Abstract
Object:
Predictor of the risk of subclinical nodal metastasis in early oral cancer would be of significant benefit in determining the need for elective neck dissection or adjuvant therapy. Depth of invasion (DOI) and glucose transporter (GLUT-1) expression have been assigned an independent prognostic significance in predicting the risk of nodal metastasis. The present study has been undertaken to evaluate the mechanisms linking GLUT-1 expression to DOI in progressive grades of oral squamous cell carcinoma (OSCC).
Method:
The GLUT-1 expression was analysed by immunohistochemistry, and its association with the DOI was assessed in 90 patients in progressive histological grades of OSCC.
Result:
We hypothesized that OSCC characterized by high frequency of lymph node metastasis, distant metastasis, or local recurrences showed highly significant statistical correlation of DOI with the GLUT-1 over expression at the invasion front in progressive histological grades of OSCC, thereby revealing their potential role in predicting both tumour aggressiveness and prognosis.
Conclusion:
The correlation of these two parameters, that is, DOI with GLUT-1 over expression at the invasion front, used simultaneously in progressive histological grades of OSCC, might have a significant prognostic value by highlighting those cases that should be subjected to further lymph node assessment, thereby predicting their risk of occult metastasis, assisting in the selection of patients requiring more aggressive therapy.
Keywords
Introduction
Oral squamous cell carcinoma (OSCC) is characterised by mortality and morbidity rates that remain unwavered in spite of all the advancement in the field of oncology and surgery. 1 Nodal recurrence as a result of undetectable subclinical nodal metastasis is, however, the main hurdle in the management of early stages I and II carcinomas. 2
Thus, a predictor of the risk of subclinical nodal metastasis in oral cancer would be of significant benefit in determining who requires further lymph node assessment and subsequently elective neck dissection or adjuvant therapy. In the clinical perspective, one needs to identify one or at best two histological predictors of metastasis, in order to address the neck optimally. 2
Depth of invasion (DOI) is considered to be a more reliable histological feature and currently the best available predictor of occult nodal disease. 2,3 There is a general agreement that most useful prognostic information can be deduced from the invasive front of the tumour where the deepest and presumably most aggressive cells reside. 4
Malignant cells show an increased glucose uptake that serves to feed the cancer cells during their expansive activity. The up-regulation of glucose transport across the plasma membrane is mediated by facilitative glucose transporter (GLUT) proteins. GLUT-1 is also known as facilitated glucose transporter member 1. It was the first facilitative sugar transporter discovered and is able to transport primarily glucose. 5
The rapidly growing tumour quickly outstrips its vasculature. Under sustained hypoxic conditions, tumour cells undergo a variety of genetic changes allowing them to prolong their survival under hypoxia.
6
The up-regulation of glycolytic metabolism through GLUTs is a survival adaptation of tumour cell under hypoxia. GLUT-1 has been identified as endogenous markers of chronic hypoxia in solid tumours
7,8
Thus, this study is undertaken to evaluate the prognostic value in predicting the risk of nodal metastasis of GLUT-1 over expression at the invasive front of primary lesions of OSCC and its correlation with the DOI so as to analyse their biological significance in the progression of OSCC, thereby having a significant prognostic value by highlighting those cases that need to be subjected to further lymph node assessment.
Materials and methods
Tissue samples
The study sample comprised formalin-fixed and paraffin-embedded tissue specimens from 90 patients with T1/T2 N0M0 OSCC, who presented to our department in the period 2007–2014, and 30 samples of normal mucosa as the control group. Normal mucosa tissue samples were obtained from gingiva of patients who did not have clinically detectable lesions and were undergoing disimpaction of maxillary canine and mandibular third molars. A total of 120 patients with age ranging from 21 to 83 years were selected. There was male predominance of 61.7% (a total of 74 males), whereas only 38.5% (a total of 46 females) of females were part of the study group.
Stringent inclusion criteria were applied. The staging was performed on the basis of clinical assessment; lymph node status was assessed by palpation. Only those patients were included in whom it was possible to retrieve surgical pathology material that could be reviewed as per our guidelines.
The maximum tumour depth is quantifiable measurements and more likely to be precise and accurate. 2 In the pre-neoplastic lesions of tumour, an elevation of the GLUT-1 expression could be observed. The changes in GLUT-1 expression were stated to be an early event during the development of not only squamous cell carcinoma (SCC) but also other carcinomas. 11
Through this study, we wanted to study the progressive change in the expression of both these parameters, that is, DOI and GLUT-1 expression levels, in the progressive histological grades of OSCC. Hence, the groups were divided into four progressive histological groups of OSCC.
Superficially invasive group consisted of lesions in which basement membrane was not intact with focal and/or superficial invasion of less than 2 mm and no deeper than lamina propria. Focal invasion was into the immediate subjacent connective tissue. In order to study the progression of invasive depth from subjacent connective tissue into deeper connective tissue, histopathologically confirmed cases of OSCC were considered and grouped into superficially invasive, well, and moderately differentiated OSCCs.
Therefore, the study design consisted of 30 patients each, in the four groups of normal mucosa, superficially invasive OSCC, well-differentiated OSCC, and moderately differentiated OSCC. Oesophageal carcinoma tissue served as external positive control.
Two sections (3 μm) of each specimen from all the study groups were prepared. One section was stained with haematoxylin and eosin (H and E) for calculating the DOI of the malignant epithelial cells in the underlying connective tissue in the three study groups of OSCC. The other section was stained immunohistochemically to assess the expression of GLUT-1 protein at the TIF in the three study groups of OSCC.
Depth of invasion
“DOI” means the extent of cancer growth into the tissue beneath an epithelial surface. 12 Depth from the basement membrane is more considerable, since theoretically, lymph node metastasis occurs after the tumour crosses the basement membrane. 13
The TIF has been defined as the most progressed, three to six tumour cell layers or detached tumour cell groups at the advancing edge of the OSCCs. It is often different from the central or superficial part of tumour, as TIF frequently shows a lower degree of differentiation and a higher grade of cellular dissociation in comparison with other parts of the tumour constituting the most aggressive subpopulation of tumour cells that ultimately will invade, spread locally, and metastasize. 14,15 Therefore, this tumour–host interface that provides more adequate prognostic information on tumour “aggressiveness” was taken into consideration.
The H and E stained slides were observed under the research microscope with computer-assisted image analyser to evaluate the depth of the tumour invasion.
In accordance with various studies by Moles et al., 16 Kane et al., 2 and Melchers et al. 17 for calculating the DOI, microscopic tumour depth was taken as the vertical extent of the infiltrative portion of the tumour which extended below from the level of an imaginary line reconstructing the basal membrane of the adjacent normal mucosa to the deepest point of tumour invasion under 4× magnification. A perpendicular line was drawn from these two reference points, and this distance was then measured in millimetres, using the measurement tool on the computer-assisted image analyser.
Immunohistochemical analysis of GLUT-1 antigen at the TIF
Previous reports have demonstrated that the GLUT-1 over expression at the tumour periphery, especially at the invasion front, to be associated with both tumour progression and aggressiveness. 7,10,18
Immunohistochemical staining was performed using the Super Sensitive One-Step Polymer-horseradish peroxidase (HRP) staining method 19 on the tissue sections of all the four study groups and external positive control group.
In brief, formalin-fixed and paraffin-embedded sections were dewaxed with xylene, hydrated with graded alcohol, Ease (EZ) Antigen Retrieval system was used for the antigen retrieval (pH 6). The slides were heated in the microwave oven for 20 min. Then, bench cooled for 20 min and washed three times with Phosphate buffered saline (PBS) for 5 min each. Sections were kept in running tap water for 5 min and then rinsed in distilled water. Then, they were treated with hydrogen peroxide for 10 min at room temperature to block any endogenous peroxidase activity. After rinsing with Tris-buffered saline, the sections were then incubated with power block for 10 min at room temperature to block non-specific immune reactions. Sections were covered with pre-diluted monoclonal primary antibody (Biogenex, Netherland) and incubated in a humidifying chamber at room temperature for 60 min. After super enhancer reagent application for 20 min and buffer wash, poly HRP reagent was added to cover the specimen and incubated for 30 min at room temperature. Subsequently after washing with TBS, the sections were incubated with 3,3 ¢ -diaminobenzidine tetrahydrochloride. Next, after washing in flowing water for 2 min and rinsing with distilled water, all the sections were stained with haematoxylin, dehydrated in graded ethanol and xylene, and finally mounted onto cover glasses. All slides were then examined by light microscopy.
Assessment of immunohistochemical expression of GLUT-1 antigen
Sections stained with anti-GLUT-1 antibody were observed under research microscope with computer-assisted image analyser, to assess its expression in basal layers of normal mucosa and its over expression at TIF in the three study groups of OSCC by evaluation of staining intensity and percentage of GLUT-1 positive cells, according to the method used by Etemad-Moghadam et al. 20,21 Tumour–host interface that provides more adequate prognostic information on tumour “aggressiveness” was taken into consideration. The staining index was evaluated by the average of 10 random fields at the invasion front.

Results
Various cutoff values for DOI have been proposed in the literature with 4 and 5 mm most frequently correlating with the risk of cervical metastasis.
22–24
In the absence of an international consensuses, the cut-off value to be used,
22
a cutoff value of 4 mm for DOI was used in this study and the groups were separated into two subgroups.
OSCC cases with DOI <4 mm. OSCC cases with DOI >4 mm.
Evaluation of GLUT-1 expression in OSCC
Erythrocytes and perineurium within the connective tissue were the internal positive control 7,8,18,25 and known oesophageal SCC samples were used as external positive control 7,8,26,27 showed strong staining intensity in all the cases (Figure 1). One external positive control was included for each immunohistochemical cohort. These positive controls confirmed that the antigenic expression was correctly maintained in the study sample. The absence of non-specific staining in the negative control confirmed the specificity of primary antibody. Immunostaining was observed in the basal layers of normal mucosa and at the TIF of the remaining three study groups of OSCC.

Positive control: (a) perineurium (×4); (b) erythrocytes (×10; red arrow); and (c) oesophageal carcinoma (×4).
The normal mucosa, superficially invasive OSCC, well-differentiated OSCC, and moderately differentiated OSCC demonstrated immunostaining at varying levels for GLUT-1 protein (Figure 2). The GLUT-1 expression occurred predominantly at tumour periphery and it was much weaker in the centre of tumour areas. GLUT-1 expression in tumours was easily apparent as brown staining and occurred in both cytoplasmic and predominantly in membranous pattern of the tumour cells (Figure 2(d) and (f)).

Immunohistochemical profile. (a) Group 1 (×4, red arrow: erythrocytes showing strong staining index); (b) group II showed homogenous low staining index at TIF (×4); (c: ×4) and (d: ×10 TIF) group III showed moderate GLUT-1 staining index; (e: ×4) and (f: ×10 TIF) group III showing strong GLUT-1 staining index at TIF; (g: ×4) and (h: ×10 TIF) group IV showed strong staining index at TIF. GLUT-1: glucose transporter.
Correlation of GLUT-1 expression and DOI in the histological grades of OSCC
In the present study, we examined the potential correlation between GLUT-1 expressions at the TIF with the corresponding increase in DOI, as the histological grade progressed from superficially invasive to well-differentiated to moderately differentiated OSCC.
In order to study the changes in GLUT-1 expression at TIF in combination with DOI in the progressive histological grades of OSCC, mechanistic study, that is, receiver operating characteristic (ROC) curve analysis was done. Sensitivity and specificity of two parameters were derived. Sensitivity (
Discussion
Neck metastasis is the most important prognostic factor in head and neck SCCs. 28–31 While considering all stages of OSCC, occult nodal metastases are present in up to 50% of cases. 17,32,33 Therefore, an elective neck dissection is generally considered in cN0 patients when the risk for occult metastasis is considered greater than 20%. Despite these additional criteria, still 20–30% of the cN0 patients considered low risk, and consequently those who are not treated with a neck dissection, develop metastases during follow-up. 17,34
Research in head–neck oncology has focused on finding additional histological predictors for the presence of occult nodal metastases, such as lymphovascular invasion, 17,35 intra-tumoural vessel density, 17,36,37 presence of various immunohistochemical or molecular markers, 17,38 and tumour infiltration depth. 2,3,7,12,13,16
As the histological grade progresses, the proximity to blood vessels and lymphatic’s with increasing DOI is what determines an increased risk of nodal metastases developing in as much as it facilitates the tumour’s ability to expand. 12 DOI is related to the more invasive behaviour of the deeply infiltrating cancer, which may promote the entry of cancer cells into the lymphatics. 39 Currently, many studies 2,17,23,24,40–42 have demonstrated a good correlation between depth of tumour invasion and the presence of occult lymph node metastasis, prompting many institutions to adopt DOI as an indicator for necessity of lymph node dissection.
While other parameters are not only qualitative (and therefore, likely to suffer a bias due to subjectivity) but are also liable to vary from one histological section to the other. In contrast, the maximum tumour depth and thickness are quantifiable measurements and are more likely to be precise and accurate. 2
The data from the present study (Table 1) revealed that there was statistically significant increase in the number of cases with DOI more than 4 mm as the histological grade progressed from superficially invasive to well-differentiated to moderately differentiated OSCC indicating it to be associated closely with disease progression (Figure 3). Our results are consistent with the previous study findings. 2,17,23,24,40–42
Comparison of DOI among the study groups II–IV.
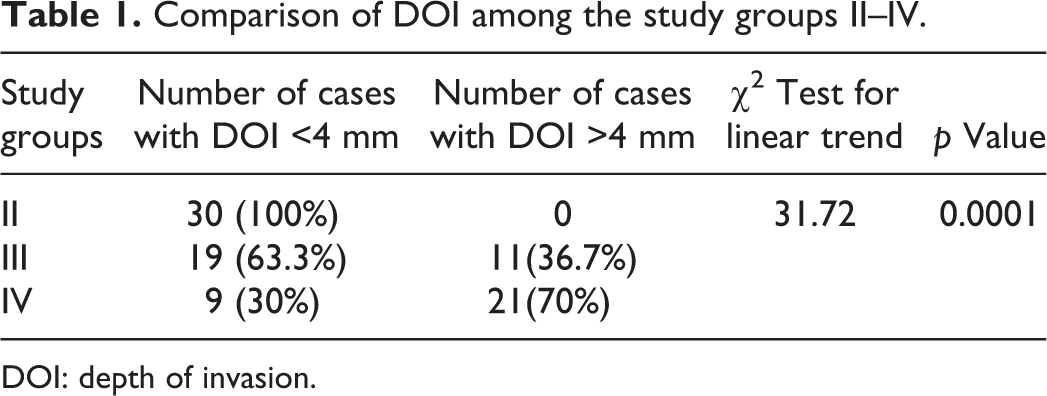
DOI: depth of invasion.

DOI measured on H and E stained section (×4 magnification). (a) Group II: DOI less than 4 mm; (b) group III: DOI less than 4 mm; (c) group III: DOI more than 4 mm; and (d) group IV: DOI more than 4 mm. DOI: depth of invasion; H and E: haematoxylin and eosin.
In the present study, 28 cases (93.3%) of normal mucosa showed focal to homogenous GLUT-1 expression confined to the basal compartment and at places in the first suprabasal cell layer (Figure 2(a)). These findings are in line with the differentiation-related expression pattern of GLUT-1 in normal human epidermis, in which GLUT-1 is expressed in the basal layer and, to a lesser extent, in the immediate suprabasal layer of the epithelium. 11
GLUT-1 expression in all the three study groups (Table 2) was consistently expressed at a measurable level in a higher percentage compared to normal mucosa (Figure 2). The GLUT-1 expression occurred predominantly at tumour periphery and it was much weaker in the centre of tumour areas. This result demonstrated that tumour cells needed much glucose as an energy source in deep invasion areas with hypoxia in comparison with wide and shallow invasion areas in the central parts of tumour. We therefore hypothesized that the GLUT-1 expression in the tumour periphery, especially at the invasion front, was associated with further progression and aggressiveness of tumour. GLUT-1 became significantly stronger when the tumour demonstrated deep invasion, since a most intense degree of hypoxia is reached in the deepest parts of tumour at TIF.
Comparison of GLUT-1 Immunohistochemistry (IHC) staining index among the four groups.

GLUT-1: glucose transporter.
It should be noted that an increase in the immunoreactivity of GLUT-1 and the membranous staining pattern have been reported as indicators of hypoxia 43,44 as its biological function is most likely related to localization at the cell membrane. The significance of GLUT-1 protein accumulation in this study appears to be as a marker of hypoxia, as the membranous GLUT-1 antibody expression was strongest at TIF (Figure 2(d) and (f)).
According to the results obtained in our study, there was consistent increase in staining index of GLUT-1 at the TIF in the three progressive histological grades of OSCC compared to the normal mucosa in group I, which was suggestive of its potential role in predicting both tumour aggressiveness and prognosis. Our findings were in concordance with those of the earlier studies. 5,7,8,10,18,25–27,45–48
Invasive tumour depth is associated with intense degree of hypoxia. GLUT-1, being an endogenous marker of hypoxia, is over expressed at the TIF, not only permit cancer cells to survive under adverse conditions such as hypoxia but enable their proliferation, progression, invasiveness, and subsequent distant metastasis. 10
In the present study (Table 3), we examined the potential correlation between progressive changes in GLUT-1 expression at the TIF with the corresponding increase in the DOI, as the histological grade progressed from superficially invasive to well-differentiated to moderately differentiated OSCC.
Correlation of GLUT-1 staining index at the TIF with DOI in all the three study group cases of OSCC.

DOI: depth of invasion; OSCC: oral squamous cell carcinoma; GLUT-1: glucose transporter; TIF: tumour invasive front.
It was observed that the staining index of GLUT-1 at the deep invasive front became stronger with the progressive histological grades of tumour, with corresponding increase in the DOI. In other words, the expression of GLUT-1 became significantly stronger when the tumour demonstrated deep invasion (Figure 4).

Correlation of GLUT-1 staining index at the TIF with DOI in study group cases of OSCC. (a), (b) and (c) Group II: low GLUT-1 staining index with DOI less than 4 mm; (d) and (e) group III: moderate GLUT-1 staining index with DOI less than 4 mm; (f) and (g) group III: strong GLUT-1 staining index with DOI more than 4 mm; (h) and (i) group IV: strong GLUT-1 staining index with DOI more than 4 mm. DOI: depth of invasion; GLUT-1: glucose transporter; TIF: tumour invasive front.
In few cases, as the histological grade progressed, some of the tumours with strong GLUT-1 expression at the invasion front showed DOI to be <4 mm. Insufficient depth of incisional biopsy specimen in these higher histological grade cases may have resulted in an artefactually diminished measurement of DOI, indicating GLUT-1 expression to be an independent negative marker in the prognosis as its expression was closely associated with disease progression.
Correlation between GLUT-1 expression and tumour depth may be indicative of the relationship existing among poor prognosis, hypoxia, and GLUT-1 expression, since a most intense degree of hypoxia is reached in deepest parts of tumours.
Our findings are in accordance with the study conducted by Ohba et al. on 24 cases of OSCC in which they concluded that GLUT-1 served as a marker indicating that tumours with deep invasion (more than 4 mm) tend to result in a worse prognosis in patients due to either lymph node metastasis, a recurrence of the primary lesion or distant metastasis. 7
The data from the present study is consistent with the previous findings that there was statistically significant increase in DOI in progressive histological grades of tumour, indicating its expression to be associated closely with disease progression. In addition, there was consistent increase in staining index of GLUT-1 at the TIF which was significantly stronger when tumour demonstrated deeper invasion with progressive increase in the histological grades of OSCC. Mechanistic correlation (Figure 5) to study together both parameters, that is, DOI and GLUT-1 expression, was evident by statistically significant ROC curve, thereby giving evidence of the accuracy of the curve.
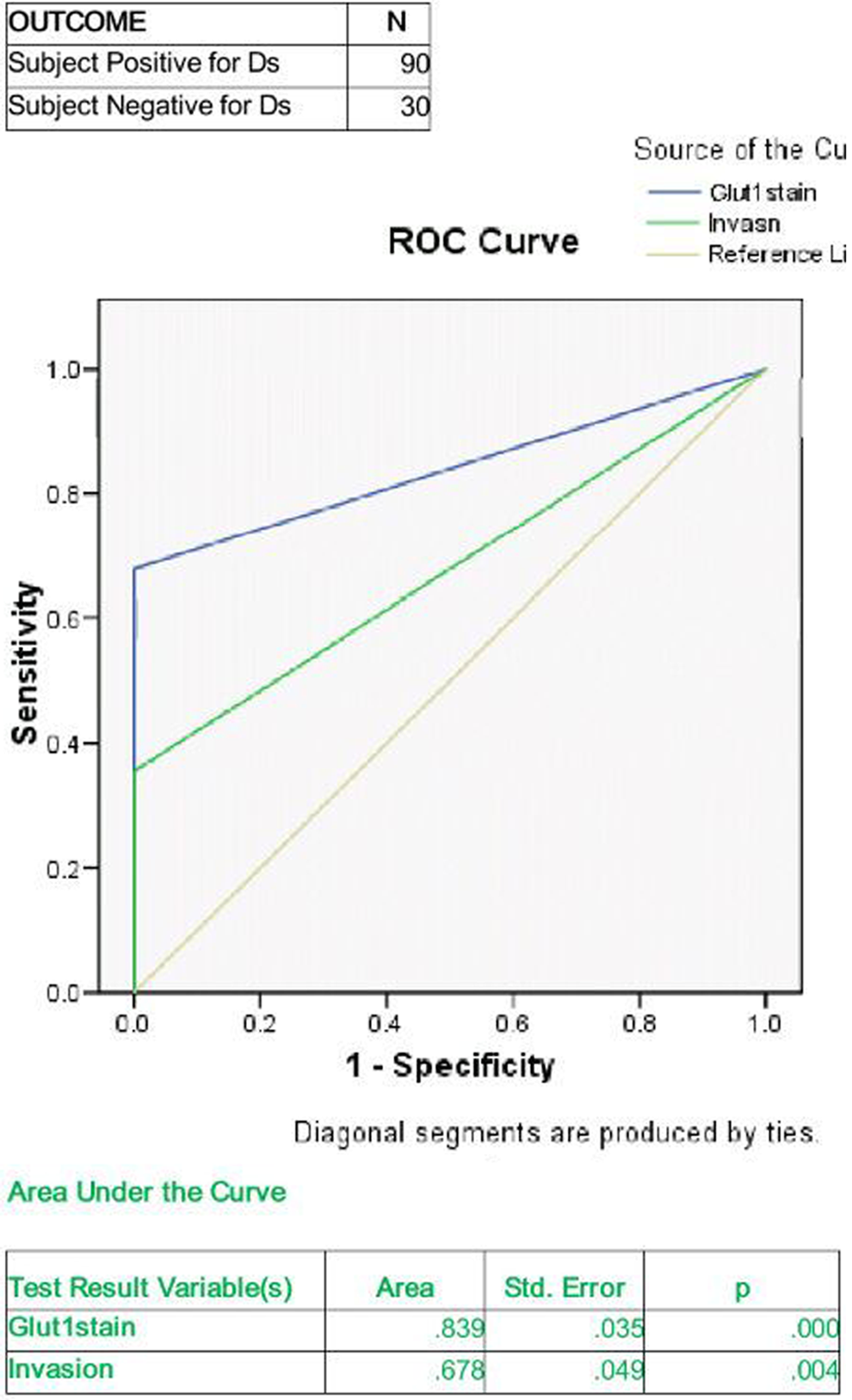
ROC curve analysis: An area under the ROC curve of 0.839 means that a randomly selected case from the groups have chance of being tested correctly 80% of the time for GLUT-1 stain indicating good balance of sensitivity and specificity. Area under the curve for invasion of 67% shows fair balance of sensitivity and specificity. Blue line indicate the readings of the test for GLUT-1 stain while the green line indicates the readings for invasion and grey indicates line with zero sensitivity and zero specificity area. ROC: receiver operating characteristic; GLUT-1: glucose transporter.
Various studies have also studied the relationship data between GLUT-1 expressions associated with reduced survival time. 7,18,25–27 The limitations of our study are its retrospective nature and lack of survival data. Both these parameters further need to be coupled with survival analysis.
It has been suggested that the expression pattern of both DOI and GLUT-1 over expression at TIF may be used as a prognostic factor of malignancy progression, possibly assisting in the selection of patients requiring more aggressive therapy.
Clarification of the role of glucose uptake and GLUTs in cancer could potentially lead to effective modes of early tumour detection and treatment. A disruption of glucose uptake via GLUT proteins may alter the metabolism of malignant cells, leading to reduced tumour growth. A first approach could be the blockade of signalling components altered in cancer that enhance the expression of sugar transporters. Several selective GLUT inhibitors, such as fisetin or apigenin, are selective agents that act by blocking the glucose uptake from tumour cells and avoid the activation of the signalling pathway. Furthermore, inhibitors of glycolysis or glycolytic pathways are being searched to identify therapeutic agents that can inhibit enzymes that leads to cancer growth and development. For example, 3-bromopyruvate, a highly active alkylating agent, was reported to target HK-2 and/or GAPDH. As chemosensitivity is enhanced by counteracting the acidification of tumour’s microenvironment,inhibitors of glycolytic enzymes may also impose an alkalizing effect in tumour’s microenvironment particularly at the tumour tissue level. This may have a more specific and powerful role in enhancing the sensitivity of tumour cells to basic chemotherapy drugs. 5,49
Conclusion
Not only a complete resection of the primary lesion but also the control of both lymph node metastasis and distant metastasis is important, to improve the therapy for OSCC. If metastasis could be recognized in advance, improvement of the prognosis might be possible through elective neck dissection. 42
DOI and GLUT-1 over expression at TIF are the two parameters that were investigated separately in a variety of tumours in many previous studies and each has been assigned a prognostic significance. Our study is among those few studies in which both parameters were used simultaneously to investigate their correlation in the progressive histological grades of OSCC, which was found to be highly statistically significant.
To conclude, these two parameters used simultaneously provide baseline by highlighting the cases that need further lymph node assessment, thereby having significant prognostic value in predicting their risk of occult metastasis.
This would probably help in removing the hurdle in the effective management of clinically negative neck in early SCC (T1–T2/NO) of oral cavity and could be a deciding factor for elective neck dissection which would further improve the survival rates of OSCC. Both intensive treatment and a careful follow-up were therefore considered to be necessary when a high GLUT-1 expression is observed in the invasion front of the tumour.
To improve the predictive potential further and to provide additional strong evidence for the biologic and clinical relevance of glucose transport and metabolism in tumour progression, both these parameters need to be correlated with lymph node status by further survival analysis and [18F]-2-fluoro-2-deoxy-D-glucose- Positron Emission Tomography (FDG-PET) scan or other recent diagnostic measures in future.
Footnotes
Acknowledgements
The authors would like to thank the staff members of Department of Oral Pathology and Microbiology, Government Dental College and Hospital, Nagpur, Maharashtra, India, for their valuable contribution and M Chopade for his technical assistance in the work of immunohistochemistry.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Department of Oral Pathology and Microbiology, Government Dental College and Hospital, Nagpur.
