealed enhancing lesions in the right temporal lobe and cervical spinal cord. The pathological diagnosis of GBM was confirmed after biopsy of the cervical spinal cord lesion, which showed hyperaccumulation of fluorodeoxyglucose (FDG) on FDG-positron emission tomography. This case indicates that metastatic lesions can also occur in Meckel’s cave.
Introduction
Meckel’s cave is a subarachnoid space near the apex of the petrous portion of the temporal bone. 1 Meckel’s cave, also known as the trigeminal cistern,2–4 contains cerebrospinal fluid (CSF) and the Gasserian ganglion, which divides into the ophthalmic (V1), maxillary (V2), and mandibular (V3) nerves. 5
Meckel’s cave is mainly affected by neoplasms and inflammatory processes. The frequency of leptomeningeal metastasis to Meckel’s cave in glioblastoma (GBM), IDH-wildtype is not clear. Moreover, we could not find any reports on cases of leptomeningeal metastasis of GBM to Meckel’s cave. To the best of our knowledge, only one case of leptomeningeal metastasis of malignant glioma to Meckel’s cave has been reported. 6
Herein, we present the case of an older female patient with leptomeningeal metastasis of GBM to Meckel’s cave.
Case presentation
A 62-year-old woman presented with pain around the right eye. She had been undergoing treatment for hypertension, dyslipidemia, and chronic apical periodontitis.
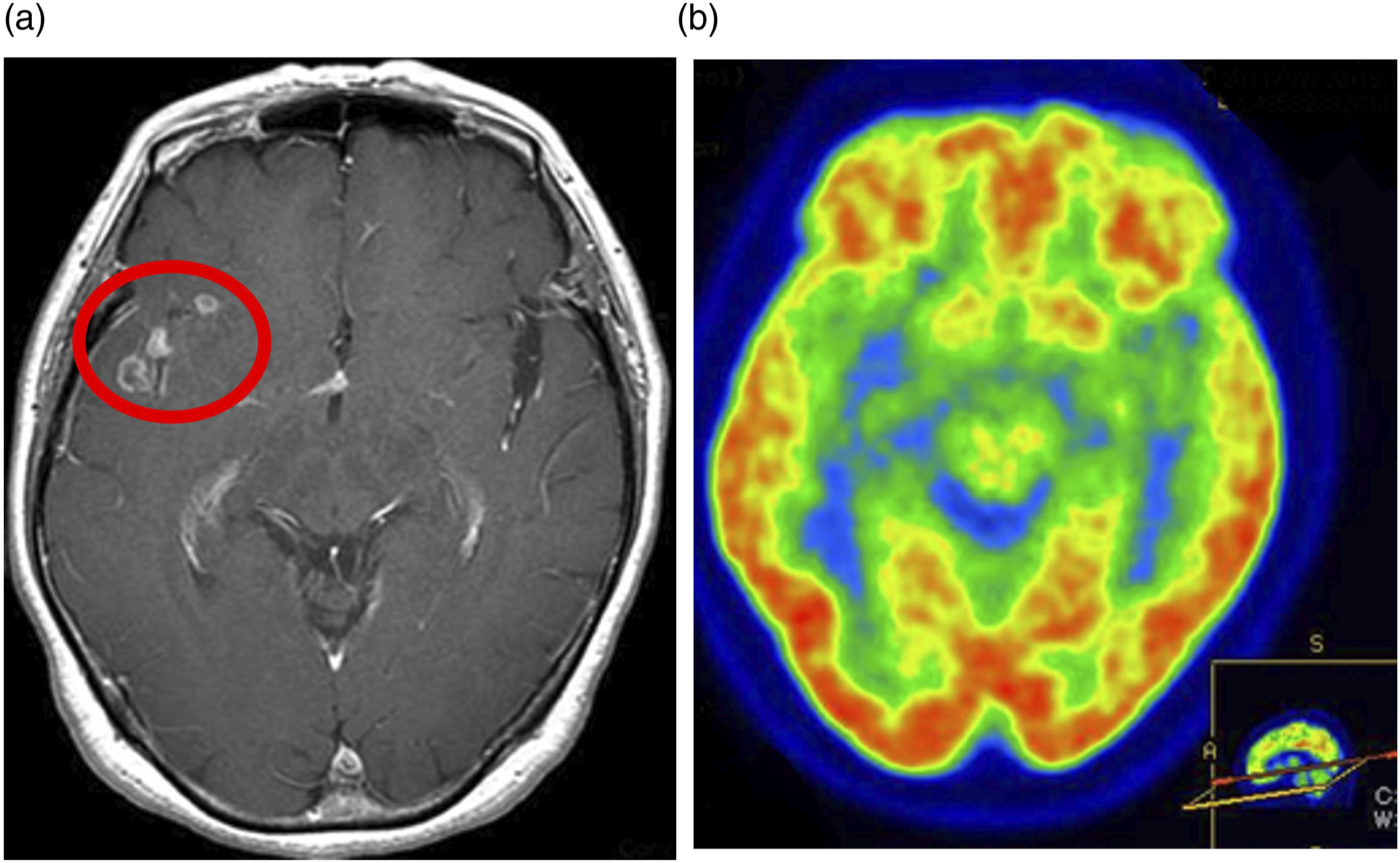
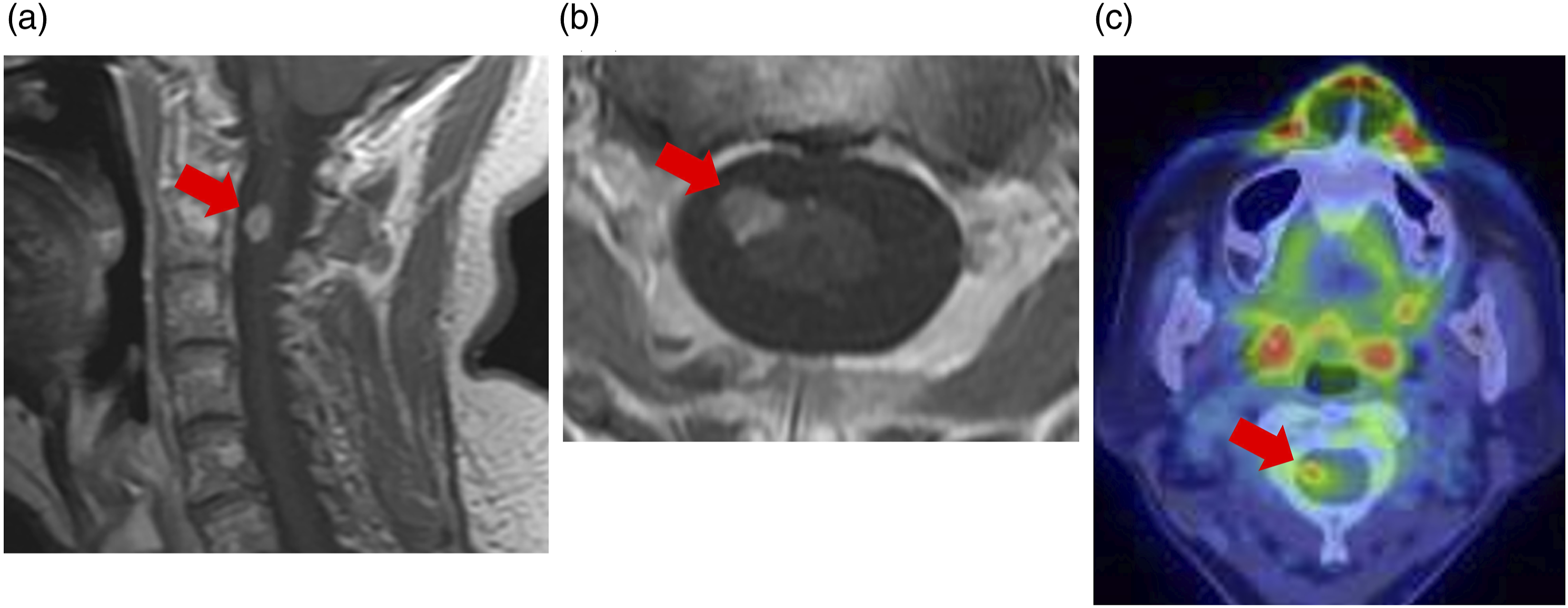
She had no relevant family history. Previously, when she visited another hospital for right eye pain, no neurological abnormalities were detected on physical examination. However, brain magnetic resonance imaging (MRI) revealed an enhancing mass lesion in the right Meckel’s cave (Figure 1) and gadolinium (Gd)-enhancing cystic lesions in the right temporal lobe and along the right Sylvian fissure (Figure 2). MRI of the spine revealed a Gd-enhancing nodule at the C2 level (Figure 3). The lesion in Meckel’s cave. (a) The axial T2-weighted image shows a mass in the right Meckel’s cave (red circle). (b) The axial T1-weighted image shows that the mass is iso-intense compared with the brain substance (red circle). (c) The axial gadolinium (Gd)-enhanced T1-weighted image shows marked Gd enhancement of the mass (red circle). (d) On fluorodeoxyglucose (FDG)-positron emission tomography, a slightly higher FDG accumulation of FDG is observed in the right Meckel’s cave (red circle) than that in the contralateral Meckel’s cave. Lesions in the right temporal lobe and right Sylvian fissure. (a) The axial gadolinium (Gd)-enhanced T1-weighted image shows Gd-enhancing cystic lesions in the right temporal lobe and along the right Sylvian fissure (red circle). (b) The fluorodeoxyglucose (FDG)-positron emission tomographic image shows no accumulation of FDG in the right temporal lobe and right Sylvian fissure. The cervical spinal cord lesion. (a) The sagittal gadolinium (Gd)-enhanced T1-weighted image reveals a Gd-enhancing nodule at the C2 level (red arrow). (b) The axial Gd-enhanced T1-weighted image reveals a Gd-enhancing nodule at the C2 level (red arrow). (c) The fluorodeoxyglucose (FDG)-positron emission tomographic image shows high accumulation of FDG in the nodule at the C2 level (red arrow).
The patient was referred to our hospital for detailed examinations and was admitted.
The patient was afebrile and had a blood pressure of 145/103 mmHg and a heart rate of 101 beats/min. Neurological examination revealed pain around the right trigeminal nerve (V1 and V2) area. No involvement of the other cranial nerves was identified. Reflexes remained intact. No weakness, sensory disturbance, and ataxia were evident. No signs of meningeal irritation were identified.
Laboratory tests, including those for blood count and blood biochemistry, revealed no abnormal findings. The levels of soluble interleukin-2 receptor and angiotensin-converting enzyme were not elevated. Blood tests for cryptococcal antigen, Aspergillus antibody, β-D-glucan, and interferon-gamma release were negative.
Cerebrospinal fluid examination revealed a white blood cell count of 3/mm3 (100% monocytes), a protein level of 32 mg/dL, a glucose level of 59 mg/dL, and negative results for cryptococcal antigen, Aspergillus-DNA, candida antigen, and bacterial culture.
FDG-PET showed high accumulation of FDG in the right Meckel’s cave lesion, but no accumulation in right temporal lobe lesions (Figure 2). In addition, high accumulation of FDG was seen in the cervical spinal cord lesion (Figure 3).
Contrast-enhanced computed tomography of the thoracic and pelvic regions showed no obvious neoplastic lesions. FDG-PET showed no abnormal accumulation of FDG in the thoracic and pelvic regions.
Based on the MRI and PET findings, we suspected malignant Meckel’s cave and cervical cord lesions as a radiological differential diagnosis.
Progress after hospitalization
Since it was difficult to confirm the diagnosis through various examinations, a biopsy of the cervical spinal cord lesion was performed.
Pale yellow tumor tissue protruding from the right anterior side of the spinal cord was observed at the level of the superior border of the C2 vertebral arch. Part of it was removed. The pathological diagnosis was GBM (WHO grade Ⅳ). On immunostaining, atypical astrocytes were glial fibrillary acidic protein (GFAP)-positive and showed high MIB-1 positivity (more than 50%) (Figure 4). In addition, intraoperative spinal fluid cytology was negative. Hematoxylin and eosin staining and immunostaining results. (a) The hematoxylin and eosin-stained section shows necrosis, microvascular proliferation, and atypical mitotic figures. (b) Atypical astrocytes are glial fibrillary acidic protein (GFAP)-positive. (c) MIB-1 positivity is high (more than 50%).
Based on these results, the patient was started on temozolomide and radiotherapy.
Temozolomide was administered at 100 mg/day for 42 days, followed by a 28-day rest period, and then 220 mg/day for 6 days. Radiotherapy was administered: 30 Gy to the whole brain, 30 Gy to the right temporal lobe and focal lesions in Meckel’s cave, and 50 Gy to the cervical spinal cord. However, the lesions increased in size and number after 3 months of starting treatment (Figure 5). A mass was also observed in the contralateral Meckel’s cave (Figure 5), and the cervical cord lesion was found to be enlarged. Imaging results after initiating treatment. (a) The axial gadolinium (Gd)-enhanced T1-weighted image shows a marked increase in the size of temporal lobe lesions (red circle) and (b) The axial Gd-enhanced T1-weighted image shows an enlargement of the lesion in the right Meckel’s cave (red arrow), and a mass is seen in the contralateral Meckel’s cave as well (red circle).
Five months after being referred to our hospital, the patient was transferred to another hospital for transition to best supportive care. She was confirmed dead about 6 months later.
Written informed consent was obtained from the patient for publication of this case report.
Discussion
Effacement or abnormal enhancement of Meckel’s cave might suggest infectious, inflammatory, congenital, or neoplastic lesions. 7 In the present case, the lesion in Meckel’s cave was a leptomeningeal metastatic lesion of GBM.
Metastasis to Meckel’s cave can be caused by hematogenous dissemination from head and neck tumors, according to articles describing leptomeningeal metastasis to Meckel’s cave. 1 Because the CNS lacks lymphatic vessels, distant metastases generally reach the CNS by the hematogenous route. The blood–brain barrier (BBB) cannot prevent metastatic cells from entering the CNS. 1 Several mechanisms beyond the BBB, including the destruction of endothelial cells by tumor cells, which causes increased vascular permeability, and solitary metastatic masses causing relative ischemia that leads to increased vascular permeability, have been proposed.8,9
In the present case, leptomeningeal metastasis occurred not only in the right Meckel’s cave but also in the contralateral Meckel’s cave. This might be related to the fact that Meckel’s cave is a blind end structure.
It was reported that spinal leptomeningeal metastasis was infrequent in the early stage of GBM.10,11 In previously reported cases of spinal leptomeningeal metastasis, symptoms were unlikely to occur in patients. It is speculated that the reason for the lack of symptoms is that the tumor cells infiltrate between nerves rather than causing the destruction of nerves. 12
Dissemination to the CSF is estimated to occur in 15–25% of patients with supratentorial GBM and 60% of patients with infratentorial GBM.12–14 However, CSF cytology has poor sensitivity for detecting malignant cells and diagnosing metastases.15–18
MRI has been reported to be useful for diagnosing spinal leptomeningeal metastasis. 10 MRI was useful for diagnosing spinal leptomeningeal metastasis in the present patient as well.
It is difficult to determine whether a lesion in Meckel’s cave is malignant or benign based on clinical symptoms and radiological findings. 1 Differential diagnosis of a lesion in Meckel’s cave includes meningioma, schwannoma, leptomeningeal metastasis, malignant lymphoma, sarcoidosis, herpes simplex virus infection, chronic inflammatory demyelinating polyneuropathy, Tolosa-Hunt syndrome, and pituitary macroadenoma. Skull base imaging is most frequently performed for differential diagnosis of lesions in Meckel’s cave. Key differentiators on imaging include loss of CSF signal in Meckel’s cave, moderate enhancement over perineural vessels, nerve enlargement, infiltration of the nerve, and extension into the bony foramen. 7 In FDG-PET for glioma, hyperaccumulation of FDG reflects tumor cell proliferation in anaplastic types. 19 However, GBM may have necrosis, in which case, there is a decrease in the accumulation of FDG. 20 In the present case, the lack of accumulation of FDG in right temporal lobe lesions may have been due to necrosis. On the other hand, FDG-PET has been reported to be effective in detecting spinal leptomeningeal metastasis.21,22 In the present case, the cervical cord lesion diagnosed pathologically as GBM also revealed hyperaccumulation of FDG on FDG-PET.
In conclusion, we experienced a case of GBM with leptomeningeal metastasis to Meckel’s cave.
The case indicates that malignant lesions can occur in Meckel’s cave, and therefore, it is necessary to include them in the differential diagnoses.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This paper was prepared in accordance with the ethical standards of the institutional ethics committee and with the 1964 Helsinki Declaration. Our institution does not require ethical approval for reporting individual cases.
Informed consent
Written informed consent was obtained from the patient for publication of this case report.
