Abstract
Objective
To investigate the association between driving pressure (ΔP) and 90-day mortality in patients following lung transplantation (LTx) in patients who developed primary graft dysfunction (PGD).
Methods
This prospective, observational study involved consecutive patients who, following LTx, were admitted to our intensive care unit (ICU) from January 2022 to January 2023. Patients were separated into two groups according to ΔP at time of admission (i.e., low, ≤15 cmH2O or high, >15 cmH2O). Postoperative outcomes were compared between groups.
Results
In total, 104 patients were involved in the study, and of these, 69 were included in the low ΔP group and 35 in the high ΔP group. Kaplan-Meier analysis of 90-day mortality showed a statistically significant difference between groups with survival better in the low ΔP group compared with the high ΔP group. According to Cox proportional regression model, the variables independently associated with 90-day mortality were ΔP and pneumonia. Significantly more patients in the high ΔP group than the low ΔP group had PGD grade 3 (PGD3), pneumonia, required tracheostomy, and had prolonged postoperative extracorporeal membrane oxygenation (ECMO) time, postoperative ventilator time, and ICU stay.
Conclusions
Driving pressure appears to have the ability to predict PGD3 and 90-day mortality of patients following LTx. Further studies are required to confirm our results.
Introduction
Lung transplantation (LTx) is an effective treatment for most end-stage lung diseases. 1 The process of lung transplantation includes procurement, organ preservation, implantation, and reperfusion, all of which are multiple stresses which may ultimately lead to primary graft dysfunction (PGD). 2 PGD is a syndrome of acute lung injury that occurs in the first 72 hours following LTx, and is characterized by pulmonary oedema with diffuse alveolar damage. 3 Clinical manifestations of PGD include, progressive hypoxemia and radiographic pulmonary infiltrates without other identifiable causes. 3 According to the International Society for Heart and Lung Transplantation (ISHLT) guidelines, PGD is graded based on partial pressure oxygen (PaO2) to fraction of inspired oxygen (FiO2) (P/F ratio) and chest radiographs. 4 The severity of PGD should be assessed daily at several time points following lung transplantation. The presence of radiographic infiltrates consistent with pulmonary oedema and a P/F ratio of <200 mmHg after 72 hours of final lung perfusion is classified as Grade 3 PGD (PGD3). 5 The incidence of any PGD grade has been reported to be up to 80%, and the incidence of PGD3 at any time point in the first 72 hours has been reported to be up to 30%. 6 Importantly, PGD is associated with significant morbidity and mortality, with PGD3 showing worse long-term survival.5,6 Multiple studies have reported that PGD is associated with prolonged length of mechanical ventilation, intensive care unit (ICU) stay, increased costs and short-term mortality.7,8
PGD shares clinical and pathological similarities with acute respiratory distress syndrome (ARDS). 9 Interestingly, several studies have shown an association between high driving pressure and poor outcomes in patients with ARDS.10–12 However, the association between driving pressure and prognosis in patients after LTx who develop PGD remains unclear.13,14 Therefore, the purpose of our study was to investigate the association between driving pressure and 90-day mortality in patients following LTx who developed PGD.
Methods
Patients
This prospective, observational study involved consecutive patients who, were admitted to our intensive care unit (ICU) following a LTx from January 2022 to January 2023. Patients included in the study were ≥18 years of age and had a postoperative survival time ≥72 hours. Exclusion criteria were: re-transplant/multi-organ transplant; postoperative thoracotomy to stop bleeding; incomplete measurements of driving pressure. The study was approved by the medical ethics committee of the Affiliated Wuxi People’s Hospital of Nanjing Medical University (No. KY23039). All patients provided written informed consent. All data were collected and retained anonymously. The reporting of this study conforms to STROBE guidelines. 15
Driving pressure
The ratio between tidal volume (VT) and end-expiratory lung volume represents the strain applied to the lung. Therefore, this ratio, also known as driving pressure, can be considered a substitute for lung strain. Driving pressure (ΔP) is calculated as the difference between plateau pressure (Pplat) and total positive end-expiratory pressure (PEEP). 16
In the ICU, most patients were supine without spontaneous respiration and required mechanical ventilation. The remaining patients received sedatives, analgesics, and muscle relaxants before volume-controlled ventilation. The ventilator (Dräger Evita 4, Germany) delivered a square waveform flow. To determine the correct Vt, it was necessary to calculate the patient’s predicted body weight (PBW) using the formula: men: PBW = 50 + 0.91 × (height in cm–152.4) kg; women: PBW = 45.5 + 0.91 × (height in cm–152.4) kg. 17 . Initial settings were as follows: VT, 6 ml/kg of PBW; PEEP, 5 cmH2O; respiratory rate (RR), 12/min. Adjustments were made according to the patient’s condition. 13 During extracorporeal membrane oxygenation (ECMO), the driving pressure was kept <15 cmH2O by adjusting the VT and PEEP. Simultaneously, the ECMO gas flow rate was adjusted to keep the partial pressure of carbon dioxide (PaCO2) <45 mmHg.
Diagnosis of PGD
PGD grades were calculated according to the recommendation of the ISHLT working group. 4 Patients were graded according to P/F ratio and chest radiographs at time points, T0, T24, T48, and T72 hours. The first time point (T0) was 2 hours after arrival in ICU when the patient’s ventilation and hemodynamic condition had stabilized.
Patients without diffuse allograft infiltrates on chest radiograph were classified as PGD grade 0 (PGD0) and were considered to not have PGD. Patients with diffuse allograft infiltrates on chest radiograph and P/F ratio >300 mmHg were classified as PGD1, patients with diffuse allograft infiltrates and P/F ratio of 200–300 mmHg PGD2, and patients with diffuse allograft infiltrates and P/F ratio of <200 mmHg as PGD3. Patients with diffuse allograft infiltrates on chest radiograph while on ECMO were automatically classified as PGD3. Patients on ECMO without diffuse allograft infiltrates on chest radiograph were considered ungradable.
For extubation patients had the following: successful spontaneous breathing trial (SBT) for two hours; not receiving, or, on low-dose vasopressor drugs; Glasgow coma scale score ≥13; no upper airway resistance and have controllable secretions; ameliorative imaging of chest X-ray; pH > 7.35; blood haemoglobin (Hb) levels >9 mg/l; temperature ≤38°C. Reintubation was defined as an endotracheal intubation required within 72 hours of successful extubation.
Data collection
The following data were collected from hospital medical records: sex; age; body mass index (BMI); PBW; smoking status; primary disease; concomitant diseases (e.g., hypertension, diabetes, pulmonary hypertension); forced vital capacity (FVC); forced expiratory volume in one second (FEV1); FEV1/FVC; diffusing capacity of the lungs for carbon monoxide (DLCO); Hb levels measured within 48 hours before LTx; aspartate transaminase (AST) measured within 48 hours before LTx; serum creatinine measured within 48 hours before LTx; ECMO type; surgical type before the ICU admission. At T0, the following were recorded: VT; Pplat; PEEP; RR; FiO2; PaCO2; P/F; ΔP, PGD. Other variables recorded included: postoperative ECMO time; postoperative ventilator time; reintubation; ICU stay; 90-day survival following LTx.
Statistical analysis
Statistical analysis was performed using SPSS software (version 25.0 for Windows®; IBM Corp, Armonk, NY, USA) and R software (version 4.3.0). A two-sided
To investigate the possible risk factors for ICU mortality at 90 days, pre- and postoperative variables and complications were compared between survivors and non-survivors and variables with
Results
Demographic data
Of the 114 patients who underwent a LTx at our centre during the study period, 104 were eligible for our study (Figure 1). Of these, 69 (66%) patients were in the low ΔP group (≤15 cmH2O) and 35 (34%) in the high ΔP group (>15 cmH2O). Prophylactic use of ECMO following LTx reduces the risk of severe PGD, but prolonged use can lead to complications (e.g., coagulation disorders, postoperative bleeding, and catheter-associated infections). At our centre, we conduct an ECMO weaning assessment within 24 hours of admission to the ICU by attempting to shut off the air source and reduce the flow rate to assess the patient’s tolerance. In total, 90 patients received ECMO (56 in low group and 34 in high group) (Table 1). Of the 90 patients, 39 (43%) had ECMO removed within 24 hours, and 61 (57%) had ECMO for more than 24 hours. Of the 61 who had ECMO weaned >24 hours, 28 (46%) were in the low ΔP group and 19 (54%) were in the high ΔP group, but this difference was not statistically significant. Indeed, no significant differences were detected between low and high ΔP groups for any the baseline demographic or clinical variables (Table 1).

Patient flow chart. Abbreviations: LTx, Lung transplantation; ΔP, driving pressure.
Patient characteristics before admission to ICU.

Data are expressed as, n (%) or mean ± standard deviation or median (interquartile range).
Abbreviations: ICU, intensive care unit; BMI, body mass index; MV, mechanical ventilation; P/F, ratio of partial pressure oxygen (PaO2) to fraction of inspired oxygen (FiO2); PBW, predicted body weight; IPF, idiopathic pulmonary fibrosis; COPD, chronic obstructive pulmonary disease; FVC, forced vital capacity; FEV1, forced expiratory volume in the first second; DLCO, diffusing capacity of carbon monoxide; Hb, haemoglobin; AST, Aspartate transaminase; ECMO, extracorporeal membrane oxygenation; LTx, lung transplantation; ns, not statistically significant.
Post-operative data
There were no significant differences between groups in the number of patients who required reintubation, but PaCO2 was significantly lower in the low ΔP group compared with the high ΔP group (
Postoperative data and observed outcomes.

Data are expressed as, n (%) or mean ± standard deviation or median (interquartile range).
Abbreviations: VT, tidal volume; Pplat, plateau pressure; PEEP, positive end expiratory pressure; ΔP, driving pressure; RR, respiratory rate; PaCO2, partial pressure of arterial blood carbon dioxide; P/F, ratio of partial pressure oxygen (PaO2) to fraction of inspired oxygen (FiO2); PGD, primary graft dysfunction; ECMO, extracorporeal membrane oxygenation; MV, mechanical ventilation; AKI, acute kidney injury; ICU, intensive care unit; ns, not statistically significant.
Importantly, significantly fewer patients in the high ΔP group compared with the low ΔP group survived to 90-days (63% vs 83%,

Pearson’s correlation coefficient (r) was determined between mechanical ventilator (MV) time (h) and driving pressure (ΔP) cmH2O. There was a significant correlation between ΔP and postoperative MV time (r = 0.399;
To investigate the possible risk factors for ICU mortality at 90 days, variables were compared between survivors and non-survivors and those with
Results from Cox proportional hazards regression model.
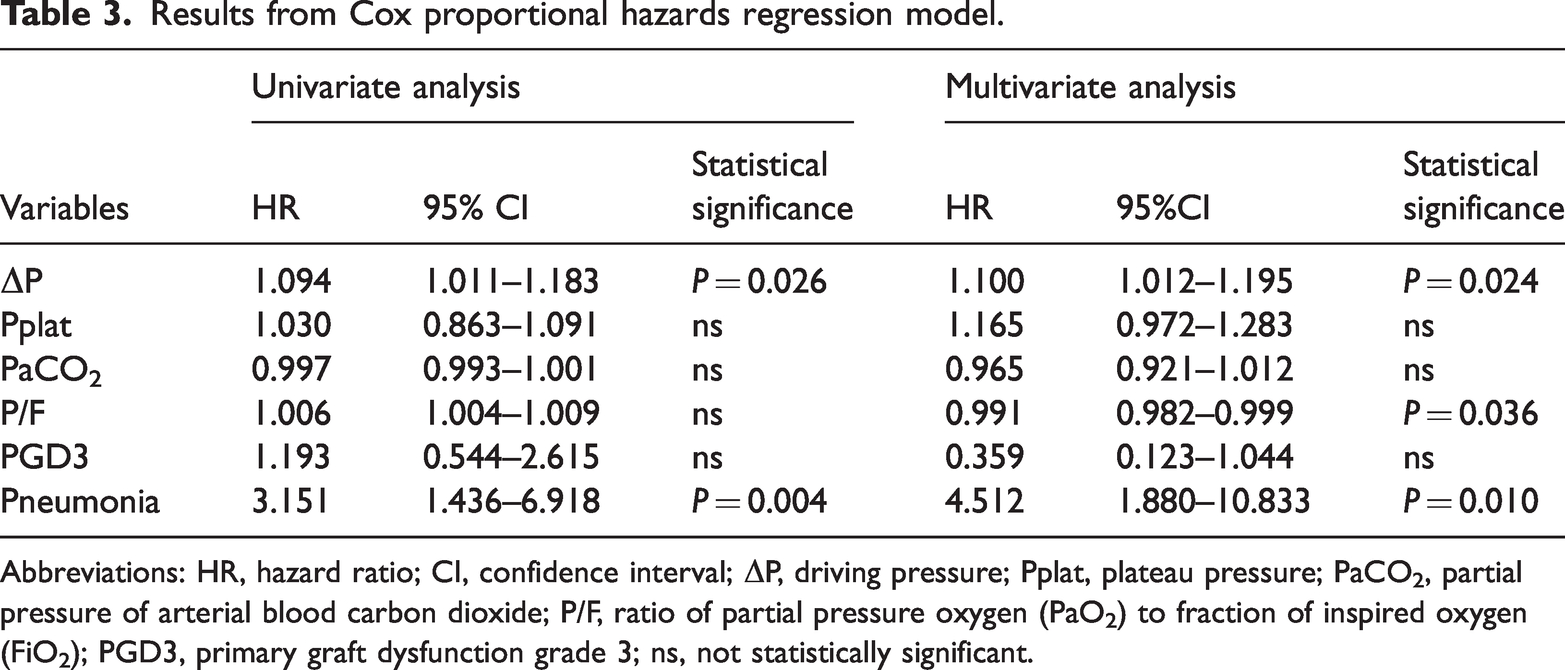
Abbreviations: HR, hazard ratio; CI, confidence interval; ΔP, driving pressure; Pplat, plateau pressure; PaCO2, partial pressure of arterial blood carbon dioxide; P/F, ratio of partial pressure oxygen (PaO2) to fraction of inspired oxygen (FiO2); PGD3, primary graft dysfunction grade 3; ns, not statistically significant.
Using Pearson’s correlation analysis, we found a significant correlation between ΔP and postoperative ventilator time (r = 0.399;

Survival curve analysis of the percentage of patients on mechanical ventilation (MV percent) by low (≤ 15 cmH2O) and high (>15 cmH2O) driving pressure (ΔP) groups. The difference between groups was statistically significant (

Kaplan-Meier curves for 90-day mortality for low (≤15 cmH2O) and high (>15 cmH2O) driving pressure (ΔP) groups. Curves were compared using a log-rank test. The difference between ΔP groups was statistically significant (
Patients with PGD3 had statistically significantly higher ΔP than PGD0/1 (

Comparison of driving pressure (ΔP) cmH2O in the different grades of primary graft dysfunction (PGD) groups.

(a) Receiver operating characteristic (ROC) curve of the predictive ability of driving pressure (ΔP) for PGD3. Area under the curve (AUC) was 0.709 (95% CI, 0.608–0.810;
Discussion
Since patients are prone to PGD following LTx, it is necessary to improve their prognosis by identifying risk factors for PGD during their ICU stay. Prompt intervention and management of PGD will undoubtedly improve patient outcomes. PGD is an acute lung injury of the donor lung that results in a decrease in functional lung volume and decreased lung compliance, and so, in many ways, is similar to ARDS. 9 Several studies have found an association between high driving pressure and poor outcomes in patients with ARDS.10–12 Therefore, we assumed that driving pressure would have the ability to predict the occurrence of PGD3 and 90-day survival in patients with PGD following LTx.
In a previous study in patients with ARDS, driving pressure was closely related to hospital mortality, even among patients who received protective ventilation. 10 In that study, the cut-off value of driving pressure for high mortality was approximately 15 cmH2O. Therefore, for our study, we used 15 cmH2O as the cut-off value for high and low driving pressure groups. Similar to the previous study, 10 we found that the 90-day mortality was higher in high ΔP group than in low ΔP group, and each unit increase of ΔP (1 cmH2O) was associated with a 10% increment in mortality. Our analysis showed that driving pressure was independently associated with increased risk of death and the ΔP was higher in non-survivors than in survivors, though the difference between groups was only approximately 2 cmH2O. Using ROC curve analysis to predict LTx outcomes, we established accurate cut-off values. The cut-off value for △P for PGD3 was approximately 15.5 cmH2O and for 90-day mortality was approximately 13.5 cmH2O. The AUC of △P for PGD3 was higher than that for 90-day mortality suggesting that △P is more predictive for the severity of PGD than mortality. This finding is not surprising since death will probably be influenced by several factors. In addition, we found that patients with PDG3 had significantly higher ΔP than patients with lower grades of PGD. We speculated that patients in the low ΔP group had better lung compliance than those in the high ΔP group, which would have led to better gas exchange with a higher P/F and lower PaCO2 as we observed in our results. 18
Prolonged MV has been associated with impaired mid-term and long-term results following LTx and, has been linked with the possibility of ventilator-induced lung injury (VILI) and lung infections.19,20 The difficulty for clinicians is to identify which patients are suitable for early extubation after undergoing LTx. 21 In our study, postoperative MV time was significantly greater in the high ΔP group compared with the low ΔP group. Therefore, △P might help identify patients who would wean successfully away from MV as early as possible after LTx.
Our study included both single and double lung transplantations, and it was assumed that more patients with double lung transplantation would be in the low △P group. However, our results showed no significant difference in numbers of patients in the low and high △P groups. While our initial settings for MV were, VT, 6 ml/kg of PBW; PEEP, 5 cmH2O; respiratory rate (RR), 12/min, adjustments could be made according to the patient’s condition. In addition, some patients with ECMO required ultra-pulmonary protective ventilation and △P was kept <15cmH2O by adjusting the VT and PEEP. Perhaps △P-guided lung protective ventilation may improve outcomes for patients following LTx. This hypothesis needs to be confirmed by future clinical studies.
Our study had some limitations. For example, it was a single-centre study, and so the generalisability of our results may be limited. Additionally, interventional studies are required to assess whether postoperative △P-guided lung-protective ventilation would improve outcomes for patients following LTx.
In conclusion, our results showed that ΔP was an independent risk factor for 90-day mortality in patients following LTx, and, ΔP was higher in non-survivors than in survivors. Significantly more patients in the high ΔP group than the low ΔP group had PGD3, pneumonia, required tracheostomy, and had prolonged postoperative ECMO time, postoperative ventilator time, and ICU stay. The evaluation of ΔP following weaning from MV may be useful clinically. Driving pressure appears to have the ability to predict PGD3 and 90-day mortality of patients following LTx. Further studies are required to confirm our results.
Footnotes
Acknowledgements
We are grateful to acknowledge Yachen Shi for his valuable suggestions and discussions.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
This research received no specific grant from funding agency in the public, commercial, or not-for-profit sectors.
