Abstract
Cerebral tuberculosis (TB) presents most frequently as meningitis in the basilar cistern; however, it can also manifest in various other ways, such as localized encephalitis, abscess, and tuberculoma. Here, focusing on imaging findings, we report an immunocompetent case who demonstrated multiple parenchymal lesions and was diagnosed with cerebral TB after testing positive on QuantiFERON (QTF); her clinical signs/symptoms and laboratory findings responded well to anti-TB medication therapy. The patient was a 60-year-old woman with the chief complaints of headache and consciousness disturbance. On admission, cerebrospinal fluid (CSF) examination showed increased monocyte predominance. T2-weighted images showed multiple, widely distributed hyperintense lesions in the periventricular and deep white matter. Gadolinium-enhanced three-dimensional gradient echo T1-weighed images revealed numerous granules or faint, small, enhanced foci in lesions in the periventricular and deep white matter, central gray matter, and hippocampus. Some abnormal sulcal enhancement was detected in the pia mater, indicating meningitis. Clinically, the diagnosis was difficult to make, but as the QTF result was positive, anti-TB drugs were administered, after which both the symptoms and CSF cell count showed improvement.
Keywords
Introduction
Tuberculosis (TB) in the central nervous system (CNS) may present as tuberculoma, meningitis, cerebritis, abscess, or miliary TB via hematogenous spread (1). Among these conditions, basal meningitis is a well-known subtype. In contrast, miliary type tuberculomas are extremely rare in immunocompetent individuals. Herein, we report a case of an immunocompetent patient with miliary tuberculomas, with extensive perivascular dissemination in the cerebral parenchyma, which were detected on magnetic resonance imaging (MRI).
Case report
A 60-year-old woman, who was from a non-endemic area, presented with symptoms such as headache, short-term memory impairment, and mild consciousness disturbance. Her medical history included traumatic subarachnoid hemorrhage nine months before presentation, but her family history was unremarkable. The patient had a medical history of using cough suppressants, expectorants, anti-allergy agents, and corticosteroid nasal drops. About five years previously, pigeons had nested in the veranda of the patient’s home for a period of time.
In the neurological examination, her consciousness level was Glasgow Coma Scale E4V4M6 as disorientation was present, and the Babinski and Chaddock signs were positive on the left. Other neurological symptoms or signs were negative. Blood biochemical test findings revealed an elevated white blood cell count of 8900/µL (normal range = 3300‒8000/µL) and elevated C-reactive protein level of 0.05 ng/mL (normal 0‒0.14 ng/mL). In the cerebrospinal fluid (CSF) test, cell count was elevated (212/µL), with an increased monocyte predominance, a low glucose level of 48 mg/dL (normal range = 50–75 mg/dL), and a high protein level of 61 mg/dL (normal range = 10–40 mg/dL), suggestive of meningitis. Plain chest radiograph and CT were performed but were unremarkable. CSF analysis was sent for both acid-fast staining and tuberculosis culture, both of which were negative. Erythrocyte sedimentation rate (ESR) was not evaluated in this case.
For magnetic resonance imaging (MRI) examination, a 3-T MR system (Signa HDxt; GE Healthcare, Milwaukee, WI, USA) was used. In each clinical stage, the patient underwent serial examinations that included T2-weighted (T2W) images (TR/TE = 5800/84 ms) and gadolinium (Gd)-enhanced, spoiled gradient echo (GRE) T1-weighted (T1W) images (TR/TE = 7.12/3.15, flip angle [FA] = 13°) with Gadteridol 0.2 mL/kg.
Cerebral MRI examination was performed on admission. On T2W imaging, multiple poorly demarcated, variously sized, hyperintense lesions were observed from the periventricular to the deep white matter of the bilateral lateral ventricle, including in the right inferior horn, bilateral triangle, right anterior horn, and body of the ventricle. The right hippocampus, bilateral basal ganglia, and thalami, as well as the white matter in the bilateral temporal and occipital lobes were also affected (Fig. 1a–c).

MRI at admission. (a–c) (From cephalad to caudal): T2W imaging shows multiple poorly demarcated hyperintense lesions in the periventricular white matter of subependymal zone to the deep white matter (a), anterior horn of the lateral ventricle, bilateral basal ganglia, thalami, white matter of the temporal and occipital lobes (b), and right hippocampus (c). (d–f) (same levels as a–c): Gd-enhanced T1W imaging shows multiple enhanced miliary foci measuring up to 2 mm in size, partially confluent along the subependyma and the deep white mater. (g) Three-dimensional susceptibility-sensitive gradient echo imaging shows linear hypointense changes in bilateral thalami and both triangles of the lateral ventricle in the hyperintense lesions.
On Gd-enhanced three-dimensional (3D) GRE T1W imaging, multiple enhanced miliary foci, measuring up to 2 mm in size, were partially confluent along the subependyma and the deep white matter, indicating perivascular space involvement (Fig. 1d–f). Some abnormal sulcal enhancement was detected along the pia mater of the parietal and occipital lobes, suggestive of meningitis. Similarly, in the right internal auditory canal and geniculate ganglion area of the facial nerve, faint enhancement was demonstrated. The ventricle was slightly enlarged and a CSF circulatory disorder was suspected. On 3D susceptibility-sensitive GRE T2* images, the bilateral thalami and triangles of the lateral ventricle showed linear hypointense changes, suspected to represent venous congestion (Fig. 1g).
Course
Although cryptococcus antigen and CSF polymerase chain reaction for Mycobacterium tuberculosis complex were negative, the initial India ink staining gave an equivocal result. From these laboratory data and the suspicious history of possible exposure to a pigeon parasite, cryptococcal meningitis was suspected, and therefore antifungal agent administration was begun soon after admission. Serology was negative for various possibly causative viruses. At this stage, although the CSF laboratory data showed a tendency toward improvement, the patient’s symptoms did not improve.
On the 12th hospital day, a positive QuantiFERON (QTF) test result was obtained, and the possibility of brain tuberculoma or tuberculous meningitis was considered. Therefore, combined anti-TB drug and steroid therapy was started. MRI performed one month after initiation of the therapy revealed an increased number of hyperintense lesions in the lateral periventricular white matter, subcortical white matter, basal ganglia, and thalamus on T2W imaging (Fig. 2a–c). The lateral ventricle was prominently enlarged, as compared with the corresponding images obtained at admission (Fig. 2). In contrast to T2W imaging, the number of enhancing miliary foci had decreased on post-contrast T1W imaging (Fig. 2). Furthermore, the patient’s cognitive function showed improvement, allowing hospital discharge, with her subsequent course continuing to be smooth.

MRI after commencing anti-TB drugs. (a–c) (same level as Fig. 1a–c): T2W imaging shows an increased number of lesions (arrows). (d–f) (same level as a–c): Gd-enhanced T1W imaging shows a decreased number of enhanced miliary foci.
On MRI obtained two months after discharge, the aforementioned hyperintense lesions on T2W imaging showed some improvement (Fig. 3a–c), while the contrast-enhanced miliary foci showed significant improvement (Fig. 3d–f). Hydrocephalus gradually worsened during the course after discharge, which was revealed on serial follow-up MRI studies. Because enlargement of the ventricle ceased, and clinical symptoms associated with hydrocephalus were not observed, treatment to prevent the enlargement of the ventricles was not performed.
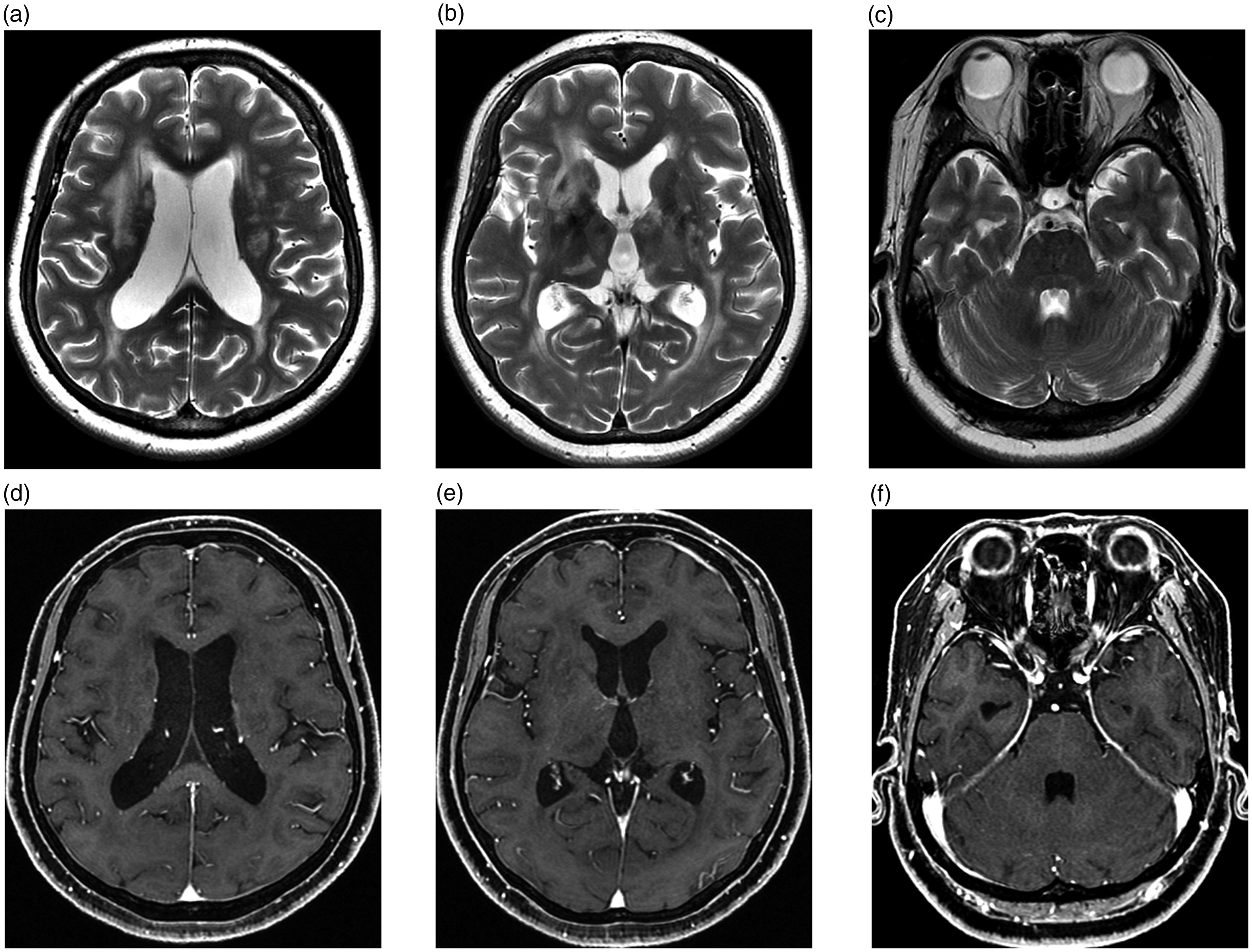
MRI at four months after discharge. (a–c) (same level as Fig. 1a–c): The lesions have decreased in size, but there is a tendency for hydrocephalus. (d–f) (same level as a–c): Gd-enhanced miliary foci are unclear and have improved.
Discussion
We experienced an extremely rare case of an immunocompetent patient with miliary tuberculomas in the CNS. The frequency of the subtypes of tuberculosis in the CNS has been reported to be as follows: meningeal, 61%; tuberculoma, 33%; infarct, 16%; hydrocephalus, 22%; and epidural, 11% (1). In each subtype, small tubercular lesions known as Rich foci initially develop in the CNS, either in the meninges, brain, or spinal cord, and become active after a quiescent period (1,2) via distant hematogenous spread with low-level bacteremia (2).
The imaging findings of parenchymal miliary tuberculomas have been described in only a few case reports and research articles (1,3–17), published mostly from developing countries. It has been reported that intraparenchymal tuberculomas occur in the frontal and parietal lobes in adults, in contrast to the infratentorial region in children (10). In general, tuberculomas appear as hypointense lesions on T1W imaging and hypo- or hyperintense lesions on T2W imaging. Tuberculomas are in the range of 2 mm–6 cm on MRI. It has been reported that 88% of such tuberculomas are < 2.5 cm in diameter (3). They show initial giant cell-predominant inflammation, without central caseation, and with poor capsule formation, and are reflected as a homogenous enhancing lesion on post-contrast-enhanced T1W imaging. At a later stage, the tuberculoma develops central caseation, showing ring-enhancement and a collagen-rich capsule with a hypointense caseating center (16). In one study, tuberculomas including caseating cystic-centered granulomas were observed in 85.9% of cases, and those including noncaseating granulomas were observed in 14.1% of cases (4). On T2W imaging, tuberculoma has a central hypointensity, indicating necrosis and/or hypocellularity, with an increased number of macrophages and gliosis, and with a peripheral hyperintense area due to edema (16) as a target sign. The detection of miliary lesions < 5 mm is often suboptimal on non-contrast T2W imaging.
In our case, Gd-enhanced T1W imaging showed non-caseating miliary lesions < 2 mm in size disseminated in the cerebral parenchyma. T2W imaging showed no hypo- or hyperintense miliary lesions in multiple extensive hyperintense changes. These findings indicated that these were early lesions. This makes our case rare in terms of the disease stage and immune status and is considered worth reporting; miliary lesions were detected on Gd-enhanced images.
Pathologically, the brain tissue immediately underlying the tuberculous exudate shows various degrees of edema, perivascular infiltration, and a microglial reaction, a process known as “border zone reaction” (18). Direct contact of the exudate with the brain surface causes the border zone reaction, which damages the underlying brain tissue. Rich and McCordock ascribed most of these changes to a hypersensitivity response (19,20). In our case, the miliary lesions were minimal, showing continuity from the subependymal region to the deep white matter, suggesting a condition having a minimal infiltration along the perivascular space (21). Such findings in the brain should thus be considered suggestive of TB, regardless of the patient’s immune status.
Finally, in our case, the initial hyperintense lesions on T2W imaging showed an increase in size during the anti-tuberculum treatment, despite which the clinical symptoms gradually improved. Such a “paradoxical response” has been previously reported (22,23). A patient with miliary tuberculomas in whom the clinical symptoms resolved, but the radiological abnormality persisted, has been described previously (5). A case series of 10 patients with CNS TB showed the occurrence of a paradoxical response in one patient (5,6). Such a response may occur within days or even within one year after starting chemotherapy, despite regular anti-tubercular chemotherapy. No clear explanation for the occurrence of a paradoxical reaction in an immunocompetent patient is yet available; however, it is surmised to have an immunological basis.
Concerning the diagnostic approach for cerebral TB, during CSF analysis, it would be prudent to assess acid-fast staining and TB culture. The Mantoux test is useful in the diagnosis of TB; ESR can be used as a simple, supplementary tool to follow-up for treatment response in endemic areas (16). Our report is not from an endemic country and cerebral TB is very rare. In our case, the aforementioned analyses were positive or were not performed; moreover, positive results of QFT and MR findings were clues to the diagnosis of TB and allowed ruling out of possible diseases. Serious findings on cerebral MRI required diagnostic treatment in our case, without sufficient evidence of cerebral TB in our non-endemic country. ESR is not examined routinely in our hospital for follow-up of rare cerebral TB.
With respect to therapy, British Infection Society guidelines generally recommend 12 months of treatment with rifampicin and isoniazid, with pyrazinamide and ethambutol stopped after two months, in cases such as ours. They also recommend that all patients with TBM receive adjunctive corticosteroids, regardless of disease severity at presentation. The regimen in adults should be as follows: dexamethasone 0.4 mg/kg/24 h, with reduction over 6–8 weeks. Accordingly, anti-TB drugs and corticosteroids were administered to our patient (24).
In conclusion, we here reported a rare case of an immunocompetent patient with miliary tuberculomas. Gd-enhanced 3D GRE T1W imaging showed multiple minimally enhancing foci with partial confluence along the perivascular spaces in the white matter or perforating territories. Local meningeal enhancement was also seen. Imaging findings of multiple granular to nodular lesions in the brain should lead to consideration of cerebral TB regardless of the patient’s immune status. In addition, attention should be paid to the paradoxical response.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
