Abstract
Background
Cone-beam computed tomography (CBCT) has been widely used during transcatheter arterial chemoembolization for hepatocellular carcinoma.
Purpose
To evaluate the sensitivity of CBCT for the detection of hepatocellular carcinomas and the tumor feeders by comparing celiac artery (CA) and common hepatic artery (CHA) injection.
Material and methods
We retrospectively enrolled 30 patients (52 hepatocellular carcinoma lesions) who had undergone CBCT-assisted transcatheter arterial chemoembolization. In 17 procedures (28 hepatocellular carcinomas) we acquired CBCT scans using CA injections (CBCT-CA) and in 18 (24 hepatocellular carcinomas) we used CHA injections (CBCT-CHA). Of the 30 patients, 5 underwent CBCT-CA and CBCT-CHA at different transcatheter arterial chemoembolization procedures. We performed inter-group comparisons of the detectability of hepatocellular carcinoma, the feeding artery, the intrahepatic artery branch order, and the tumor-to-liver contrast.
Results
CBCT-CA detected all 28 hepatocellular carcinomas and 27 of their feeders (96.4%); CBCT-CHA identified 22 of 24 hepatocellular carcinomas (91.7%) and 21 of their feeders (95.5%). There was no significant inter-group difference in the detectability of hepatocellular carcinoma lesions (p = 0.21) or feeding arteries (p = 0.69). CBCT-CHA was superior for the assessment of the tumor-to-liver contrast and the intrahepatic artery branch order (both: p < 0.01).
Conclusion
CBCT-CA and CBCT-CHA were equally useful for the detection of hepatocellular carcinoma and of the feeding artery, although CBCT-CHA yields better visualization of hepatocellular carcinoma and the hepatic artery. Thus CA injection seems sufficient for lesion and vessel detection when the insertion of an angiographic catheter into the CHA is difficult.
Keywords
Introduction
Transcatheter arterial chemoembolization (TACE) of the liver is one of the most common treatments for hepatocellular carcinoma (HCC), a hypervascular malignant liver tumor. Tumor detection and assessment of the tumor-feeding vessel(s) are important for an effective treatment, while limiting non-target embolization. 1
HCCs not readily apparent on digital subtraction angiograms (DSA) may be detectable on C-arm-mounted cone-beam computed tomography (CBCT) scans whose spatial and contrast resolution is superior to DSA. 2 –5 CBCT yields accurate, real-time information on target lesions.3,5 Although the signal-to-noise ratio (SNR) of CBCT is lower than that of multidetector CT (MDCT), improvements in the imaging capabilities and shortening of the reconstruction time have led to the utilization of CBCT during TACE. Common hepatic artery (CHA) or proper hepatic artery injection has usually been used for dual-phase CBCT during hepatic angiography (CBCT-HA).6,7 Before the availability of gadolinium ethoxybenzyl diethylenetriamine pentaacetic acid (Gd-EOB-DTPA), the acquisition of CT scans during hepatic angiography (CT-HA) and during arterial portography (CT-AP) was considered a reliable, highly sensitive imaging method for the detection of HCC. Corona enhancement at dual-phase CT-HA is an essential finding for distinguishing between HCCs and arterioportal shunts. 8 Tada et al. 9 demonstrated that the sensitivity and specificity of Gd-EOB-DTPA-enhanced magnetic resonance imaging (MRI) was equal or superior to combination of dual-phase CT-HA and CT-AP. Consequently, diagnostic dual-phase CT-HA has become less important.
CHA injection for dual-phase CT-HA or CBCT-HA requires the insertion of a conventional angiographic catheter or a microcatheter into the CHA. The former can be technically demanding; however, the risk of iatrogenic artery injury is pretty low. The latter may yield insufficient contrast due to a low flow rate of contrast medium especially when a thin microcatheter suitable for ultra-selective TACE is inserted. The flow rate in high-flow microcatheters is adequate; however, they are not suitable for ultra-selective TACE because their use decreases selectivity.
Because toxicity to adjacent structures and non-target TACE should be avoided, obtaining single-phase CT-HA or CBCT-HA images is beneficial.3,5,10–13 An earlier systematic review and meta-analysis of the role of CBCT in patients with HCC treated by TACE suggests that CBCT can significantly increase detection of tumors and tumor-feeding arteries. 14 Single-phase CBCT scans can be acquired by celiac artery (CA) injection (CBCT-CA) when they are performed before the contrast medium returns from the splenic to the portal vein. CA is simpler than CHA injection (CBCT-CHA) because the latter requires the insertion of an angiographic catheter or a microcatheter into the CHA. As the detectability of HCC and of the feeders on CBCT-CA images remained to be ascertained, we compared CBCT-CA and CBCT-CHA images acquired before TACE in patients with HCC.
Material and Methods
Study design
This retrospective study was approved by our institutional review board; prior informed patient consent was waived. Between April 2017 and March 2018, we performed 116 TACE procedures in patients diagnosed HCC.
Included for analysis were patients who had CBCT during TACE and dynamic contrast-enhanced CT and/or Gd-EOB-DTPA-enhanced MRI two months before TACE. Exclusion criteria were poor breath holds, status post-lobectomy, and status post-arterial redistribution for percutaneous port catheter placement. Also excluded were patients who had undergone another TACE 10 days earlier, and patients with hepatic arterial anomalies such as a replaced right hepatic artery (RHA) from the superior mesenteric artery, a replaced left hepatic artery (LHA) from the left gastric artery, and patients whose RHA arose from the CA. Small tumors less than 10 mm were also excluded. 15 Based on these criteria, our retrospective study included 30 patients with 52 HCCs. We reviewed their pre-TACE angiography, CBCT, CT, and/or MRI findings. Of the 30 patients, 5 underwent CBCT-CA and CBCT-CHA at different TACE procedures. In 17 procedures (28 HCCs) we acquired CBCT scans using CA injections (CBCT-CA, group 1) and in 18 (24 HCCs) we used CHA injections (CBCT-CHA, group 2). Table 1 shows the characteristics of the patients and their hepatic tumors.
Characteristics of the 30 patients and their tumors.
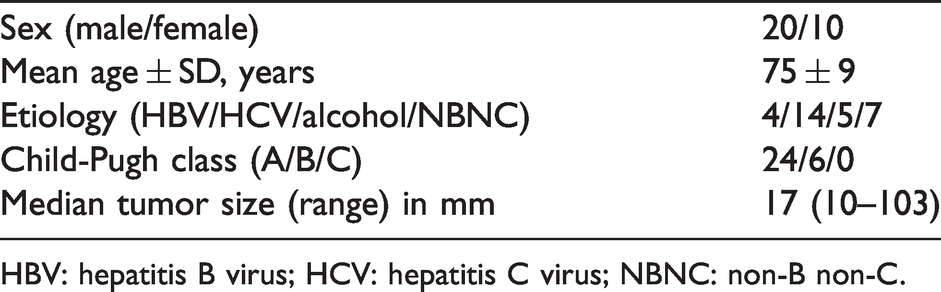
HBV: hepatitis B virus; HCV: hepatitis C virus; NBNC: non-B non-C.
Imaging technique
Before TACE, DSA of the celiac trunk was performed. The contrast medium (20 ml, 350 mgI/ml iohexol (Omnipaque)) was injected at a rate of 4 ml/s through a 4F catheter.
CBCT images (XperCT; Philips Medical Systems) were acquired on an angiography unit (Allula Xper FD 20/20; Philips Medical Systems, Best, The Netherlands). The operator chose CA or CHA injection. The contrast medium (30 ml, 350 mgI/ml iohexol (Omnipaque)) was injected at a rate of 2 ml/s through a 4F catheter; scanning was started 8 s after the start of contrast medium injection. During a single breath hold, we obtained 312 projection images. The x-ray acquisition parameters were 117 to 123 kVp, with an acquisition time of 5.2 s, 240° rotation around the patient.
Image analysis
Dynamic contrast-enhanced CT or MRI scans performed before TACE represented the gold standard for evaluating the detectability of HCC on DSA and CBCT images. The HCC diagnosis was based on arterial contrast enhancement and contrast medium washout in the equilibrium phase on CT scans, on arterial contrast enhancement and hypointensity in the hepatobiliary phase on MRI scans, 16 and on a correspondence between the arterial contrast enhancement on CBCT and CT or MRI scans. In patients with multiple HCCs, the three largest tumors were assessed.
A radiologist with eight years of experience recorded the detectability of the feeding artery, defined as the artery adjacent to the HCC on CBCT images (Fig. 1(b)). To rate HCC visualization, the tumor-to-liver contrast (TLC) was calculated;
17
circular regions of interest (ROI) with a minimum size of 0.5 cm2 were placed and the mean attenuation value (in Hounsfield units, HU) in the most strongly enhanced portion of the HCC (ROItumor) and the surrounding liver parenchyma (ROIliver) was recorded. Special care was taken to avoid placing the ROI in areas with pronounced artifacts or within vessels. The TLC was calculated for each HCC using the formula
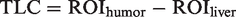

(a) Arterial-phase contrast-enhanced computed tomography (CT) demonstrated hepatocellular carcinoma at segment 5 of the liver (arrow). (b) Cone-beam CT also revealed hepatocellular carcinoma (arrow). A feeding artery adjacent to the tumor was visualized (arrowhead).
To evaluate visualization of small intrahepatic artery branches, the highest branch-order of the intrahepatic artery was counted by a radiologist with eight years of experience. The LHA and RHA are primary branches, the anterior and posterior branches of the RHA are second-order branches, and the segmental branches of the RHA are third-order branches. The presence of fourth-order or higher order branches was considered to represent a bifurcation between vessels of similar diameter.
Statistical analysis
The Chi-square test was used to determine inter-group differences in HCC detectability on DSA images, Fisher’s exact test to assess differences in HCC and feeding artery detectability on CBCT-CA and CBCT-CHA images, and the Welch t-test to determine differences in the TLC. To determine differences in the branch-order number on CBCT scans, we applied the Mann-Whitney U-test. All data were analyzed with statistical software (Statcel4; OMS, Tokyo, Japan).
Results
Of the 28 HCCs in group 1 (CA injection), 20 (71.4%) were detected by DSA, as were 17 of the 24 HCCs in group 2 (CHA injection, 70.8%). All 28 lesions in group 1 were observable on CBCT-CA images as were the feeders of 27 of these HCCs (96.4%). CBCT-CHA identified 22 of the 24 group 2 HCCs (91.7%) and 21 feeders (95.5%). As shown in Table 2, there was no significant inter-group difference with respect to the visualization of HCCs or their feeders.
Difference between digital subtraction angiography- and cone-beam computed tomography images in the detection of hepatocellular carcinoma and the feeder.

CA: celiac artery; CHA: common hepatic artery; CBCT: cone-beam computed tomography; HCC: hepatocellular carcinoma; DSA: digital subtraction angiograms.
The TLC was significantly higher on group 2 than group 1 CBCT images (p < 0.01, Table 3 and Fig. 2(a) and (b)). It was recorded a significantly greater number of branch orders on group 2 than group 1 CBCT images (p < 0.01, Table 3 and Fig. 3(a) and (b)).
Tumor-to-liver contrast and number of intrahepatic artery branches identified on cone-beam computed tomography scans performed during angiography.

CA: celiac artery; CHA: common hepatic artery; TLC: tumor-to-liver contrast.

(a) CBCT-CA image showing a 10-mm diameter hypervascular HCC (arrow). The tumor-to-liver contrast (TLC) was 311 HU. (b) CBCT-CHA image showing a 17-mm diameter hypervascular HCC (arrow). The TLC was 717 HU.

(a) CBCT-CA (maximum intensity projection) showing sixth-order branches of the hepatic artery. (b) CBCT-CHA (maximum intensity projection) showing seventh-order branches of the hepatic artery.
Discussion
CBCT-CA detected all 28 HCCs and 27 of their feeders. On CBCT-CHA images, 22 of 24 HCCs and 21 of their feeders were visualized. There was no significant inter-group difference in the detectability of HCCs and their feeders. The rate of HCC detection on CBCT images was similar to that reported in an earlier systematic review and meta-analysis of the role of CBCT in patients with HCC treated by TACE. 14
Our observations suggest that CA and CHA injections yield similar CBCT findings. CA injection is sufficient for lesion and vessel detection when the insertion of a guiding catheter into the CHA is difficult. CA injection is simple and, different from CHA injection, on CBCT-CA images, a potential benefit is that extrahepatic feeders such as the omental, gastric, and the inferior phrenic artery arising from the CA are observable.
Post-CA injection, 71.4% of HCCs were observable on DSA images; 70.8% were identified after CHA injection. Our findings are in accordance with those reported by Pung et al. 14 There was no significant difference between group 1 (CA) and group 2 (CHA) with respect to the detectability of HCCs on DSA images.
On the other hand, the TLC was stronger and the order number of visualized branches was greater after CBCT-CHA, suggesting a better visualization of the HCC and of small branches of the intrahepatic artery. As some HCCs are fed by two or more arteries, 18 all feeders must be identified before ultra-selective TACE and CBCT-CHA studies are necessary. CBCT data can be used for 3D-vessel tracking by software that identifies the tumor feeders automatically.11,19 However, the overlay of the portal venous system may hamper their detection by the software, following CBCT-CA.
Referring to earlier studies,3,5,7,19 our injection rate for CBCT-CHA was 2 ml/s; it was the same for CBCT-CA. As the detection of HCCs and their feeders was similar on CBCT-CA and CBCT-CHA images, the delivery of contrast medium at the rate of 2 ml/s appears to be appropriate although Meyer et al. 4 performed CBCT-CA at an injection rate of 3 ml/s. Studies are underway to identify the optimal injection rate for the acquisition of diagnostic CBCT-CA images.
Our study has some limitations. Our sample size was small, the study was non-randomized, retrospective, and it included patients seen at a single center. As the injection site was unilaterally chosen by the operator, we cannot deny selection bias. Lastly, we cannot deny the possible presence of non-visible feeding arteries in patients where a single feeder was identified.
In conclusion, as on CBCT-CHA images, HCCs and their arterial feeders were identifiable on CBCT-CA images, although smaller branches of the intrahepatic artery are more readily identifiable on CBCT-CHA images. Thus, CBCT-CA imaging seems sufficient for lesion and vessel detection when the insertion of an angiographic catheter into the CHA is difficult. A controlled prospective study is needed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Kazuo Awai received research grants from Canon Medical Systems, Hitachi, Fujitsu, Nemoto-Kyorindo, Eizai, Daiichi-Sankyo, Fuji Seiyaku Kogyo, and Fuji Film Toayama (research grants were paid to the institution). He is also a board member of the Japanese Radiological Society. Any other competing interests, such as employment, consultancy, patents, products in development, or marketed products, do not exist regarding this manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
