Abstract
The aim of this study was to investigate the prevalence of fatigue among lung recipients 1–5 years after transplantation (LuTx) and to explore its relationship to distressing symptoms such as dyspnea, sleep problems, pain, and decreased appetite, as well as psychological and transplant specific wellbeing. A cross-sectional, multicenter cohort study was conducted. A total of 117 lung recipients with a mean age of 54 years (SD 13 years) who were due for their annual follow-up were included at 1 year (n = 35), 2 years (n = 28), 3 years (n = 23), 4 years (n = 20), and 5 years (n = 11) after lung transplantation. Several instruments were used: the Multidimensional Fatigue Inventory, the Organ Transplant and Symptom Well-being Instrument (OTSWI), the Pain-O-Meter, and the Psychological General Well-Being Index. Overall 56% reported high or severe fatigue. The Psychological General Well-Being sum score makes the strongest contribution (p < .0001) to explaining the variance in general fatigue (Beta –.628). The results of the study indicate that efforts both to identify fatigue and to test interventions are needed after LuTx.
Introduction
The rationale behind this study is that no comprehensive analysis of fatigue after lung transplantation (LuTx) has been performed. As self-management and health-related quality of life (QoL) are important outcomes after lung transplantation, it is essential to explore the nature of fatigue and its possible impact on lung recipients’ wellbeing. A key concern for nurses in transplant care is how to change and support health behaviors to engage lung recipients in the extensive task of self-management and in following recommendations from healthcare professionals. Symptom management is one important aspect of self-management after lung transplantation. How the lung transplant recipient perceives the recovery process and what they report as their main concern is fundamental in developing self-management support. Symptoms during the recovery process, psychological distress and decreased wellbeing are all potential threats to adherence to prescribed recommendations, but foremost to health and wellbeing. Therefore, an increased understanding of the possible symptom burden is vital in promoting long-term health outcomes after lung transplantation. This articles narrows the knowledge-gap regarding how wellbeing is affected by fatigue and related symptoms and thus contributes to the health-promotion efforts of LuTx nurses. Thus, the aim of this study was to investigate the prevalence of fatigue among lung recipients 1–5 years after transplantation and to explore its relationship to distressing symptoms such as dyspnea, sleep problems, pain, and decreased appetite, as well as psychological and transplant-specific wellbeing.
Background
Fatigue, a multifaceted symptom characterized by the sense of exhaustion, lethargy, and distress, has been explored among other solid organ recipients, mainly liver recipients. Liver transplantation (LTx) is associated with improvement in fatigue in patients with primary biliary cirrhosis (PBC). However, a substantial proportion of patients continue to suffer from significant fatigue after 2 years. 1 Patients with an alcoholic or cryptogenic cirrhosis history had significantly poorer physical function and experienced more physical fatigue than liver recipients with other diagnoses. 2 Fatigue is also common among patients with liver cirrhosis and associated with impaired QoL. 3 Psychological distress, severity of cirrhosis, and low cortisol level determined general fatigue. Anemia and impaired renal function also contributed to physical fatigue and remain of concern after LTx due to cirrhosis. In total, 76% experienced high fatigue severity and 77% had poor sleep quality after LTx. 4 Fatigue predicted daily function and all health-related QoL domains. Sleep quality, anxiety, and depression were associated with fatigue severity. 5 Van den Berg-Emons et al. concluded that fatigue is a major problem after LTx. 6
Fatigue affects the activities of daily living after kidney transplantation (KTx), reducing the ability to perform activities. 7 Several psychological and behavioral factors, including negative mood, low QoL, napping, and caffeine consumption, are related to sleep disturbance among pre- and post-KTx patients. 8 Fatigue was found in 59% of the kidney recipients although only 13% had this symptom listed in their medical records. 9 Furthermore, in another study, physical fatigue negatively impacted QoL among 22% of kidney recipients. 10 High Body Mass Index (BMI), poor sleep quality, and mood disturbance were predictors of fatigue after KTx. The 39% of respondents reporting severe fatigue after KTx had more functional impairments as opposed to non-severely fatigued recipients, in addition to higher BMI, more pain, less social support, depressive symptoms, and sleep problems. 11 No differences in fatigue were found when comparing living donor kidney recipients with deceased donor kidney recipients as measured using The Multidimensional Fatigue Inventory (MFI-20). 12
Fatigue is rarely explored after heart transplantation (HTx) but according to Reyes, Evangelista, Doering et al., 13 fatigue is common among female heart recipients and associated with both physiological and psychological factors. Anemia, functional status, and depression were significant predictors of fatigue, explaining 69% of the variance. 13 Finally, Chou et al. reported that after HTx, fatigue interference had a greater influence on QoL domains than fatigue intensity. 14 Organ transplant recipients experience symptom distress and transplant nurses have a great task when promoting health and supporting symptom management. This study is the first to comprehensively explore fatigue 1–5 years after LuTx to answer the question: What is the prevalence and impact of fatigue up to 5 years after lung transplantation?
Design
Study participants’ diagnosis leading to lung transplantation, type of transplantation and immunosuppressive medication.
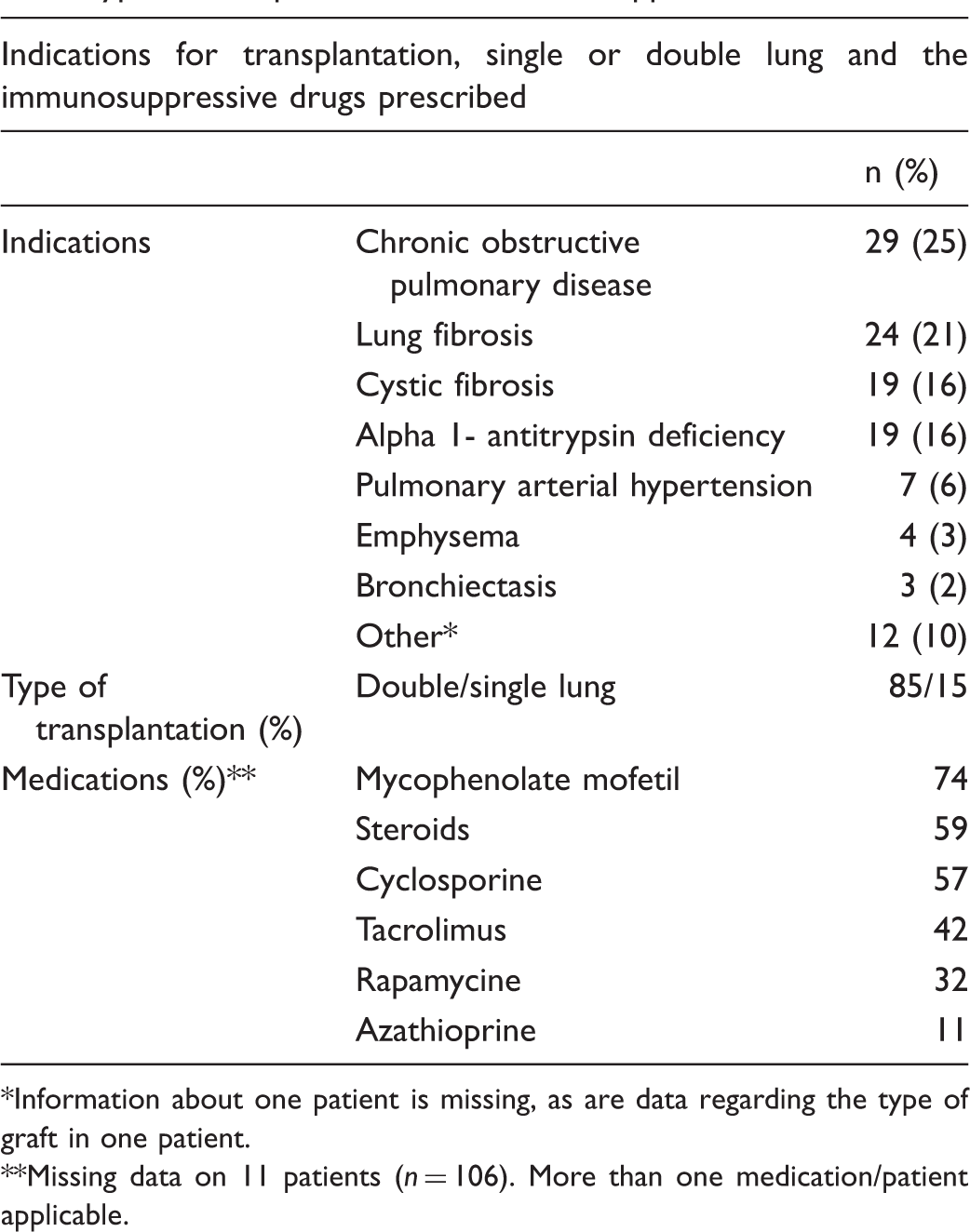
Information about one patient is missing, as are data regarding the type of graft in one patient.
Missing data on 11 patients (n = 106). More than one medication/patient applicable.
Demographic characteristics.
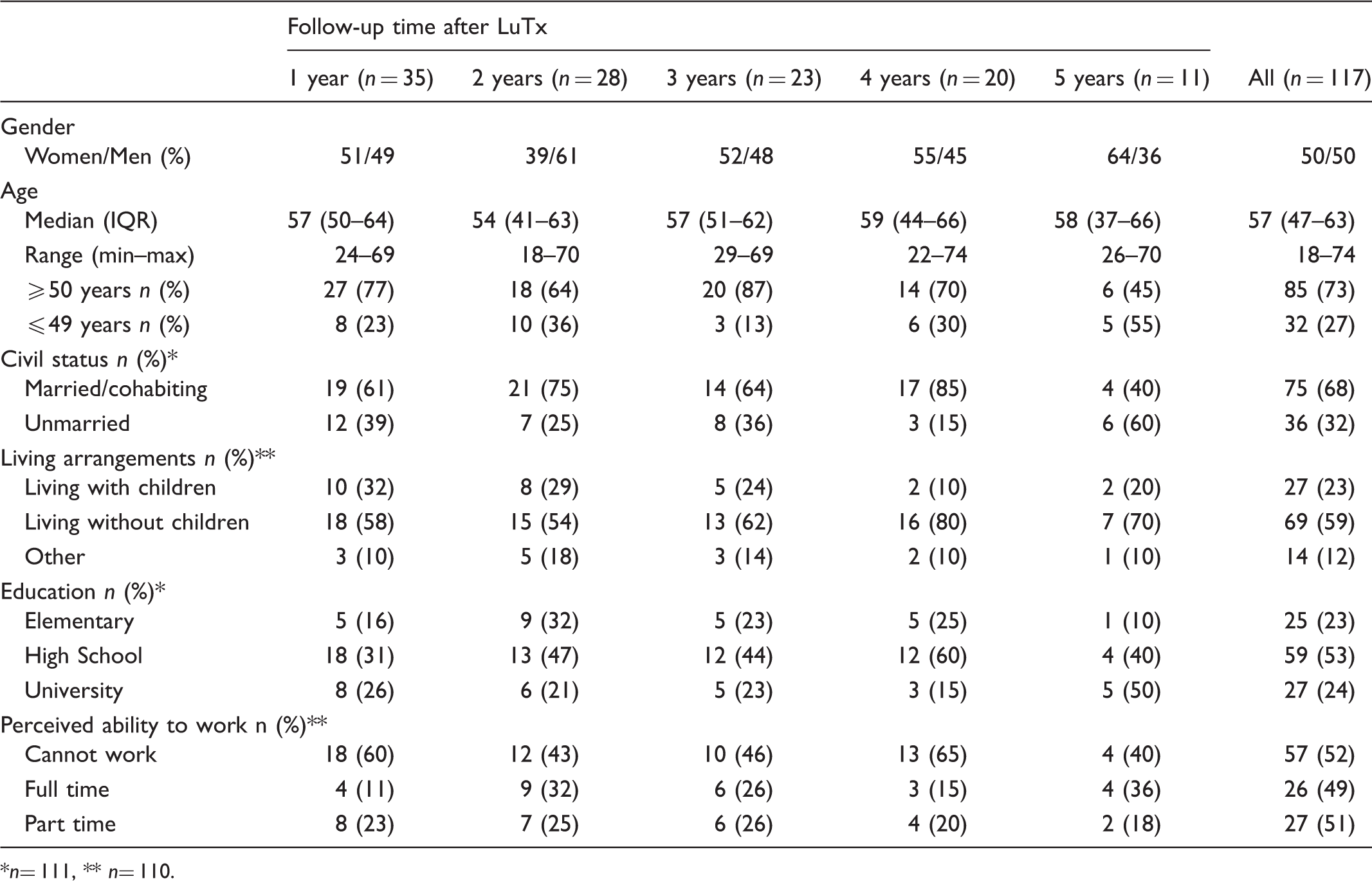
n= 111, ** n= 110.
Methods
The Multidimensional Fatigue Inventory (MFI-20) measures five dimensions of fatigue, i.e. general fatigue, physical fatigue, reduced activity, reduced motivation, and mental fatigue. 15 A 19-item version was used in line with the results of the Swedish validation of the instrument. 16 Fatigue in the preceding days is rated on a five-point Likert scale from ‘yes, that is accurate’ to ‘no, that is not accurate’. Subscale scores range from 4 to 20, where a high score represents greater fatigue. Cronbach’s alpha for the various subscales was > 0.70. Based on previous studies,17,18 scores in the general fatigue subscale were grouped (4–11, 12–15, 16–20) and served as a cut-off for low, high, and severe fatigue. Fatigue was also measured with the fatigue subscale in the Organ Transplant Symptom and Well-being Instrument (OTSWI-F) assessed on a five-point Likert scale from 0 to 4: ‘not at all’ = 0, ‘a little’ = 1, ‘somewhat’ = 2, ‘quite a bit’ = 3, and ‘very much’ = 4. 19 The three-item subscale score for fatigue ranges from 0 to 12. The OTSWI also contains 20 single items with the same response scale, which measure distress from 20 transplant specific symptoms.
For this study the single items dyspnoea and decreased appetite were used. 19 Pain assessment was performed by means of the Pain-O-Meter (POM).20,21 The POM provides information about pain intensity, sensation, location, and duration. It consists of a 10 cm straight line to indicate pain intensity, the POM-VAS. For each pain location the lung recipients were asked to indicate the intensity on the POM-VAS. A total VAS-score for each pain location was calculated. The minimum VAS-score is 0, while the maximum depends on the number of reported pain locations. There is also a list of sensory and affective words, POM-WDS. All affective POM-WDS in the Swedish version of the instrument were used: agonizing, annoying, troublesome, tiring, terrifying, miserable, sickening, nagging, killing, unbearable, and torturing. Each word has an assigned intensity value (range 1–5). The sensory and affective scores are added together to form a total POM-WDS pain intensity score (PIS) illustrating the total burden of pain, where the minimum is 0 and the maximum depends on the number of sensory and affective words reported for each pain location. This instrument has undergone reliability and validity testing in different patient populations and has been used among organ recipients. 22
The Swedish version of the Psychological General Well-Being (PGWB) instrument was used to measure psychological wellbeing and distress. 23 The instrument contains 22 items that constitute six dimensions; anxiety, depressed mood, positive wellbeing, self-control, general health, and vitality. Inter-item correlation values range from 0.53 to 0.79 and Cronbach’s alpha ranges from 0.61 to 0.89. The PGWB total sum score is 132, with higher scores indicating better health status and psychological wellbeing. A normal sum score is 100–105, and women tend to report lower wellbeing than men. 24 The sum of scores was dichotomized into low scores (0–100) and respectively good scores (101–132) indicating physiological wellbeing
Statistical analysis and ethical considerations
The hypotheses tested regarding fatigue, concurrent symptoms and wellbeing.
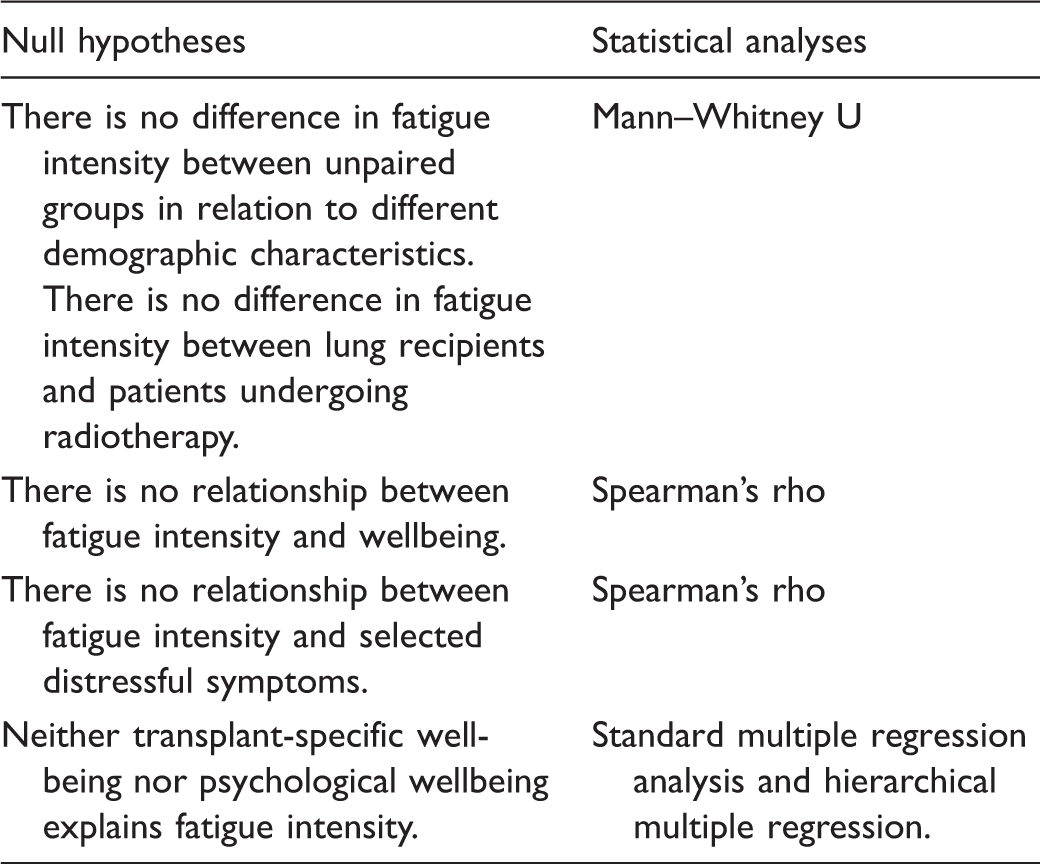
When applicable, values of p < .05 (two-tailed) were considered statistically significant. Due to the small sample sizes at each follow-up, the correlations between fatigue, selected symptoms, and wellbeing included all patients, regardless of the time since their LuTx. The stepwise analysis was performed as follows:
Exploration of proportions and intensity of fatigue in all recipients related to the number of years from the time of LuTx. Exploration of intensity of fatigue in relation to selected demographic characteristics. Exploration of possible differences in fatigue between two unpaired groups, e.g. men and women, and lung recipients in addition to patients treated for malignancy. Exploration of intensity of fatigue and associations with selected symptoms, transplant specific and psychological wellbeing. Analysis of possible explanatory factors.
Regarding the regression analysis, when checking for multicollinearity we decided not to remove the independent variable PGWB sum even if it was > .70. The tolerance of .38 was good, and the variance inflation factor (VIF) was 2.63, in other words below 10. In addition, Cook’s distance was < 1. The normal probability plot lay in a reasonably straight diagonal line from bottom left to top right, suggesting no major deviation from the norm. Finally, the distribution of the scatterplot was roughly rectangular.
A cohort of Swedish patients (mean age 68 years, 68% women and 32% men) undergoing radiotherapy for different malignancies was used as a comparison group. This cohort was chosen because such patients experience fatigue and several concurrent symptoms during treatment, as well as uncertainty in relation to survival. The cancer cohort is a part of one of the authors’ published thesis. Permission to carry out this study was granted by the Regional Ethical Review Board in southern Sweden (D-nr 2014-124).
Results
The magnitude of fatigue 1–5 years after lung transplantation
Fatigue in lung recipients at different follow-up times after transplantation.
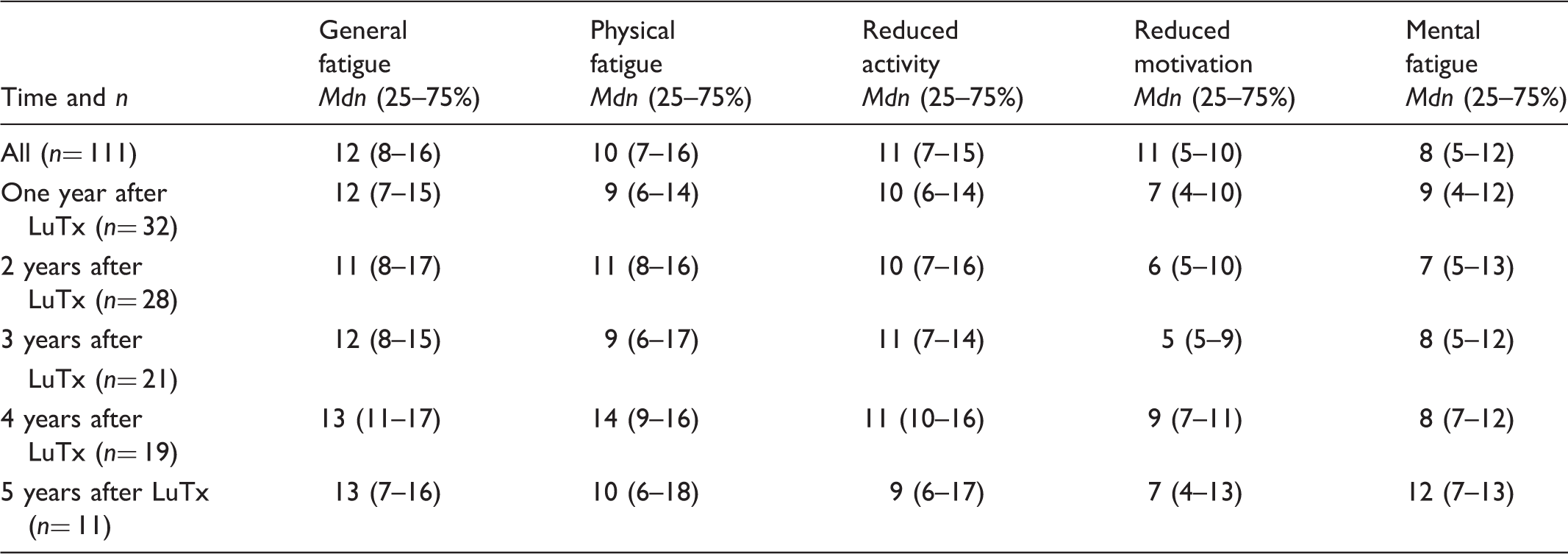
A total of 56% reported high or severe fatigue. The proportion of recipients with severe fatigue ranged from 22% to 45% depending on follow-up time (Figure 1). Fatigue measured with the OTSWI-F subscale had a median value of 3 (IQR 1–6).
Proportions of lung recipients (%) with different intensity of fatigue at different time-points after transplantation. Groups based on general fatigue scores in the MFI-20, low (<12), high (12–15) or severe (16–20) intensity of fatigue.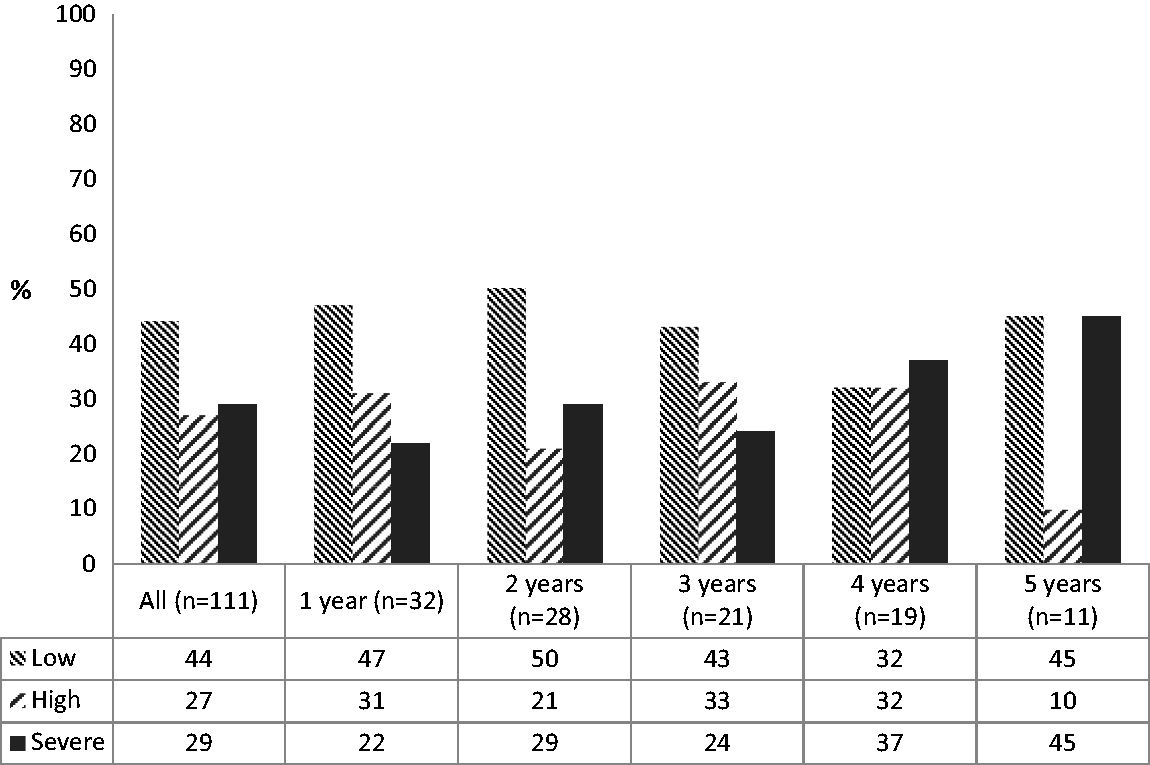
Median levels of fatigue based on demographic characteristics.
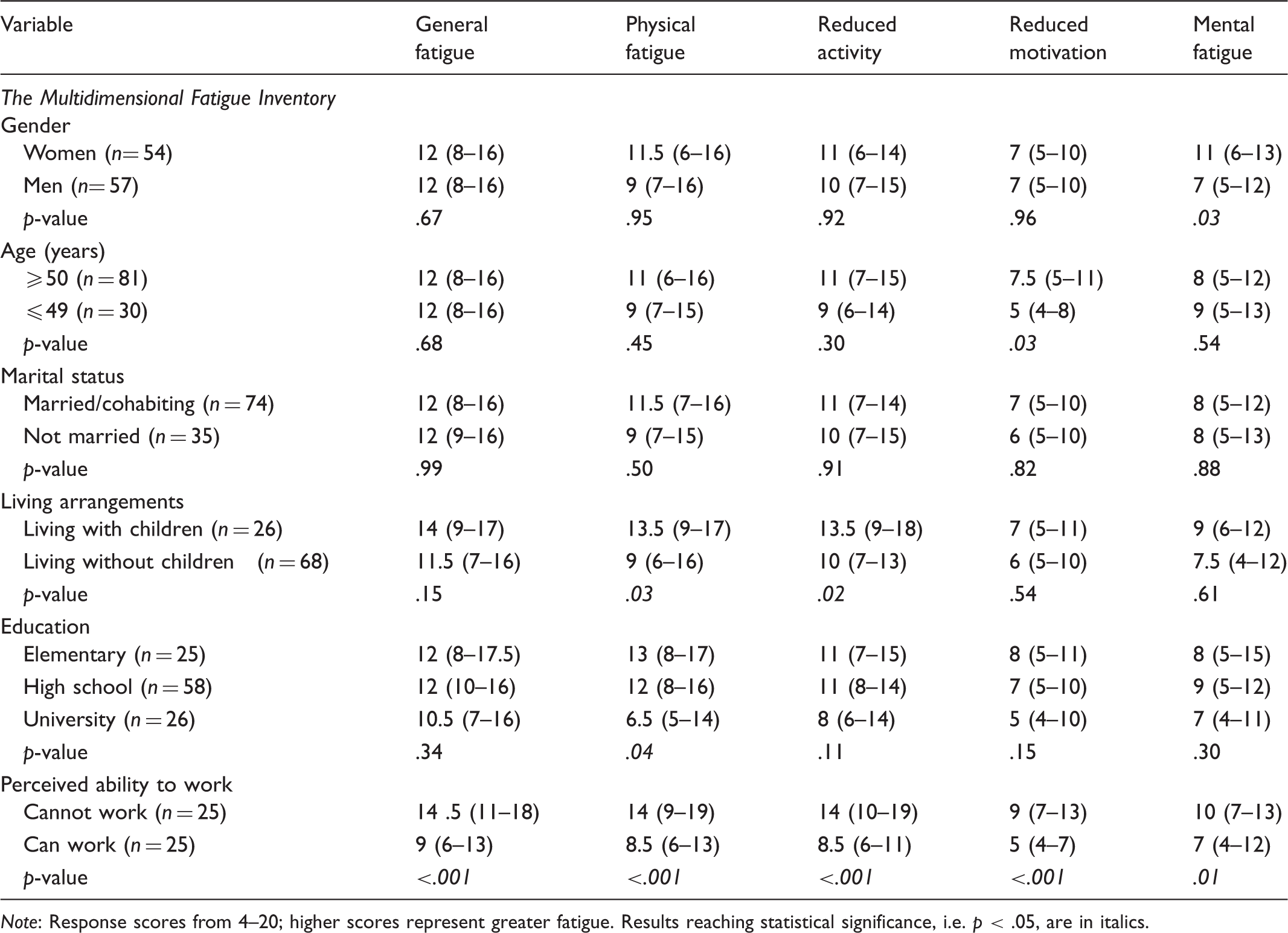
Note: Response scores from 4–20; higher scores represent greater fatigue. Results reaching statistical significance, i.e. p < .05, are in italics.
As illustrated in Figure 2, lung recipients reported significantly lower fatigue intensity than patients undergoing radiotherapy.
Comparisons in median scores of MFI-20 subscales between lung recipients and patients with abdominal cancer undergoing radiotherapy.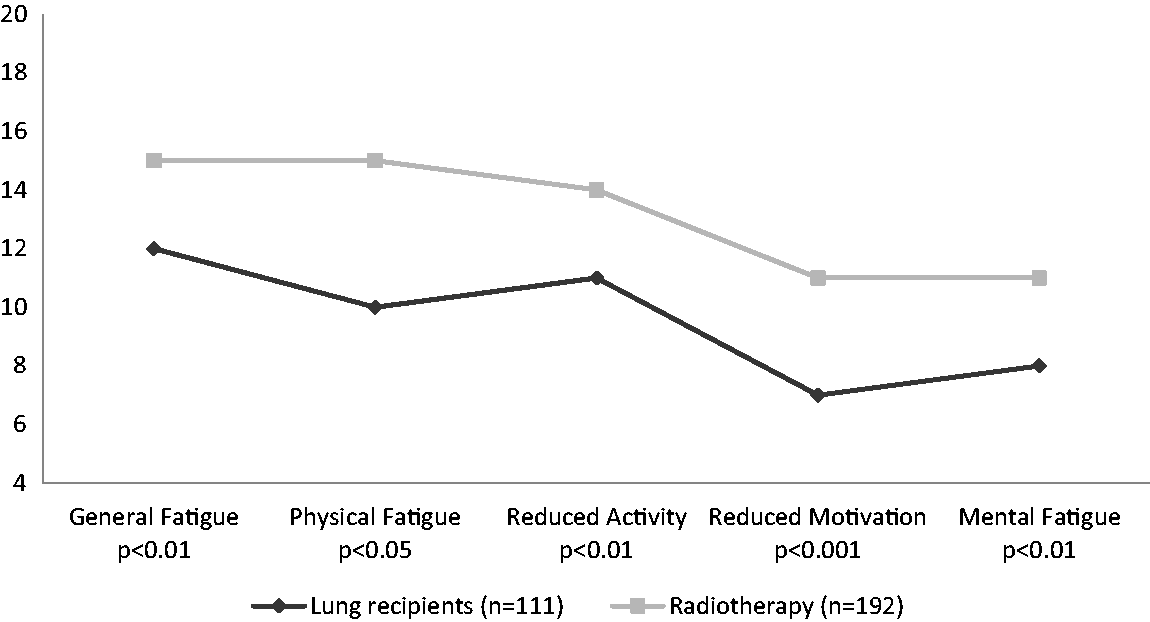
Fatigue and distressing symptoms
Lung recipients with pain were not more fatigued than those without pain. There was a relationship between dyspnea and all dimensions of fatigue; general fatigue (.670), physical fatigue (.679), reduced activity (.559), reduced motivation (.497), and mental fatigue (.353). Dyspnea when resting was also related to all fatigue dimensions: general fatigue (.590), physical fatigue (.710), reduced activity (.566), reduced motivation (.501), and mental fatigue (.354). The relationship between pain and all five fatigue dimensions was weak or moderate with correlation rs values below 0.4. The correlation between sleep problems and general fatigue was .528 as well as between decreased appetite and fatigue (.393). There were no relationships between fatigue and the following variables: time on ventilator, time in the ICU, length of hospital stay, and number of rejections.
Fatigue and wellbeing
Patients with low psychological wellbeing (46%, based on a cut-off score of 100 on the PGBW) had higher median levels in all fatigue dimensions (p < .001). The correlation between general fatigue and overall transplant-specific wellbeing (OTSWI sum) was rs .749 with p < .001. The correlation between general fatigue and the PGWB sum was rs–.818 with p < .001.
Possible explanatory factors
The regression model explained 63.6% of the variance in general fatigue (p < .0001) (R2 = .636). The PGWB sum makes the strongest unique contribution (p < .0001) to explaining the variance in general fatigue (beta .628, in contrast to the OTSWI sum: beta .203, p = .034). Hierarchical multiple regression was used to assess the ability of the PGWB sum and the OTSWI sum to predict perceived general fatigue levels, after controlling for the influence of age and sex. Preliminary analyses were conducted to ensure no violation of the assumptions of normality, linearity, multicollinearity, and homoscedasticity. In step one, age and sex explained 0.2% of the variance in perceived general fatigue. After addition of the two scales in Step 2, the total variance explained by the model as a whole was 64.3% when age and sex were statistically controlled for (R2 = .643, F change = 95.147, p < .001). In the final model two control measures were significant, with the PGWB sum recording a higher beta value (beta = .628, p < .001) than the OTSWI sum (beta = .203, p = .034). The design did not permit exploring causation, so we reported which factor explained the variance in the regression model where the direction of the relationship is as follows: the higher fatigue score the lower score on the PGWB. A high fatigue score suggests severe fatigue while a low score on the PGWB suggests poor psychological wellbeing.
Discussion
The key findings in this study were that 56% reported high or severe levels of fatigue, regardless of the number of years since
As with liver recipients, the PGWB determines fatigue after LuTx. 3 Our sample was found to suffer less from fatigue than other cohorts of both liver recipients 4 and kidney recipients, where this kind of problem was found in 59%. 8 Anemia, functional status, and depression were significant predictors of fatigue, explaining 69% of the variance after HTx, 13 which supports our results that psychological wellbeing is a strong predictor of fatigue, indicating the need for interventions to enhance mental health.
At the 4 and 5-year follow-ups patients report high levels of general fatigue. The peak severity of fatigue at these time-points was also evident when distinguishing between different dimensions of fatigue. When analyzing other aspects in the SMATT study, e.g. pain, the severity of pain burden after LuTx was also most profound at the 4-year follow-up, suggesting an overall peak in perceived illness at that time.25,26 However, the association between fatigue and pain was not as evident.
Cancer patients undergo demanding treatment and must cope with uncertainty regarding life expectancy in the same way as lung recipients. We were pleased to note that lung recipients were less fatigued in all five dimensions compared to patients undergoing radiotherapy. The difference in median values was most evident in physical fatigue. Not surprisingly, lung recipients who reported inability to work scored higher on all fatigue dimensions than those working full or part time. Therefore it reasonable to believe that an intervention decreasing fatigue might improve the ability to return to work. As expected, there was a strong relationship between fatigue and shortness of breath as well as between fatigue and sleep problems in the whole group. Women reported more mental fatigue than men, but due to the study design no firm conclusion can be drawn from this. However, it is known that in general women tend to report more symptoms and worse symptom burden than men after organ transplantation. 19 Thus, interventions should focus on how to handle and reduce mental fatigue in female lung recipients.
In line with Talkawar, 27 our study highlights the importance of identifying appropriate interventions targeting fatigue after LuTx. A 12-week rehabilitation program proved effective after liver transplantation as the participants were less fatigued after the intervention. 28 Furthermore, cardiorespiratory fitness was the only fitness component which related to the severity of fatigue after liver transplantation. 29 Reviews on fatigue interventions among cancer patients show that combined exercise and psychological procedures are most effective and that interventions based on pharmaceutical treatment and education are insufficient.30,31 We argue that increased efforts to both identify fatigue and test interventions are needed after LuTx.
In conclusion, high or severe fatigue is common 1–5 years after LuTx but may vary depending on follow-up time. Poor psychological general wellbeing is a strong contributor to fatigue, indicating that efforts to improve lung recipients’ psychological wellbeing could decrease their fatigue levels. This is a new target area for health promotion by transplant nurses for lung transplant recipients.
Limitations
The cross-sectional study design and small patient groups at each time-point after LuTx hindered analysis of variation in fatigue levels between different years of follow-up. For that reason, a longitudinal study is ongoing. In addition, all measurements were self-reported and not supported by blood tests or physiological performance tests. It is not optimal if the correlation between the two independent variables is too high as in this case the correlation between psychological general wellbeing (PGWB) and transplant-specific wellbeing (OTSWI) was > .70. The ideal would be a correlation less than .70. However, these instruments measure various factors and are developed in two different contexts. Yet they are probably overlapping in their overall sum score in some way and thus the findings from the regression analysis should be interpreted with caution.
Footnotes
Author contributions
Anna Forsberg: design, data analysis, drafting article, statistics and securing funding. Annette Lennerling: data collection and critical revision of the article. Annika Kisch: critical revision of the article. Sofie Jakobsson: data analysis, drafting the article, statistics.
Acknowledgements
We acknowledge Ulrika Nibble, Elisabeth Svebring, Marie Stiernspetz and Kristine Kappelin at the two transplant units who facilitated the data collection.
Funding
This work was supported by the Thure Carlsson foundation.
Conflict of interest
The authors declare that there is no conflict of interest.
