Abstract
Background
Active surveillance of men with prostate cancer relies on accurate risk assessments because it aims to avoid or delay invasive therapies and reduce overtreatment.
Purpose
To compare the diagnostic performance of pre-biopsy biparametric magnetic resonance imaging (MRI) with confirmatory multiparametric MRI in selecting men for active surveillance.
Material and Methods
The study population included biopsy-naïve men with clinical suspicion of prostate cancer undergoing biparametric MRI followed by combined (standard plus MRI targeted) biopsies. Men diagnosed with prostate cancer who were subsequently enrolled in active surveillance and underwent a confirmatory multiparametric MRI within three months of diagnosis were included in the study. Discrepancies between the pre-biopsy biparametric MRI and the confirmatory multiparametric MRI were assessed.
Results
Overall, 101 men (median age = 64 years; median prostate-specific-antigen level = 6.3 ng/mL) were included. Nine patients were re-biopsied after multiparametric MRI for the following reasons: suspicion of targeting error (three patients); a new suspicious lesion detected by multiparametric MRI (five patients); and an increase in tumor volume (one patient) compared with biparametric MRI. Confirmatory biopsies showed a Gleason grade group (GG) upgrade of ≥2 in 4/6 patients with suspicion of more advanced disease (missed suspicious lesion, increase in tumor volume) on multiparametric MRI. However, although multiparametric MRI subsequently detected a GG ≥ 2 prostate cancer lesion missed by biparametric MRI in 4% (4/101) of included men, the difference did not reach statistical significance (McNemar, P = 0.133).
Conclusion
Biparametric MRI could be used to select men eligible for active surveillance and a confirmatory multiparametric MRI performed shortly after inclusion seems unnecessary.
Introduction
Treatment for prostate cancer (PCa) ranges from invasive radical interventions, such as radical prostatectomy and radiotherapy, to non-invasive observational strategies that include either watchful waiting or active surveillance (AS) (1). AS is a well-established surveillance program for men with low-risk PCa that aims to reduce overtreatment and avoid or delay invasive interventions with their inherent side effects, e.g. impotence and incontinence (2). Men enrolled in AS are monitored closely using prostate-specific-antigen (PSA) measurements, digital-rectal examinations, and biopsies. Active treatment can be initiated in response to disease progression (2). However, AS is heavily dependent on accurate risk assessments including tumor localization, volume, and Gleason score (GS) grading. Diagnosis of PCa currently relies on transrectal ultrasonography-guided biopsies (TRUS-bx). However, TRUS-bx are prone to sampling errors and clinically significant PCas may be missed by the random untargeted sampling method (3). Multiparametric (mp) magnetic resonance imaging (MRI) has emerged as a non-invasive diagnostic tool for improved PCa risk assessment with a high negative predictive value (NPV) for ruling out significant disease. Thus, a confirmatory mpMRI may be used in men enrolled in AS for lesion characterization and staging to identify potentially missed clinically significant PCas for confirmatory targeted biopsies or to rule out aggressive disease and confirm eligibility for AS (4–7). At present, the European Association of Urology guideline recommends using mpMRI when a patient is enrolled in AS to confirm their eligibility (1). However, it is not specified at what time point during follow-up it should be performed. Furthermore, it was recently demonstrated that biparametric (bp)MRI improves PCa risk stratification, has a high NPV for ruling out significant disease in biopsy-naïve men, and may be used to avoid unnecessary biopsies (8–11). Compared with mpMRI, bpMRI is simpler and quicker with fewer scan sequences and no contrast agent. In addition, mpMRI is associated with greater costs and prolonged magnet-time. However, although bpMRI seems to improve PCa risk stratification of biopsy-naïve men in an initial detection setting (suspicion of PCa), it is unclear whether it can be further used for lesion characterization and staging to select men eligible for AS, which constitutes a different patient cohort with known low-risk PCa. Therefore, we compared the diagnostic performance of pre-biopsy bpMRI and confirmatory mpMRI scans in selecting men eligible for AS and assessed whether bpMRI missed suspicious PCa lesions that were subsequently detected by mpMRI.
Material and Methods
Our study population was derived from a prospective database (12) used to assess the diagnostic accuracy of bpMRI for PCa detection in biopsy-naïve men. This database included 1020 biopsy-naïve men with clinical suspicion of PCa who underwent bpMRI followed by combined biopsies (TRUS-bx and targeted biopsies of any Prostate Imaging Reporting and Data System [PI-RADS] ≥ 3 lesion). Overall, PCa was detected in 655 of the 1020 men; 180 of these 655 men were subsequently enrolled in AS based on national guidelines (12) or patient preference. The primary inclusion criterion for AS was men with low-risk PCa (i.e. tumor stage ≤T2a, PSA ≤ 10 ng/mL, and GS ≤ 6). However, well-informed men with limited intermediate-risk PCa (cT2b, PSA = 10–20 ng/mL, or GS = 3 + 4 in only 1–2 biopsy cores) could be enrolled based on patient preference. All patients underwent a confirmatory mpMRI as part of the departments’ diagnostic workup within the first year of entering AS. Of these, 101/180 men fulfilled the inclusion criteria and underwent an mpMRI within the first three months. These men constituted the final patient cohort for this study.
BpMRI and MpMRI
All MRIs (bpMRI and mpMRI) were performed using a 3-T MRI magnet (Ingenia version 5.3.1, Philips Healthcare, Best, the Netherlands) with a 16-channel surface coil and a built-in table coil (Philips Healthcare) positioned over the pelvis. No post-acquisition filtering was applied outside what is denoted “default” by the scanner vendor (Philips). BpMRI was performed before biopsies of the prostate. The bpMRI protocol included axial T2-weighted (T2W) and diffusion-weighted (DWI) imaging with reconstructions of the corresponding apparent diffusion coefficient maps and image acquisition times of approximately 15 min. In contrast to the bpMRI protocol, the mpMRI protocol included triplanar T2W imaging and dynamic contrast-enhanced T1-weighted (T1W) imaging sequences in the axial plane. Furthermore, anti-peristaltic drugs (hyoscinbutylbromid [20 mg/mL Buscopan injection fluid; Boehringer Ingelheim GmbH, Ingelheim am Rhein, Germany] and glucagon [1 mg GlucaGen; Novo Nordisk A/S, Bagsværd, Denmark]) were administered if tolerated. The mpMRI procedure consisted of preparing and positioning the patient, establishing intravenous access (approximately 15 min), and image acquisition (approximately 30 min). This enables us to perform two bpMRI scans per hour or one mpMRI scan per hour. The mpMRI and bpMRI protocol details are listed in Table 1. All MRI scans were reviewed by the same prostate MRI physician (>6 years of experience), who registered and scored any suspicious lesions on a 5-point scale according to their likelihood of being significant PCas (1 = highly unlikely; 2 = unlikely; 3 = equivocal; 4 = likely; and 5 = highly likely) using the PI-RADS version 2 criteria (13). The reader was aware of the bpMRI when reporting the mpMRI. Patients with no suspicious lesions were assigned a PI-RADS score of 1. However, as the bpMRI protocol did not include dynamic contrast-enhanced imaging, scoring of lesions in the peripheral zone relied solely on DWI findings (i.e. the dominant sequence). Therefore, an equivocal score of 3 was not upgraded to a score of 4 due to a lack of positive dynamic contrast-enhanced findings. An MRI suspicion score ≤ 2 was considered a low-suspicion or negative MRI result. MpMRI results were compared to the initial bpMRI examination and deemed equivalent if no new lesions were detected or if a lesion did not increase in PI-RADS score or volume. Because the bpMRIs only included one-dimensional axial T2W images, three-dimensional tumor volumes could only be assessed on mpMRI. As a result, an increase in tumor volume comparing bpMRI and mpMRI results was assessed subjectively by the reader for example by comparing slices for comparing, if relevant. All patients were discussed at a multidisciplinary conference that included urologists and radiologists, on which biopsy and MRI results were analyzed in consensus. If there was a mismatch between the PI-RADS assessment category/MRI suspicion (e.g. large tumor, low ADC value, invasive behavior) and the biopsy results (e.g. benign targeted biopsy or limited GS 6 tumor), then biopsy targeting error was suspected and a re-biopsy was recommended. Similarly, any patient with a new suspicious lesion or an increase in tumor volume on mpMRI was referred for re-biopsy if the result of such could potentially change the patients clinical management. As our fusion-biopsy system does not allow for recording of the needle-tracts, prior biopsy sites could not be retrospectively checked.
MpMRI and bpMRI protocols.

mpMRI, multiparametric magnetic resonance imaging; bp, biparametric (MRI); TR, repetition time; TE, echo time; FOV, field of view; NSA, number of signal averages; Temp res, temporal resolution; DWI, diffusion-weighted images; DCE, dynamic contrast-enhanced; BW, bandwidth.
Histopathological evaluation
The same genitourinary pathologist (>15 years of experience) reviewed all biopsy samples. When reviewing the biopsy cores, the pathologist was aware whether it was a systematic biopsy or a targeted biopsy. For each PCa-positive biopsy core, the location, the GS based on the International Society of Urological Pathology 2005 consensus (14), and the percentage of cancerous tissue per core were determined. In addition, patients were assigned International Society of Urological Pathology 2014 consensus Gleason grade groups (GGs) (15). Repeat biopsy results were compared to the original diagnostic biopsies.
Statistical analysis
Descriptive statistics were used to present patient characteristics. Continuous variables (i.e. age, PSA level, PSA density, and prostate volume) are described using medians, standard deviations, and ranges. Normality was tested using Kolmogorov–Smirnov test and a P value < 0.05 was considered statistically significant. Differences between the initial bpMRI scans and the mpMRI scans were evaluated using McNemar’s and chi-square tests. PI-RADS risk assessment groups from bpMRI and mpMRI were stratified into negative (PIRADS 1 and 2), equivocal (PIRADS 3), and positive (PIRADS 4 and 5) findings; intra-reader agreement was calculated using Cohen’s Kappa. Statistical analysis was performed using Rstudio software (ver. 1.1.5; RStudio, Inc., Boston, MA, USA) and a two-sided P value < 0.05 was considered statistically significant (16).
Results
In total, 101 men (median age = 64 years; median PSA = 6.3 ng/mL) met the requirements for inclusion in the study. The variables are normality distributed.
Time from the initial bpMRI to the confirmatory mpMRI was median 84 days ± 15 SD (range = 59–114 days). There was no significant difference comparing the distribution of PI-RADS risk assessment categories from bpMRI with mpMRI (Fig. 1). The majority of patients (85%) had clinically non-palpable tumors at stage cTx–T1c, while 11% had cT2a and 4% had cT2b tumors. Patient characteristics are listed in Table 2. Overall, the MRI reader assessment changed in 11 cases after confirmatory mpMRI for the following reasons: upgrade in PI-RADS assessment category from PI-RADS 3 to 4 (two cases); increase in tumor volume (three cases); and detection of new lesions (six cases). One of the upgrades from PI-RADS 3 to 4 was caused by positive DCE, but it did not have any clinical consequence, as both PI-RADS 3 and 4 lesions were biopsied in the initial setting. The other one was re-biopsied. Although, three increased in volume, two of them were not re-biopsied because it was decided at the multidisciplinary consensus conference that it did not have any clinical consequence for the patients. The six new lesions were: one PI-RADS 3 which was considered to be sampled by the standard biopsies and therefore not re-biopsied; one PI-RADS 3, which appeared secondary to a PI-RADS 5 lesion and therefore not re-biopsied; and finally four which were re-biopsied (two PI-RADS 3 and two PI-RADS 4; Table 3). Post-biopsy hemorrhage was present in 62% (n = 63) of mpMRI scans. Where 53% (n = 54) influenced the images. BpMRI had 8% (n = 8) susceptibility artifacts with partial effect on the images, 1% (n = 1) with no effect on the images, and 1% (n = 1) with effect on the images. Concerning rectal gas, bowl movement, or patient movement: MpMRI had 7% (n = 7) susceptibility artifacts with partial effect on the outcome of the scan, 2% (n = 2) with no effect on the outcome of the scan, and 1% (n = 1) with effect on the outcome of the scan. The intra-reader agreement comparing bpMRI to mpMRI was high (Cohen’s Kappa = 0.94; Table 4). Overall, nine patients underwent a confirmatory re-biopsy after mpMRI (Table 3 and Figs. 2–7). Five of them were caused by a change in the reader assessment. The decision to refer patients for re-biopsy was initiated at the discretion of the treading urologist and radiologist in consensus at the multidisciplinary team conference and based upon: (i) suspicion of targeting error of initial biopsies (n = 3); (ii) a new lesion detected (n = 4); or (iii) a significant increase in tumor volume (n = 1) comparing mpMRI to bpMRI. However, one was re-biopsied because the histopathologist was not satisfied with the material required at the biopsy (patient 6, n = 1). As a result, the remaining (n = 92) patients did not undergo immediate re-biopsy, either because the mpMRIs and bpMRIs were diagnostic comparable or because a re-rebiopsy would not change the clinical management of the patient. For example, patient 4 had suspicion of targeting error of the initial biopsies, because he had a large anterior PI-RADS 5 lesion (ADC 0.51) on bpMRI, but only GG 1 PCa on targeted biopsy (Figs. 4 and 5). This might be explained by PCa heterogeneity where larger lesions often harbor areas of mixed cancer aggressiveness within the same lesion. The discrepancy between the biopsy result and the MRI lead to re-biopsy that revealed a GG 3 tumor. Of the six patients for whom mpMRI revealed a suspicion of more advanced disease, confirmatory biopsies showed a GG upgrade ≥ 2 in four (patients 3, 4, 6, and 7).
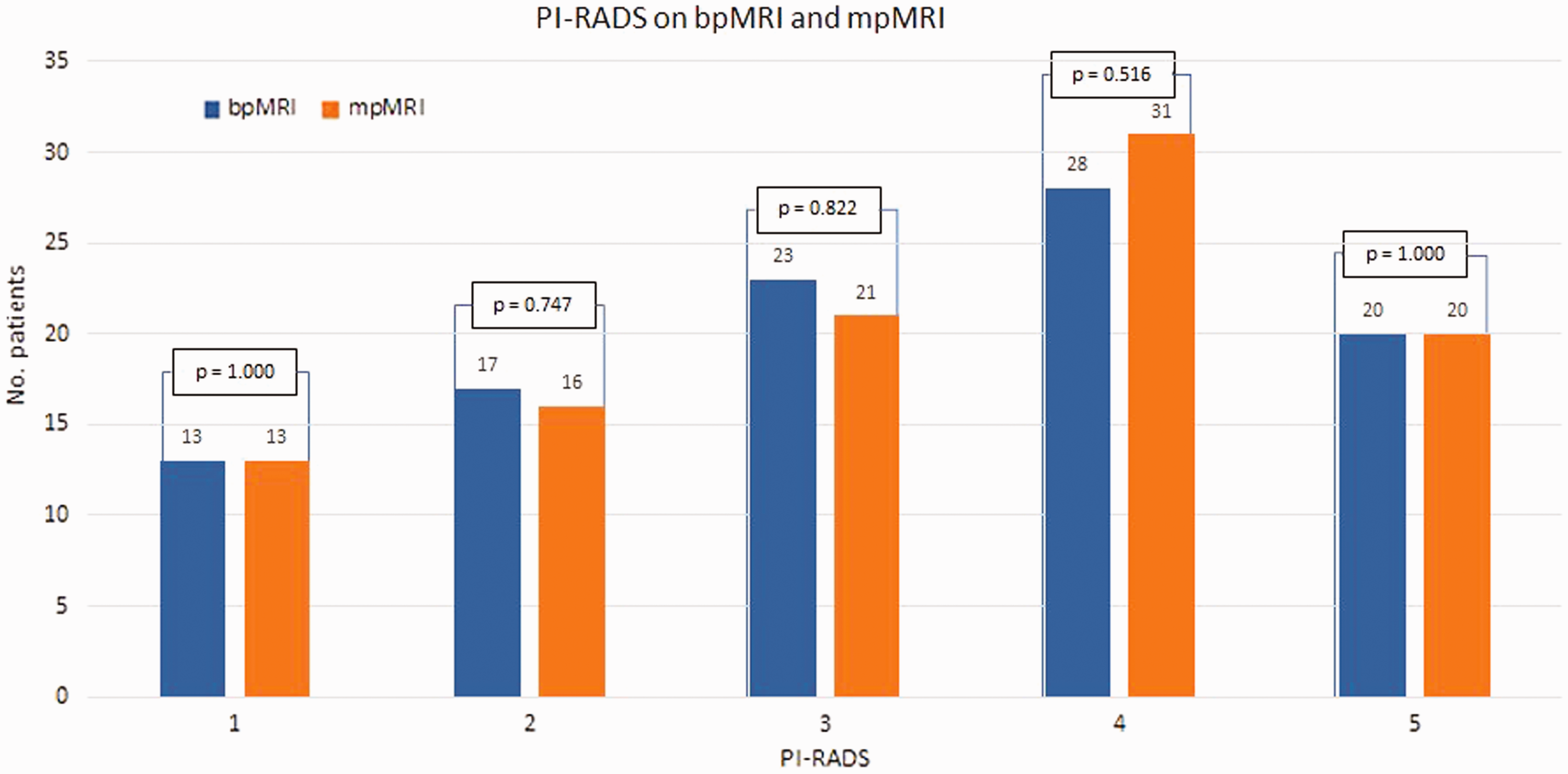
Comparison of patient PI-RADS scores obtained from bpMRI and mpMRI results PI-RADS, prostate imaging reporting and data system; mpMRI, multiparametric magnetic resonance imaging; bp, biparametric (MRI).
Baseline characteristics.

SD, standard deviation; PSA, prostate-specific antigen; PI-RADS, prostate imaging reporting and data system; bpMRI, biparametric magnetic resonance imaging; GG, Gleason grade group.
Patients who had biopsies after mpMRI.
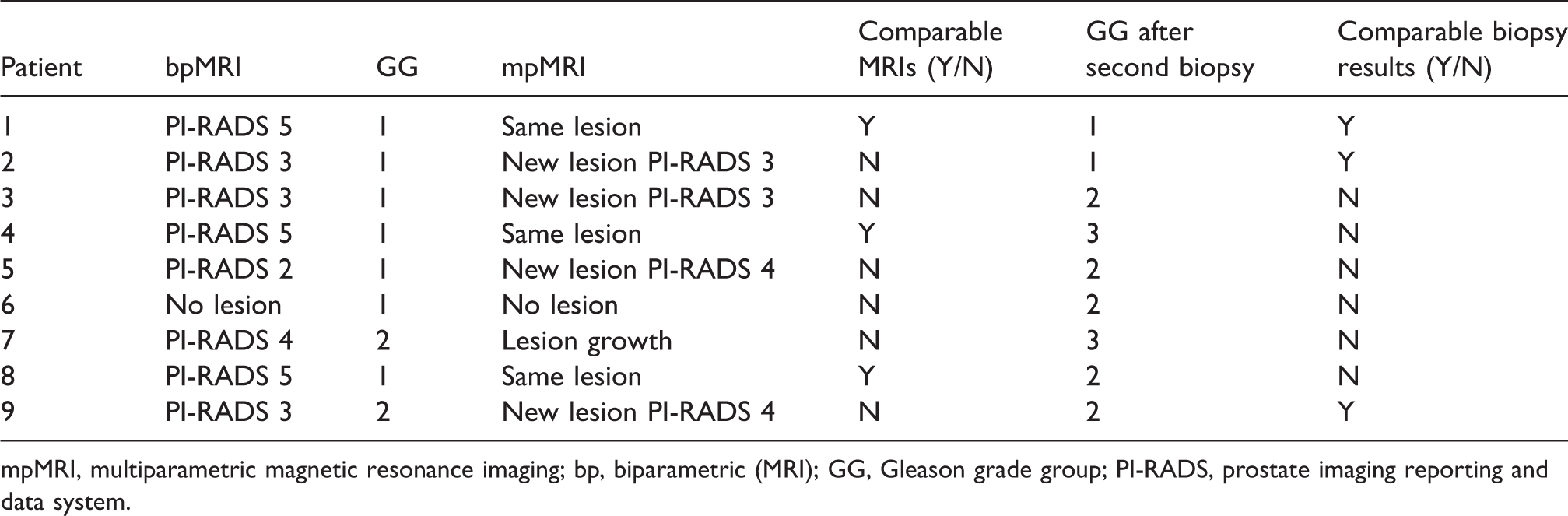
mpMRI, multiparametric magnetic resonance imaging; bp, biparametric (MRI); GG, Gleason grade group; PI-RADS, prostate imaging reporting and data system.
Reader assessment changes in PI-RADS score.

PI, prostate imaging reporting and data system; SUM, summarized.
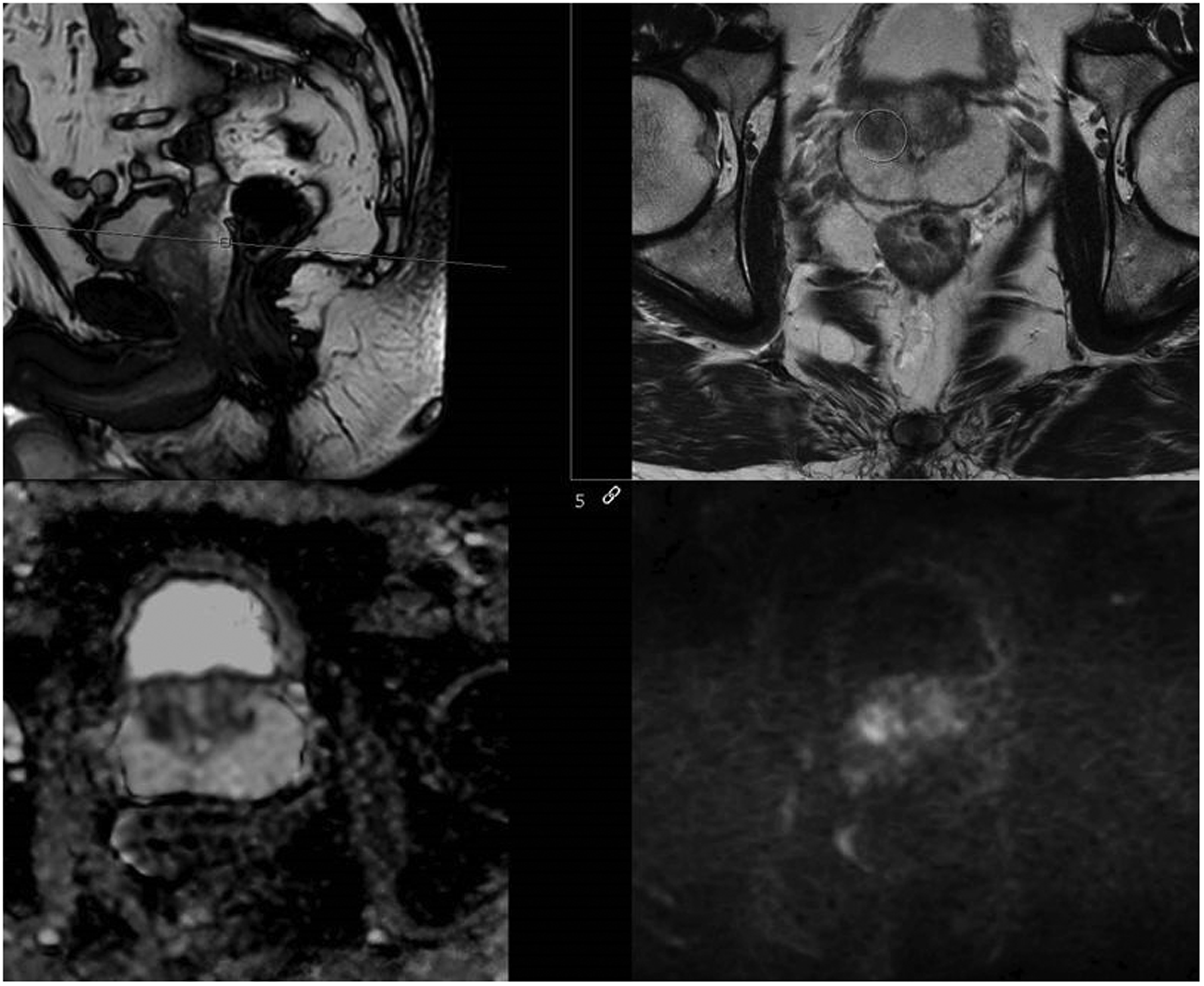
BpMRI of patient 3. BpMRI of an anterior PI-RADS 3 lesion where targeted biopsies revealed a GG 1. PI-RADS, prostate imaging reporting and data system; bpMRI, biparametric magnetic resonance imaging.

MpMRI of patient 3. A new anterior PI-RADS 3 lesion as seen on the bpMRI. Repeat targeted biopsies revealed a Gleason score upgrade to GG 2. PI-RADS, prostate imaging reporting and data system; mpMRI, multiparametric magnetic resonance imaging.

BpMRI of patient 4. BpMRI of an anterior PI-RADS 5 lesion where targeted biopsies revealed a GG 1. This raised suspicion of targeting error. PI-RADS, prostate imaging reporting and data system; bpMRI, biparametric magnetic resonance imaging.
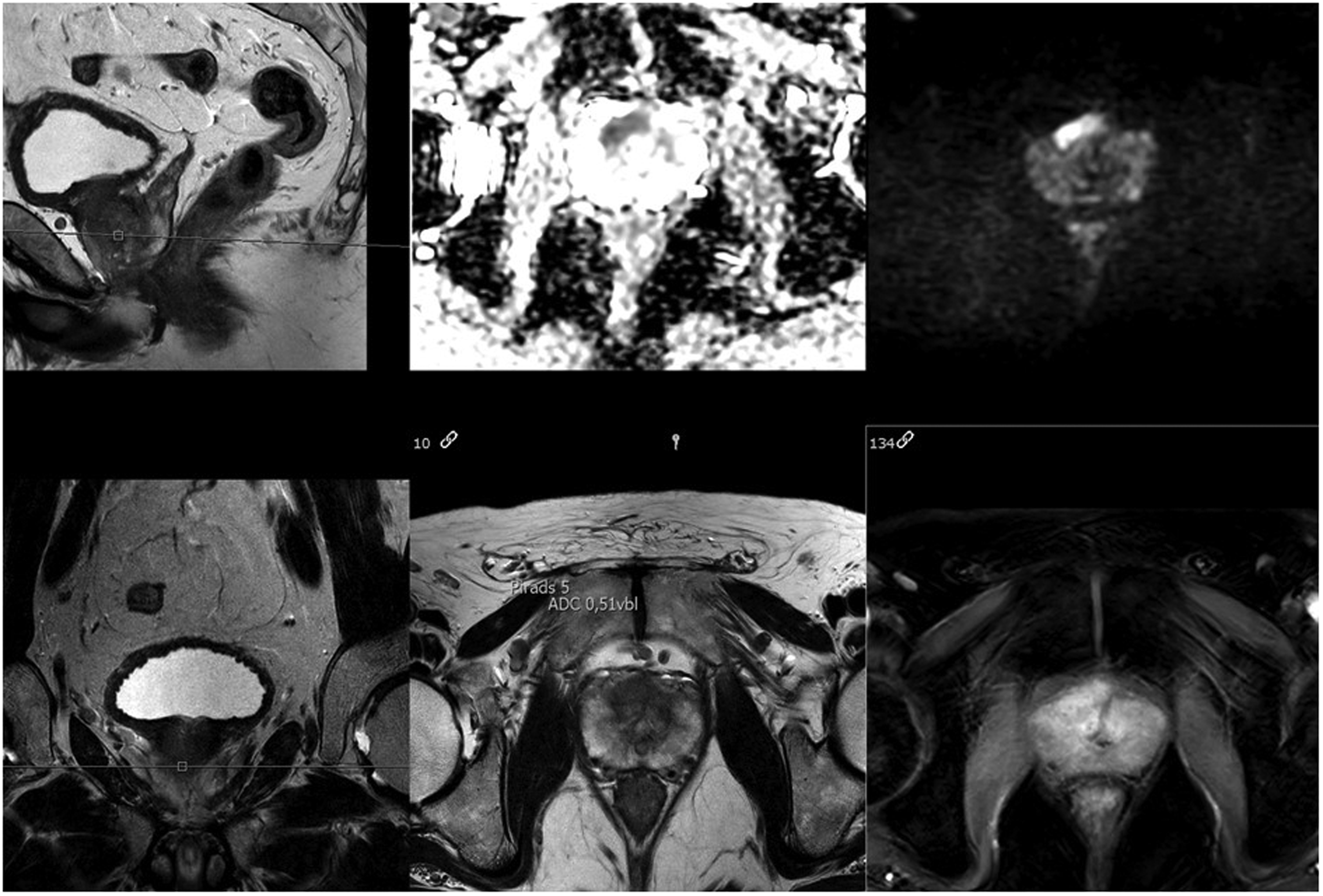
MpMRI of patient 4. The same anterior PI-RADS 5 lesion as seen on the bpMRI. Repeat targeted biopsies revealed a Gleason score upgrade to GG 3. PI-RADS, prostate imaging reporting and data system; mpMRI, multiparametric magnetic resonance imaging.
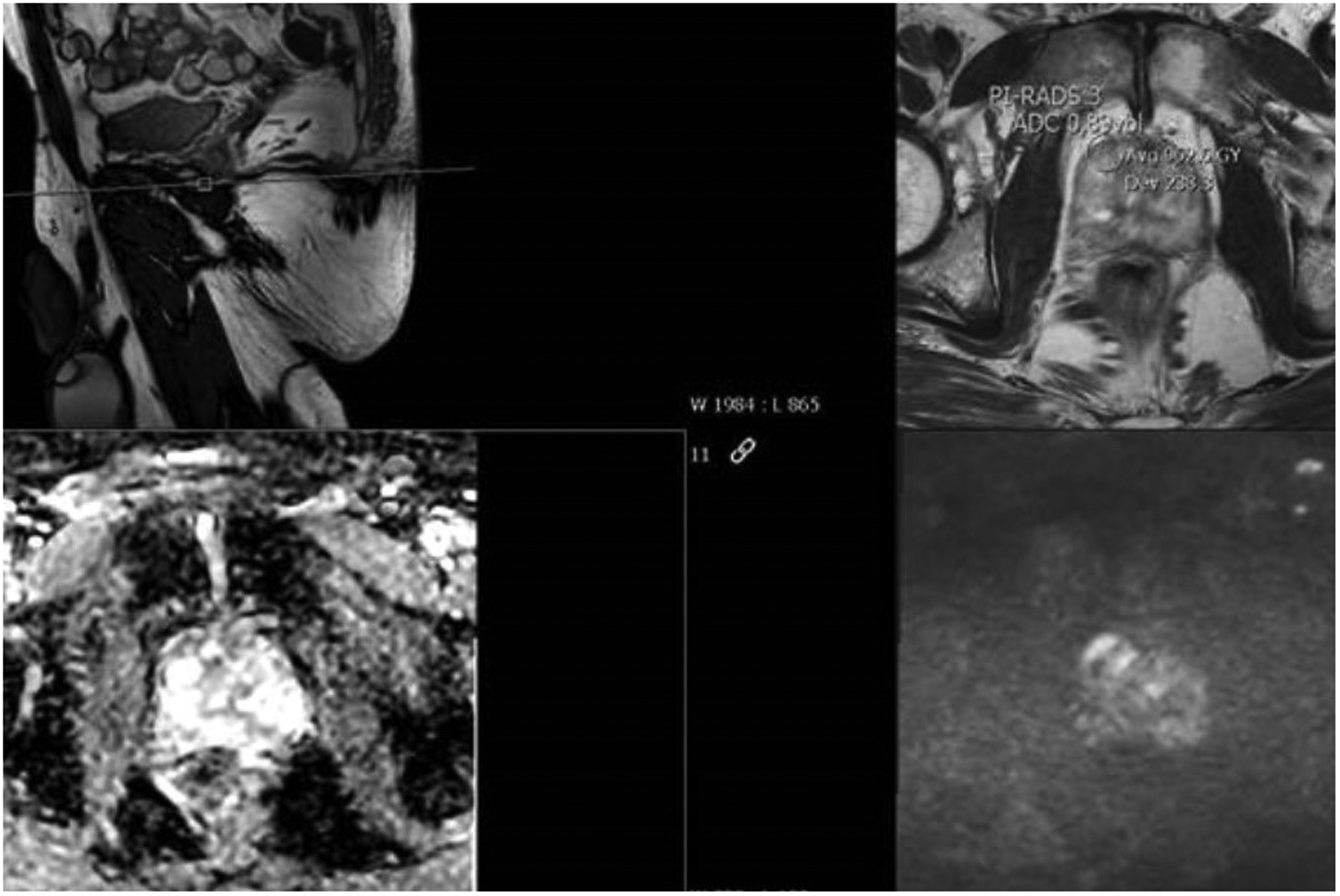
BpMRI of patient 9. BpMRI of an anterior PI-RADS 3 lesion where targeted biopsies revealed a GG 2. PI-RADS, prostate imaging reporting and data system; bpMRI, biparametric magnetic resonance imaging.

MpMRI of patient 9. The same anterior lesion as seen on the bpMRI but on mpMRI is upgraded to an PI-RADS 4. Repeat targeted biopsies revealed a GG 2 again. PI-RADS, prostate imaging reporting and data system; mpMRI, multiparametric magnetic resonance imaging.
However, the false-negative rate was 4% (4/101). The false-negative rate was defined as a lesion detected on mpMRI with GG ≥ 2 which was not detected on bpMRI. The difference did not reach statistical significance (McNemar’s test, P = 0.133).
Discussion
At present, guidelines recommend performing mpMRI in men with low-risk PCa enrolled in AS to confirm their eligibility and reduce the risk of missed occult significant cancer (1). Here we compared the diagnostic performance of a more simple and rapid bpMRI protocol with confirmatory mpMRI scans in selecting men eligible for AS and assessed whether bpMRI missed significant PCa lesions that were subsequently detected by mpMRI. The variables are normality distributed but with large enough sample sizes the violation of the normality assumption should not cause major problems (18). Overall, we found no significant difference between bpMRI and confirmatory mpMRI performed in men three months after enrolment in AS. This indicates that a rapid and simple pre-biopsy bpMRI performed in the initial setting of detecting PCa also suffices as an initial scan for AS eligibility and an additional confirmatory staging mpMRI performed shortly after AS enrolment is not necessary. Prior studies have evaluated and compared the diagnostic performance of bpMRI and mpMRI in detecting PCa (18,19). However, our study further assesses the ability of bpMRI for the specific task of selecting men for AS, which constitutes a selected patient cohort, as these men have known low-risk PCa and their eligibility for AS based on PSA, DRE, and biopsy findings is decided after all the diagnostic results are available. While limited data are available for bpMRI to select men for AS, our findings are consistent with a previous retrospective study that compared the diagnostic accuracy of bpMRI and mpMRI in detecting significant PCas in 204 patients (19). Six patients had suspicion of more advanced disease, of which five were due to new appearing lesions on mpMRI. Four of these men had significant PCa detected on confirmatory biopsies. The numbers are limited and larger prospective studies are needed to confirm our conclusion. However, despite the limited patient number, our study provides additional evidence for the reliability of using bpMRI in the initial assessment of selecting men for AS. Overall, two independent readers missed only four (4/204) and three (3/204) significant cancers using bpMRI and zero (0/204) and five (5/204) significant cancers using mpMRI, respectively. Similarly, Kuhl et al. found no significant differences in the diagnostic accuracy of bpMRI and mpMRI in 542 men with elevated PSA who underwent repeated prostate biopsies (20). However, while bpMRI seems sufficient as an initial eligibility-scan for AS, mpMRI still plays a role during follow-up in monitoring the disease. Men enrolled in AS undergo close surveillance, including repeat biopsies after one year. Ideally, these biopsies should be preceded by confirmatory mpMRI to identify possible disease progression and the limited number of bpMRI missed significant lesions, if bpMRI was used as an initial scan to confirm eligibility for AS.
Overall, mpMRI performed three months after bpMRI raised suspicion of more advanced disease in 6% of our study patients, in which confirmatory biopsies revealed a GG upgrade in 4% (4/101 patients), However, no patient had GG4 or GG5 PCa, and in comparison to the contemporary well-established AS cohorts and surveillance programs that do not include MRI findings and where PCas are missed by TRUS-bx in up to one-third of patients, the 4% false-negative rate using bpMRI seems justifiable (21–23). Moreover, as the time interval from bpMRI to mpMRI was only three months, it might be that the new lesions detected by mpMRI are not newly developed lesions, but rather lesions initially missed by the reader or the bpMRI. In the case of patient 2 (with a GG 1 on first and second biopsy set), biopsy cores from the standard biopsies of the first set of biopsies detected what was seen as a new lesion found on mpMRI. But as it is a GG 1, it cannot be expected that bpMRI or mpMRI will detect it. In the case of patient 9 (with GG 2 on first and second biopsy set), standard biopsies also detected the lesion which appeared as a new lesion on mpMRI. The remaining patients with new lesions (patients 3 and 5) were not detected by standard biopsies. But as standard biopsies are only biopsies and thereby do not cover the whole gland, it cannot be concluded that if the standard biopsy misses a lesion then it is not there. This means that even though the first set of standard biopsies misses the lesion, it might still be there. This is also supported by the very short time interval which makes the possibility of a new lesion very unlikely. Furthermore, it is important to realize that there will always be a risk of targeting error when performing image-guided biopsies despite careful efforts to align the MRI and TRUS images. Biopsy targeting error was suspected in three patients (all anterior lesions), of which two had a GG upgrade following re-biopsy. This may be explained by the fact that only 1–2 targeted biopsy cores was obtained from each suspicious lesion in the initial biopsy setting. Sampling errors may be reduced by spacing biopsies and obtain more targeted cores per lesion (focal saturation).
Our study has limitations. First, the study is a retrospective analysis of prospectively collected data with a limited number of patients who fulfilled the final inclusion criteria. Because not all patients in our AS cohort underwent a confirmatory mpMRI three months after the initial diagnostic bpMRI, there might be a selection bias for those who did. Furthermore, because the bpMRI and mpMRI scans were not performed at the same time point, we cannot theoretically rule out disease progression. However, we think it is reasonable to conclude that the additional aggressive cancerous lesions (GG ≥ 2) detected by mpMRI were not caused by disease progression, but were missed in the initial assessment of bpMRI, because low- and intermediate-risk PCas generally develop slowly (17). Second, this study was performed at a single center with one dedicated MRI physician reading all the MRI scans and two highly experienced TRUS operators performing all the biopsies. As a result, no inter-reader analysis could be done. Less experienced readers and operators might not achieve the same diagnostic yield. However, we found a high intra-reader agreement (Cohen’s Kappa = 0.94), which corresponds with our previously reported inter-reader agreement (Cohen’s Kappa = 0.84–0.87) comparing bpMRI with mpMRI (19). Third, because we used biopsy results as standard reference, PCa lesions may have been missed by both bpMRI, mpMRI, and biopsies. Furthermore, as only nine patients were re-biopsied, we cannot exclude the possibility that MRI invisible lesions would have been detected by standard re-biopsies if all men regardless of mpMRI findings underwent repeat biopsies (standard and targeted) after three months. Thus, we acknowledge the limitation of this being a retrospective study without prostatectomy specimens as ideal comparing reference to confirm presence or absence of missed PCa lesions. And that the diagnostic and staging accuracy cannot be evaluated precisely without because of this. Furthermore, our numbers are limited and larger prospective studies are needed to confirm our conclusion. However, although the PROMIS study was performed in biopsy-naïve men and not in an AS cohort, the study showed a good association between mpMRI suspicion and significant PCas, using 5-mm template sampling as standard reference (4) with only limited added value of TRUS-bx in mpMRI-negative men. Furthermore, because all patients underwent standard biopsies following initial bpMRI, including those with low-suspicion bpMRI scans, using biopsies as standard reference allowed us to compare outcomes that reflect clinical practice. Fourth, because the bpMRI scans were scored using a modified PI-RADS score, equivocal scores of PI-RADS 3 in the peripheral zone were not upgraded to PI-RADS 4 due to a lack of positive dynamic contrast-enhanced findings. However, this would only affect biopsy results stratified by PI-RADS scores and not the overall outcomes, because all PI-RADS 3 lesions were biopsied. Finally, bpMRI was performed in biopsy-naïve men and mpMRI was performed in recently biopsied men. Thus, the pre-test cancer prevalence differed (biopsy-naïve versus AS cohort), which might have influenced the diagnostic performance of bpMRI and mpMRI. Furthermore, post biopsy-related artefacts and morphological changes in the prostate may have influenced the sensitivity of mpMRI. However, it is a generally recommended to wait 6–8 weeks after biopsy before prostate MRI is performed to avoid artefact from biopsy-related hemorrhage. Still, this does not account for biopsy induced inflammation or atrophy.
Despite these limitations, our data provide additional evidence for the reliability of using bpMRI in the initial assessment of selecting men for AS.
In conclusion, a rapid and simple bpMRI could be used to select men eligible for AS and a confirmatory mpMRI performed shortly after inclusion seems unnecessary.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
