Abstract
IgG4-related disease was originally discovered in patients with autoimmune pancreatitis accompanied by elevated serum IgG4 levels and has subsequently been described in almost every organ system. IgG4-related disease presents with a variety of symptoms according to the organ affected and may be accompanied by serious complications such as organ dysfunction associated with IgG4-positive cell proliferation. We report a case of IgG4-related abdominal disease in a patient who also had involvement of the coronary artery.
Introduction
IgG4-related disease (IgG4RD) was originally discovered in patients with autoimmune pancreatitis accompanied by elevated serum IgG4 levels1,2 and has subsequently been described in almost every organ system.1–4 IgG4-related cardiovascular disease can affect the coronary arteries, heart valves, myocardium, pericardium, aorta, and peripheral vasculature. 4 IgG4RD is characterized by diffuse lymphoplasmacytic infiltrates and the presence of abundant IgG4-positive plasma cells within extensive fibrosis, frequently associated with tumorous swelling and elevated serum IgG4 concentrations.1,2 IgG4RD presents with a variety of symptoms according to the organ affected and may be accompanied by serious complications such as organ dysfunction associated with IgG4-positive cell proliferation. 4 We report the case of a 50-year-old man with IgG4RD involving abdominal and extra-abdominal organs. The diagnosis was not considered for about 10 years after the initial presentation until an excisional biopsy of the right mandibular gland was positive for IgG4.
Case report
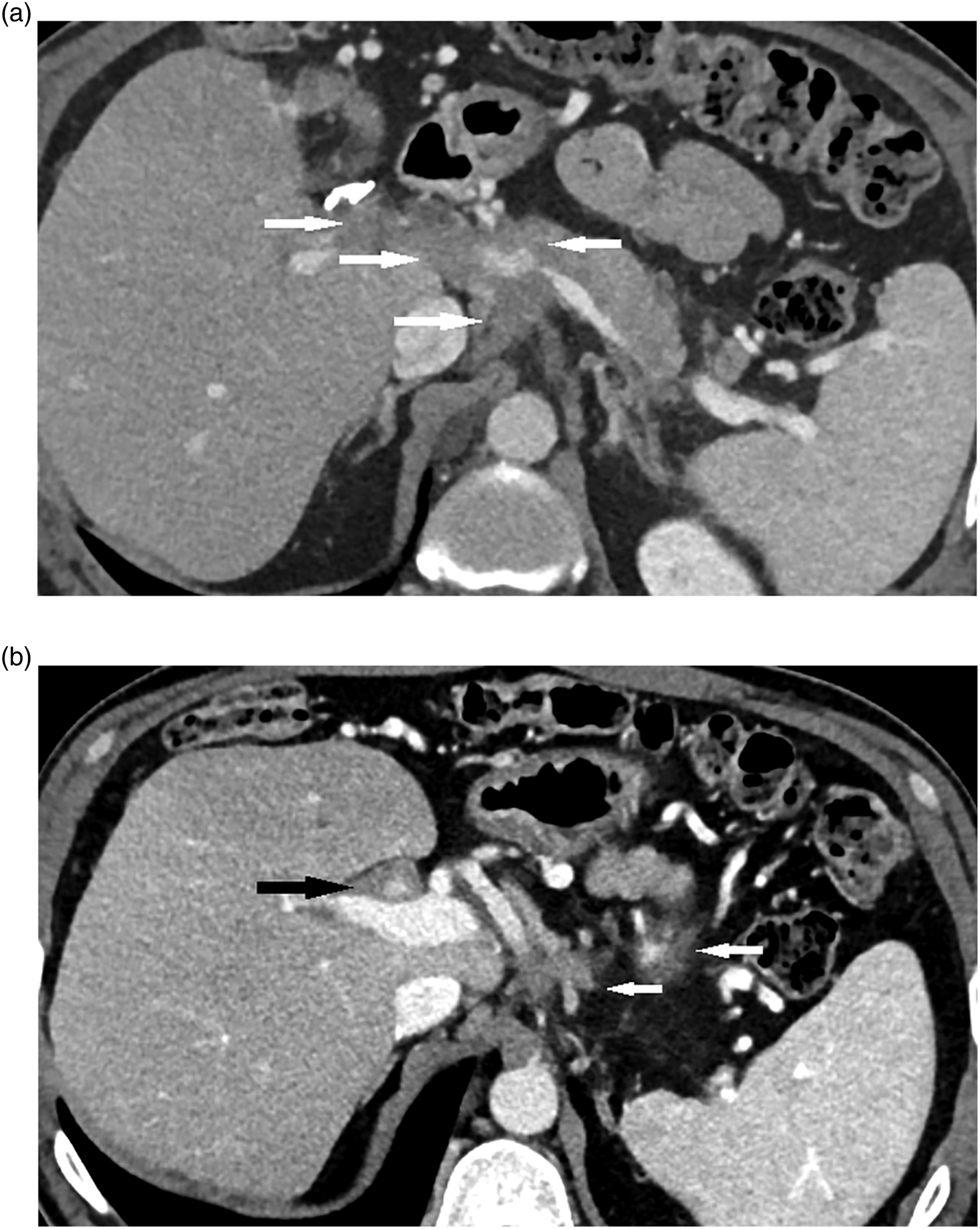
A 52-year-old man visited our institution for evaluation of possible IgG4-related disease. He had ongoing symptoms for approximately 10 years, including Raynaud’s phenomenon, recurrent diverticulitis, and lymphadenopathy. An axillary lymph node resection was performed, with inconclusive pathology. He also had chronic right upper-quadrant abdominal pain and underwent cholecystectomy, also with inconclusive pathology, but without evidence of infection or malignancy. A PET-CT did not show convincing evidence of malignancy. He had right mandibular gland swelling, which was excised after unsuccessful FNA. Pathology demonstrated a mass-like region of dense fibrosis with marked inflammation including lymphoid follicles with terminal centers with numerous plasma cells and scattered eosinophils. Additional immunohistochemical staining revealed greater than 100 plasma cells per HPF staining positive for IgG4. The gallbladder specimen was then re-stained and also demonstrated high IgG4 levels. The patient had continued symptoms, including shortness of breath and wheezing on exertion as well as ongoing abdominal discomfort. He therefore had evaluation for involvement of other organs by IgG4-related disease. A thoracic and coronary CTA showed focal soft tissue encasement of the proximal LAD with mild associated luminal narrowing (Figure 1). Abdominal–pelvic CT showed soft tissue thickening involving the retroperitoneum and porta hepatis encasing multiple vascular structures with moderate narrowing of the superior mesenteric and splenic veins (Figure 2). There was mild intrahepatic biliary ductal dilatation, with infiltrative soft tissue in the peribiliary region encasing hepatic artery branches. IgG4 was elevated to 1170 mg/dl (reference range: 2.4–121 mg/dl). He started rituximab treatment infusion and had symptomatic relief. Short axis reformatted images from coronary CT angiogram shows soft tissue encasing the mid left anterior descending coronary artery with mild luminal narrowing (arrow). Post-contrast portal venous phase axial images from abdominal CT show soft tissue encasing and narrowing of the splenic vein and main portal vein in the region of the portal confluence and extending to the porta hepatis (a and b, white arrows), with additional circumferential soft tissue encasement of the hepatic artery in the porta hepatis (b, black arrow).
Discussion
IgG4RD was originally discovered in patients with autoimmune pancreatitis accompanied by elevated serum IgG4 levels1,2 and it has been described in almost every organ system including the pancreas, bile ducts, lacrimal glands, salivary glands, central nervous system, thyroid, lung, liver, gastrointestinal tract, kidney, prostate, retroperitoneum, cardiovascular system, lymph nodes, skin, and breast.1–4 IgG4-RD affecting the heart is relatively rare, with most reported cases involving the coronary arteries followed by pericardial, aortic, myocardial, and valvular lesions.4,5–9 The disease entity is characterized by diffuse lymphoplasmacytic infiltrates and the presence of abundant IgG4-positive plasma cells in extensive fibrosis, frequently associated with tumorous swelling lesions and elevated serum IgG4 concentrations.1,2 IgG4RD presents with a variety of symptoms according to the affected organs and may be accompanied by serious complications such as organ dysfunction associated with IgG4-positive cell proliferation.1,4
For the diagnosis of IgG4RD, elevated concentrations of IgG4 in serum and tissue is helpful but not specific. Serum concentrations of IgG4 may vary widely in patients with documented disease, and approximately 30% of patients do not demonstrate elevated IgG4 in blood samples.1,10,11 A ratio of IgG4 to IgG in tissue greater than 0.5 also helps distinguish IgG4RD from other conditions.1,11,12 Clinical features in other organs may help diagnose IgG4-related cardiovascular disease in the absence of immunohistological confirmation. 4
The imaging features are frequently variable and nonspecific, and do not reliably distinguish IgG4-related disease from cancer, although a peripancreatic halo and diffuse narrowing of the pancreatic duct are classic findings in IgG4 pancreatitis and may help to distinguish this entity from pancreatic adenocarcinoma. 1 The diffuse encasement of the mesenteric vessels and periportal region in our patient is somewhat atypical for IgG4 disease and could also suggest infiltrative pancreatic adenocarcinoma or cholangiocarcinoma, although the lack of significant pancreatic or biliary ductal dilatation would be unusual. IgG4-related coronary artery lesions represent tumorous soft tissue formations surrounding the coronary arteries, or periarterial soft tissue thickening causing coronary stenosis. 4 Both focal and diffuse coronary involvement has been described, and our case likely represents a typical focal lesion. FDG-PET/CT might play a role in diagnosing IgG4-related vascular diseases6,13–15 although it is unclear whether FDG-PET/CT is useful as a diagnostic tool for other IgG4-related valvular, myocardial, and pericardial lesions. 4
There is no consensus concerning the optimal therapy for IgG4RD. 1 Aggressive treatment is needed when vital organs are involved in the setting of serious organ dysfunction and failure.1,16 Glucocorticoids are typically the first line of therapy. 1 Corticosteroid therapy with or without surgical intervention can lead to excellent outcomes when the cardiovascular system is involved. 4 With recurrent or refractory disease, other approaches such as B-cell depletion with rituximab may be useful.1,17 It is important to survey for extra-cardiovascular lesions when IgG4-related cardiovascular disease is suspected. 4 Multiorgan disease may present at diagnosis but can also evolve metachronously over months to years; therefore, careful follow-up after treatment is also necessary. 1
In conclusion, IgG4-related disease is an uncommon multi-system disorder, with potentially fatal outcomes in untreated cases. Cardiac/coronary disease should be suspected in patients with known IgG4RD and symptoms potentially related to the heart or pericardium. Further studies are needed to establish the optimal therapy for IgG4-related disease, and in the absence of immunohistological confirmation, clinical features in other organs may help to diagnose IgG4-related disease.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
