Abstract
The cerebral metabolism, such as the oxygen extraction fraction (OEF), in remote ischemic lesions following revascularization for moyamoya disease (MMD) has not yet been fully elucidated. We herein report a patient with an increased OEF in a remote ischemic lesion after revascularization in a case of adult-onset MMD. A 21-year-old woman suffered from a left parietal lobe infarction due to MMD. At 2 months after onset, left superficial temporal artery (STA)–middle cerebral artery (MCA) bypass and encephalo-myo-synangiosis (EMS) were performed. The postoperative course was uneventful. 15O-positron emission tomography (PET) performed at 2 months after the first operation revealed an increased OEF in the contralateral (right) frontal lobe that was suspected of being possible remote ischemia. The patient underwent right STA-MCA bypass and EMS. 15O-PET at 14 days after the second operation revealed an increased OEF in the contralateral (left) occipital lobe that was suspected of potentially being remote ischemia caused by a watershed shift. Two years after the second surgery, left occipital artery (OA)–posterior cerebral artery (PCA) anastomosis and EMS were performed due to transient right hemianopsia. Neither rebleeding nor ischemic complications occurred 2 years after the third surgery. We need to be alert for the possible progression of PCA stenosis in MMD after revascularization. It might induce remote ischemia after revascularization. OA–PCA bypass is therefore considered to be an effective treatment option in such cases.
Introduction
Moyamoya disease (MMD) is characterized by chronic steno-occlusion of the distal part of the internal carotid arteries (ICAs) and the proximal anterior and middle cerebral artery (MCA) bilaterally. 1 Collateral vessels develop to compensate for ischemia, including basal moyamoya vessels originating from the distal end of the ICAs and transdural anastomoses from the external carotid arteries. 2 Direct or indirect revascularization is considered the most effective management for MMD in order to re-establish the cerebral blood flow and improve cerebral ischemia. 3 Remote ischemia at the contralateral hemisphere was also reported following revascularization for MMD. 4 In addition, a few previous reports have mentioned a correlation between posterior cerebral artery (PCA) stenosis and remote ischemia.5,6 However, the cerebral metabolism, such as the oxygen extraction fraction (OEF), in the remote ischemic lesion has never been fully elucidated.
We herein report a patient with an increased OEF in a remote ischemic lesion after revascularization in a case of adult-onset MMD who underwent occipital artery OA-PCA bypass.
Case description
A 21-year-old woman suffered from left parietal lobe infarction with mild sensory aphasia, right hemispatial neglect, and incomplete Gerstmann syndrome. Magnetic resonance imaging (MRI) fluid-attenuated inversion recovery (FLAIR) at admission revealed cerebral infarction in the left temporoparietal lobe (Fig. 1(a)). Magnetic resonance angiography (MRA) showed bilateral terminal ICA and MCA steno-occlusive changes and mild stenosis of the left PCA (Fig. 1(b)). MRI T1-weighted imaging (WI) showed a large flow void, indicating moyamoya vessels in the basal ganglia (Fig. 1(c)). She also had the genetic variant RNF213 c.14576G>A. Based on the above findings, we diagnosed her with MMD. (a) Magnetic resonance imaging fluid-attenuated inversion recovery at admission revealed cerebral infarction in the left temporoparietal lobe. (b) Magnetic resonance angiography showed bilateral terminal internal carotid arteries and middle cerebral artery steno-occlusive changes and mild stenosis of the left posterior cerebral artery. (c) Magnetic resonance imaging T1WI showed a large flow void indicating moyamoya vessels in the basal ganglia.
15O-positron emission tomography (PET) revealed a low CBF, increased CBV, and low CBF/CBV, indicating a low perfusion pressure in the left frontal lobe (Fig. 2(a)). At 2 months after onset, she had recovered except for mild symptoms of incomplete Gerstmann syndrome. Left superficial temporal artery (STA)–MCA bypass and encephalo-myo-synangiosis (EMS) were performed. The postoperative course was uneventful. 15O-PET findings: (a) At the first admission, (b) 2 months after the first operation, (c) 14 days after the second operation, (d) 2 years after the second surgery, E: 14 days after the third surgery. CBF: cerebral blood flow, CBV: cerebral blood volume. CMRO2: cerebral metabolic rate of oxygen, OEF: oxygen extraction fraction. (a) We noted a low CBF and CMRO2, increased CBV, and low CBF/CBV indicating a low perfusion pressure in the left frontal lobe. (b) An increased OEF was detected in the contralateral (right) frontal lobe. (c) An increased OEF was detected in the contralateral (left) occipital lobe. (d) A low CBF, mildly increased OEF, and increased CBV were observed in the left PCA territory. (e) No obvious ischemic lesions were observed, except for those in the left parietal lobe.
15O-PET performed 2 months after the first operation revealed an increased OEF in the contralateral (right) frontal lobe that was suspected of potentially being remote ischemia (Fig. 2(b)). Fortunately, MRI FLAIR showed no ischemic lesions in the right frontal lobe (Fig. 3(a)). She then underwent right STA–MCA bypass and EMS. Although the postoperative course was again uneventful, with no new neurologic deficit, 15O-PET performed 14 days after the second operation revealed an increased OEF in the contralateral (left) occipital lobe, which was suspected of potentially being remote ischemia that might have been caused by a watershed shift (Fig. 2(c)). Furthermore, MRA indicated a progression of left P1 stenosis 28 days after the second operation (Fig. 3(b)). Since she had no new symptoms, we decided to perform careful observation this time instead of surgical revascularization, such as with OA–PCA bypass. (a) Magnetic resonance imaging fluid-attenuated inversion recovery showed no ischemic lesions in the right frontal lobe. (b) Magnetic resonance angiography indicated a progression of left P1 stenosis at 28 days after the second operation. (c) Magnetic resonance imaging fluid-attenuated inversion recovery did not show any new ischemic lesions. (d) Magnetic resonance angiography showed left P1 occlusion.
She was admitted to our hospital again due to an occipital headache and transient right hemianopsia 2 years after the second surgery. Although MRA showed left P1 occlusion (Fig. 3(d)), MRI FLAIR did not show any new ischemic lesions (Fig. 3(c)). 15O-PET showed a low CBF, mild increase in the OEF, and increased CBV in the left PCA territory (Fig. 2(d)). We ultimately decided to perform surgical revascularization.
Left OA–PCA anastomosis and EMS were performed. Three-dimensional computed tomography angiography of the status after three surgical revascularization procedures showed that the bilateral STAs and left OA were patent (Figs. 4(a)–(c)). MRI diffusion-weighted imaging (DWI) and FLAIR did not show any new ischemic lesions (Figs. 4(d) and (e)). Similarly, MRA showed left PCA occlusion, and the bilateral STAs and left OA were patent (Fig. 4(f)). The final 15O-PET images indicated no obvious ischemic lesions, except in the left parietal lobe, at 14 days after the third surgery (Fig. 2(e)). She was discharged home with modified Rankin Scale (mRS) 22 days after the third surgery. Neither rebleeding nor ischemic complications occurred for 2 years after the third surgery. (a) Right lateral view, (b) posterior view, and (c) left lateral view. Three-dimensional computed tomography angiography performed after three surgical revascularization procedures showed that the bilateral superficial temporal arterys and left occipital artery were patent. (d) Magnetic resonance imaging diffusion-weighted imaging and (e) fluid-attenuated inversion recovery did not show any new ischemic lesion. (f) Magnetic resonance angiography showed left posterior cerebral artery occlusion, and the bilateral superficial temporal arteries and left occipital artery were patent.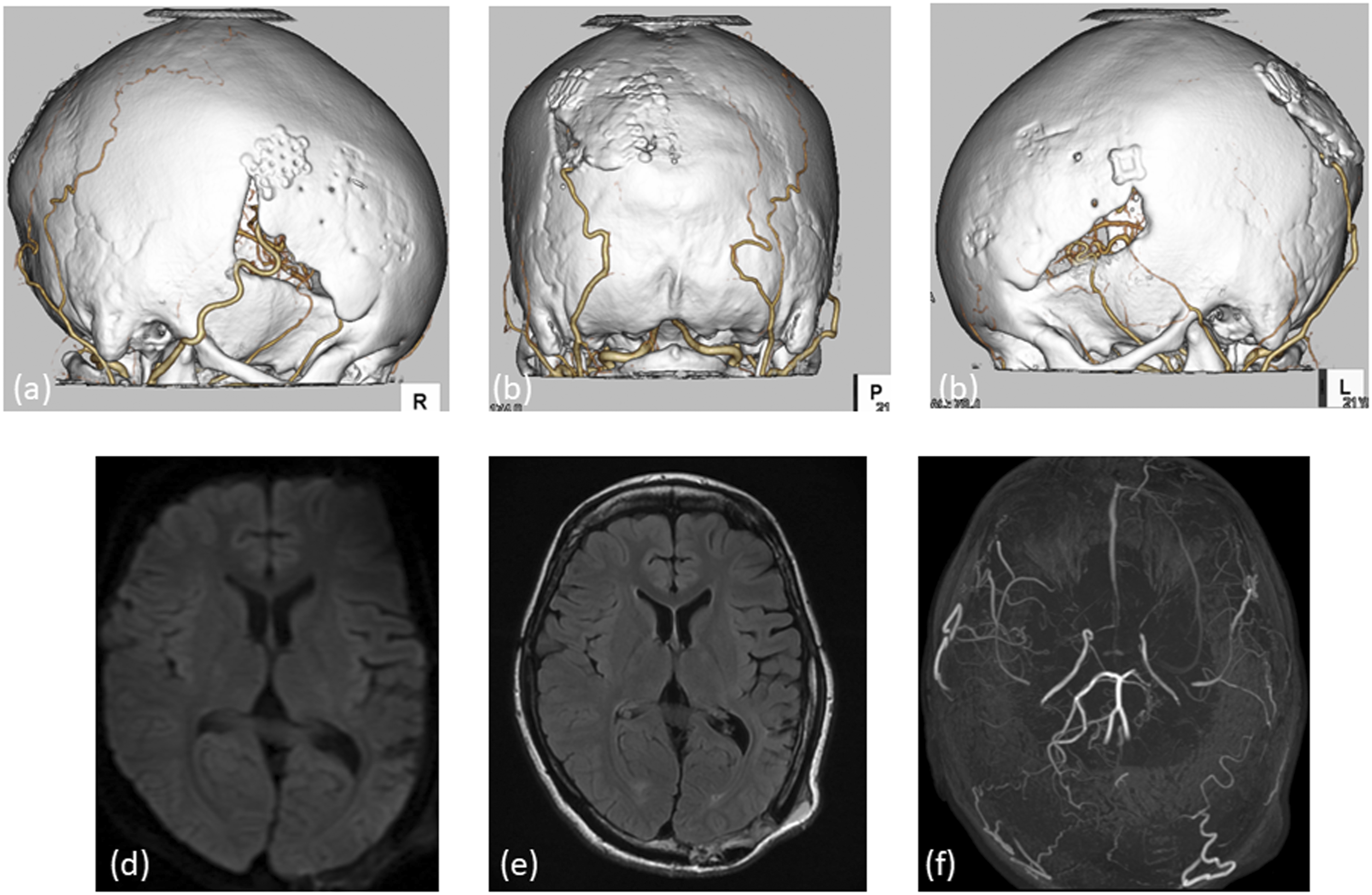
Discussion
Although remote ischemia or a watershed shift4,7 have been detected as a CBF reduction by single-photon–emission computed tomography, no reports have previously described an increased OEF.
In the present case, an increased OEF was observed in the contralateral (right) frontal lobe 2 months after the first operation and it was suspected of potentially being remote ischemia. This phenomenon was also observed in the posterior circulation following a progression of PCA stenosis. Thus, our case suggests that the progression of PCA stenosis can influence remote ischemia. Although a few previous reports have mentioned a correlation between PCA stenosis and remote ischemia,5,6 the mechanisms underlying remote ischemia remain controversial. 4 Based on the present case, remote ischemia seems to cause an increase in the OEF. However, whether PCA stenosis preceded it or it was induced by changes in the perfusion pressure due to a watershed shift to promote PCA stenosis is unclear. Whether or not the watershed shift associated with revascularization is a trigger for PCA changes should therefore be explored in future studies.
Recent studies have emphasized the involvement of the PCA in MMD as an important factor related to a poor prognosis.8,9 The efficacy of posterior circulation revascularization surgery, including OA–PCA bypass, has also been reported. 10 We need to closely monitor the progression of PCA stenosis even after revascularization, for MMD might induce remote ischemia following such a procedure. In such cases, OA–PCA bypass seems to be an effective treatment option.
In conclusion, we herein reported a patient with an increased OEF in a remote ischemic lesion after revascularization in a case of adult-onset MMD. Whether PCA stenosis preceded it or it was induced by changes in the perfusion pressure due to a watershed shift to promote PCA stenosis is unclear. Whether or not the watershed shift associated with revascularization is a trigger for PCA changes should be explored in future studies. We need to closely monitor the progression of PCA stenosis even after revascularization for MMD since remote ischemia may sometimes occur following this procedure. In such cases, OA–PCA bypass seems to be an effective treatment option.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
