Abstract
Introduction
After stroke, upper limb impairment affects independent performance of activities of daily living. We evaluated the usability, functionality, and efficacy of a myoelectric elbow-wrist-hand orthosis to provide support, limit unsafe motion, and enhance the functional motion of paralyzed or weak upper limbs.
Methods
Individuals with stroke participated in a single-session study to evaluate the device. Ability to activate the device was tested in supported and unsupported shoulder position, as well as the elbow range of motion, ability to maintain elbow position, and ability to lift and hold a range of weights while using the device.
Results
No adverse events were reported. 71% of users were able to operate the device in all three active myoelectric activation modes (Biceps, Triceps, Dual) during testing. Users were able to hold a range of wrist weights (0.5–2 lbs) for 10–120 seconds, with the largest percentage of participants able to hold weights with the device in Biceps Mode.
Conclusions
The myoelectric elbow-wrist-hand orthosis improved range of motion during use and was efficacious at remediating upper extremity impairment after stroke. All users could operate the device in at least one mode, and most could lift and hold weights representative of some everyday objects using the device.
Keywords
Introduction
Neuromuscular impairment of the upper extremity can occur from a myriad of medical issues, such as stroke, spinal cord injury, brachial plexus injury, multiple sclerosis, traumatic brain injury, and amyotrophic lateral sclerosis. The degree and presentation of upper extremity impairment can also vary widely, ranging from complete flaccidity to severe muscle synergy patterns and involuntary reflexes, which may contribute to significant impairment of the shoulder, elbow, wrist, and hand. The resultant loss of function may limit an individual’s ability to perform activities of daily living (ADLs).1–4 In fact, over 80% of stroke survivors experience upper limb impairment, and only one in three achieve a full functional recovery.5–7
Often an orthosis (or brace) is used to counteract upper limb impairment with the goal of facilitating improved function after stroke or other neurological injuries.8–10 Orthoses can be off-the-shelf, custom-fit, or custom-fabricated. The design is chosen depending on the purpose and the user’s needs, and can be non-powered or powered. Current evidence suggests that non-powered, static upper limb orthoses have limited task-specific benefits, especially for people post-stroke, 11 while powered devices often suffer from non-intuitive control structures. 12 , 13 This reduces the usability and fails to restore function to the neurologically-impaired upper limb, especially when hypertonicity or spasticity is present. Powered myoelectric devices, controlled intuitively by electromyographic (EMG) signals, may offer the potential to enhance functional use of the impaired limb.
The MyoPro® (Figure 1; Myomo Inc., Cambridge MA, USA) is a commercially available, custom-fitted powered myoelectric elbow-wrist-hand orthosis (EWHO). 14 This device is intended to be used as a supportive and assistive device in everyday tasks12,13 by individuals with a weakened or dysfunctional upper extremity. MyoPro® technology utilizes software to amplify surface EMG signals from the musculature of the affected arm, to translate these signals into orthosis motion via an externally powered motor unit. The orthosis is meant to support and restrict hypermobility of a weak or deformed arm, while the orthotic joint allow for movement of the anatomical joint. The MyoPro® has been used previously by individuals post-stroke and by individuals with brachial plexus injuries (BPI), multiple sclerosis (MS), amyotrophic lateral sclerosis (ALS), traumatic brain injury (TBI), and spinal cord injury (SCI). 13,15 The ability of severely hemiplegic stroke survivors to effectively operate a myoelectric controlled powered orthosis has been previously reported 13 and similar devices have been explored in clinical and rehabilitative contexts.16–23 For example, immediate use of the MyoPro® improved performance on the Fugl-Meyer Upper Limb Assessment in chronic stroke survivors. 24 Most of these works are either case studies for single users or implementing an orthosis in a therapeutic strategy. However, no studies have assessed, in a controlled setting, whether the device works as intended by the manufacturer for a community-dwelling, impaired population.

The MyoPro® powered orthosis from Myomo Inc. Photograph courtesy of Myomo Inc.
The purpose of this study was to evaluate the usability, functionality, and efficacy of the MyoPro® device on upper limb support and assistance in community-dwelling chronic stroke survivors. The long-term goal of this work is to inform device development and assess the basic functions of a powered upper-limb orthotic in an impaired population, for potential implementation in future clinical trials.
Methods
All study methods were approved by the Northwestern University Institutional Review Board (STU00104395). Participants provided written informed consent prior to participation in the study.
Participants
Participants were recruited through IRB-approved fliers placed in study-associated orthotic and physical therapy clinics at the Shirley Ryan AbilityLab, its affiliated greater Chicago locations, and Geauga Rehabilitation Engineering Orthotics and Prosthetics in the greater Cleveland area. These sites were chosen to target populations of potential or current MyoPro® users. To qualify for the study, participants had to meet the following inclusion criteria: (a) be an adult (over 18 years old), (b) have upper limb impairment caused by mild to moderate cerebrovascular accident (CVA), (c) be able to support the weight of the device, (d) have no fixed upper limb contractures on the affected side, (e) be able to read and comprehend the English language, and (f) have
Participant demographics, clinical evaluation, and device experience.

*User had his own device but it was unavailable on testing day so the adjustable device was utilized instead.
#MyConfig settings are available on the tablet application to set limits for the flexion and extension.
Device
The MyoPro® orthosis used in this study provides externally powered motion for elbow flexion/extension, utilizing EMG signals from the user’s biceps and triceps as control inputs. The EMG signal is smoothed using a bandpass filter technique and amplified to produce a proportional response at the motor when it exceeds a predetermined threshold. The device is custom-molded for each user to achieve optimal EMG detection and movement assistance. The weight of a fully fabricated device is approximately 2.75lbs, with some variance due to customization of the orthosis size and shape (Figure 1). The motor unit can generate a maximum of 7 Nm of rotational torque at the joint, which equates to a lifting force of approximately 2–2.5 kg (4–5 lbs) for an average-sized device. The power source is an internal, rechargeable NiMH battery.
For the purposes of the study, participants used either their own personal MyoPro® orthosis (MyoMo Classic, MyoMo Motion W, or the MyoMo Motion G, v2015), that was fitted and programmed by a MyoPro® certified practitioner prior to the study, or an adjustable MyoPro® device provided by Myomo Inc. for this study. Both types of devices used EMG signals, obtained from surface EMG sensors placed on the biceps and triceps of the affected limb, to control elbow movement. Three active modes are available on the device: Biceps Mode, Triceps Mode, and Dual Mode. In Biceps Mode or Triceps Mode, the device uses EMG signals from only the biceps or triceps, respectively, to generate corresponding elbow motion. Relaxation of the agonist muscle allows the antagonist action to occur, e.g., in Biceps Mode, elbow flexion occurs with biceps activation and elbow extension occurs with deactivation of the biceps. Dual Mode requires sequential control of biceps and triceps activation/inhibition to allow proportional joint motion. Even small EMG signals (as low as 5 µV) can be detected by the surface EMG sensors and amplified to control the device, allowing movement in a previously restricted extremity. Typical presentations of upper extremity weakness associated with chronic stroke hemiparesis make Biceps Mode the simplest to use for most individuals, and this is the most commonly selected mode for new users. In Standby Mode, the motor does not activate in response to EMG signals.
The software provided by Myomo® Inc. for adjusting and viewing the device’s settings, MyConfig, was used to view the myoelectric signals and device settings of each myoelectric EWHO during the testing, but settings were not modified during the testing process.
Testing equipment
Testing equipment included a stopwatch, a standard manual goniometer, and wraparound wrist cuff weights of 0.5 lb, 1 lb, and 2 lb. Separate custom hardware and software were also used initially to verify surface EMG signals from biceps and triceps muscles at 1000 Hz using standard Ag/AgCl disposable pre-gelled electrodes (GS-26, Bio-Medical Instruments, MI, USA).
Protocol
Screening of potential participants was performed prior to admission to the study, and qualified, interested individuals provided written informed consent. The IRB-approved testing protocol was performed on a single day by two study personnel, a licensed and registered occupational therapist and a certified orthotist. Participants’ EMG signals were first assessed using detachable sensors from Bio-Medical Instruments to visually confirm that surface EMG signals were available to control the MyoPro®.
Clinical Screening: First, tests were performed without the device to determine baseline functional mobility without assistive technology. Spasticity was quantified using the Modified Ashworth Scale to determine if velocity-dependent motion could create an unsafe scenario in which the anatomical ROM might be exceeded during device use. Elbow and shoulder active range of motion (AROM) and passive range of motion (PROM) were measured using a standard manual goniometer.
Device Acclimation and Settings: All participants were given the opportunity to acclimate to the device. During this acclimation period, the orthotist examined and adjusted the device fit for proper contact of EMG electrodes. EMG settings were previously programmed by their clinical orthotist (MyoPro trained), who used the MyConfig software during muscle activation/relaxation to optimize EMG gain and thresholds. EMG settings were customized for each patient to trigger the device without creating undesired motion. Testing began when the participant felt comfortable with the device fit and procedures.
Usability Testing: Participants were asked to demonstrate donning/doffing, turning the device on/off, and changing modes. Participants were also asked to move the device to its full range of motion in Biceps and Triceps Modes and to full flexion, full extension, and midrange stop (45–90°) in Dual Mode. Elbow support, wrist support, and hand support at the anatomical joints, as provided by the MyoPro®, were noted during each mode and position to ensure no malalignment (hyperextension or mediolateral instability) occurred. To evaluate this, clinicians (OT/CPO) observed the participant’s joint position during donning and mode changes of the device by the participant. If a device was malpositioned or malfitting (e.g., device joint unable to align with anatomical joint center), the device could create hyperextension or angulation past a participant’s available ROM once turned on. This was determined via a clinical exam of A/PROM collected initially by the OT. We also monitored for skin breakdown under the electrodes during the functional use of the device. Skin checks were completed before, during, and after each testing session, and any abnormalities were documented. Any reports or observation of pain, irritation, or erratic motion were also documented.
Functionality Testing: Functionality was defined as the ability of the user to activate the device in different modes and move it through their range of motion at multiple shoulder positions. AROM of the elbow was measured in multiple shoulder positions both with and without the device. Elbow flexion and extension were tested at four shoulder positions with neutral forearm: 450 abduction, 900 abduction, 450 flexion, and 900 flexion. AROM in these positions was compared across four conditions: (1) without the device and without therapist support (Unsupported, no device); (2) wearing the device in Biceps Mode on the affected arm, without therapist support (Unsupported, device); (3) wearing the device in Biceps Mode on the affected arm, with additional therapist support (Supported, device); and (4) without the device, with therapist supporting the affected arm (Supported, no device). Some participants (n = 14) used a shoulder saddle harness during their daily activities. Therapist support was provided as static support at the shoulder and proximal humerus to maintain the positions of shoulder abduction and flexion during each position. For individuals who used a harness, it was loosened or partially removed during a test if it prevented the user from achieving a required position (most commonly for 900 flexion/abduction). This was to ensure that ROM during device use was not restricted by additional factors.
Efficacy Testing: Efficacy was defined as the ability of the user to accurately control and operate the device to perform a range of movements and positions that mimicked ADLs, requiring controlled elbow positions while maintaining objects of varying weights using different device modes. This was tested with three different weights, chosen as representative of household items a person may need to lift on an everyday basis—2lbs (wrist weight), 1 lb (wrist weight), and 0.5 lb (canvas bag). These weights might represent utensils, food items or clothing. The subjects had the weights strapped to the distal forearm for all conditions and did not require simultaneous, coordinated grasp and elbow control. Weights were held for durations of 10 seconds, 30 seconds, 1 minute, or 2 minutes. These conditions were performed with the device in (i) Biceps Mode (with a goal of full elbow flexion) and (ii) Dual Mode (with a goal of maintaining the elbow within mid-range, 45–900). Participants were allowed three attempts to complete each weight and time condition. No therapist support was provided during this testing. If a participant was not able to hold a given weight for the given amount of time after three trials, they were not asked to perform more difficult levels of the task.
Statistical data analysis
Each participant’s ability to perform AROM tasks under each experimental condition was rated as “unable or partially able” (0) or “fully able” (1). Regression models comparing the ability to perform a task under different experimental conditions (shoulder abduction or flexion at 45° or 90° angles, with and without the device) as predictors were fitted using the Generalized Estimating Equation (GEE) approach, with within-participant exchangeable correlation structure. This model appropriately accounts for within-participant correlation between multiple measurements under different experimental conditions. Interactions between these terms were examined. Separate models were used for each of the four elbow tasks.
For functional activity assessment, performed in Biceps and Dual Modes, each participant’s weight-holding capacity was summarized as the maximum weight they could hold for at least 10 seconds. In addition, a weight-time combination index was calculated as the product of the maximum weight held and length of time that the participant could hold the weight. This metric is indicative of participant’s maximal weight tolerance during a functional activity. A Wilcoxon’s signed rank test was used to compare the maximum weight and the maximum combination index for different conditions. All statistical analyses were performed using Stata 14 software (Stratacorp, LLC).
Results
Usability
All participants (n = 18) were able to don/doff and use the device without any assistance in standby and active modes without any hyperextension and/or medio-lateral instability. No participants reported pain or skin breakdown while using the MyoPro®, and all were able to generate EMG activity in the biceps and triceps muscles during initial EMG testing.
There were no serious adverse events, such as hyperextension of the joint, pain caused by the device, or skin breakdown. Nine of the 18 participants experienced undesired events associated with a technical issue, including loss of responsiveness of the device (three participants) or unexpected motion caused by loss of battery, device migration, or suboptimal fit (six participants). These issues were more common for participants who tested with the adjustable device (4/6 participants) then those with a custom-fit device.
All participants were able to achieve full elbow flexion in Biceps Mode, while 72% of participants achieved full elbow extension in Triceps Mode. Ability to initiate the device in Dual Mode (co-contraction; shown in Table 2) indicated that 81% of participants were successful in achieving mid-range control of the elbow.
Number of participants able to initiate the device in different modes.

Functionality
More patients were able to complete the AROM task using the device than without it (Table 3). Interactions between functional task type (shoulder flexion vs. abduction, at 45° or 90°, with or without the device) were not statistically significant for any of the elbow tasks. The odds ratios (OR) from the GEE models are reported in Table 4. The odds of being able to perform a task with the shoulder at 90° were lower than at 45° for both unsupported elbow flexion (OR = 0.16, p < .001) and extension (OR = 0.21, p=.002), and unsupported elbow flexion tasks were more difficult with shoulder flexion than shoulder abduction (OR = 0.28, p < .001). Using the device increased the odds of being able to perform elbow extension (OR = 3.93, p < .001). No other significant differences were found.
Percentage of participants able to complete positional target tasks under different shoulder conditions during the AROM testing.

Note: The MyoPro® was set to biceps mode in all device conditions.
Odds ratio (OR) of completing tasks under different conditions during AROM testing.

Efficacy
Table 5 provides the time duration achieved by each participant for the weight and device conditions. Most participants were able to hold the 0.5 lb, 1 lb, and 2 lb weights for 2 minutes (77.8%, 66.7%, and 55.6%, respectively) in Biceps Mode, but fewer could accomplish this task in Dual Mode (43.8%, 56.3% and 31.3%, respectively). The maximum weight that participants could hold, regardless of duration, was significantly higher in Biceps Mode than in Dual Mode (p = 0.009, Wilcoxon signed rank test). The combination index of maximal weight tolerance was also higher for Biceps Mode (p = 0.054, Figure 2).
Time condition completed while holding weights in biceps and dual modes.
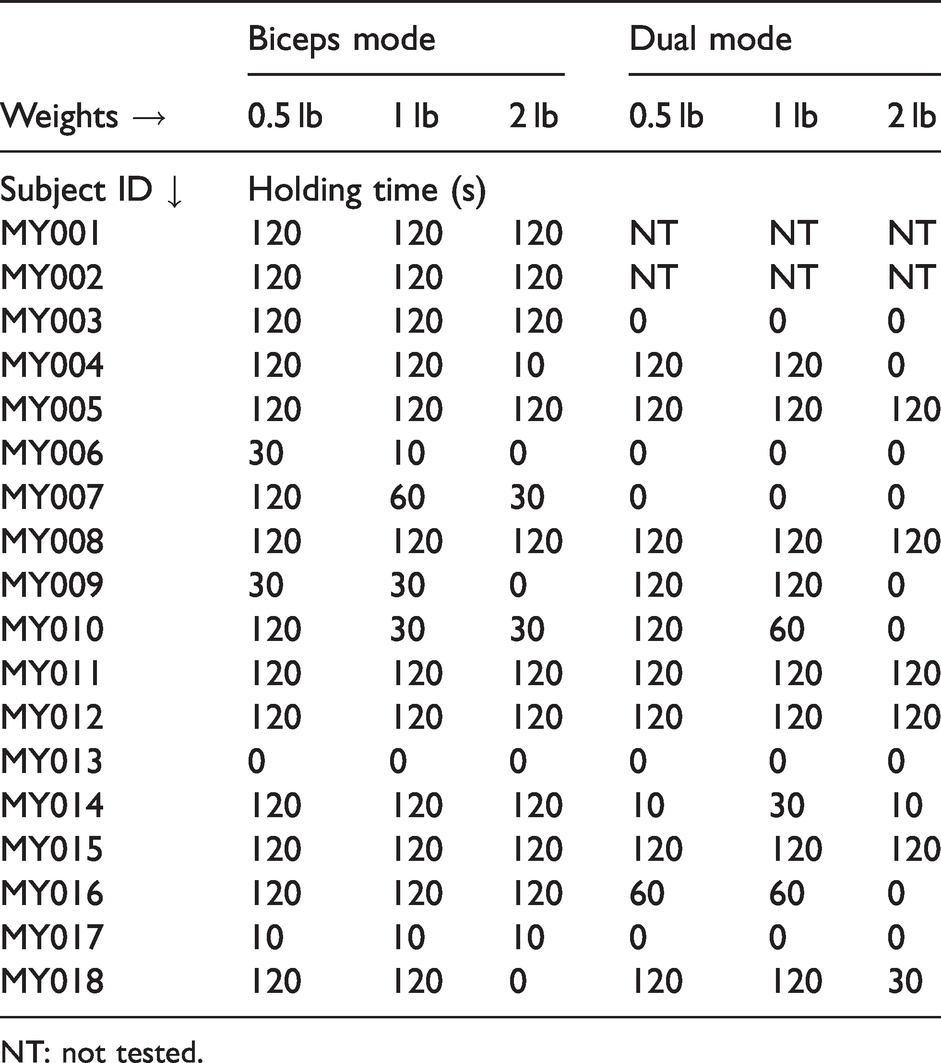
NT: not tested.

User’s ability to hold functional weights of different form factor in Biceps and Dual Modes (separated by –– line). The horizontal axis represents the percentage of users that were able hold the weights and the vertical axis indicates the duration of holding (up to 2 min).
Discussion
This study demonstrates the feasibility of using the MyoPro® myoelectric EWHO for community dwelling chronic stroke survivors, with participants able to don/doff the device, switch modes, and maintain the positioning of the impaired elbow in both active and standby modes. It also provides evidence that the device can improve active, assisted range of motion and support weight-bearing upper extremity activity in individuals with chronic stroke. Some minor undesired events occurred during the study including improper sensor placement, low battery, and migration of the device. These events did not jeopardize the participants’ safety, but usability of the device may be improved with further training for new users and with a more adjustable shoulder saddle harness to ensure proper support and suspension while maintaining ROM at the shoulder. Participants who used the adjustable device encountered technical issues with the device often related to a suboptimal fit, suggesting the need for a custom-fitting orthoses for best use. Other fitting and training issues would likely be mitigated by further orthotic and therapeutic education. Further studies are needed to assess long-term usability and functional assistance during ADLs.
Participants were able to fully initiate the device, regardless of whether they had used the device for some time or if they were a novice user. All participants were able to successfully use Biceps Mode. Although Triceps Mode and Dual Mode were more challenging, 72.22% of participants were able to operate the orthosis in Triceps Mode, 93.75% could initiate biceps movement in Dual Mode, 87.50% could initiate triceps movement in Dual Mode, and 81.25% could stop their arm mid-range while operating in Dual Mode, which is important for many functional activities. Failure to initiate the device or achieve full ROM in these modes may result from abnormal muscle synergies, which are well-documented in the upper limb after stroke. During stroke recovery, it is common for patients to experience different levels of spasticity and abnormal synergies, commonly seen as flexion patterns in the upper extremity that presents itself as shoulder internal rotation, elbow flexion, forearm pronation and wrist/finger flexion. 25 Failure of the antagonist muscle to relax when the agonist muscles contracts creates co-contraction, promoting dysfunctional movement patterns. In post-acute patients, these imbalances in muscular contraction and poor coordination of agonist-antagonist muscles likely contribute to the difficulty in controlling Triceps Mode over the Biceps Mode when using the device. The ability of most users to successfully initiate all three modes suggests that, with proper training, all three modes could be achievable for most patients with upper limb impairment. Use of Dual Mode would provide the highest functional value, as users would have direct control of joint angles, allowing them to carry objects of varying sizes or to use their affected extremity to assist in more complex bimanual tasks. More users were able to complete the activities in their active range of motion for both flexion and extension tasks with the device than without it. In addition, the odds of completing the tasks were higher when using the device, indicating that the device increased the capacity for functional movement. Subjects were better able to perform elbow flexion and extension in the device while supported by the therapist in all shoulder positions, likely due to the ability to focus coordination on a single joint action and reduction of co-contractions associated with unsupported biarticular positional control. Subjects may not be able to perform ADLs independently when a task requires more variable shoulder position. However, it was found that in most positions of shoulder abduction and flexion, subjects were more successful in the device than out of the device when achieving positional targets.
Functional task performance could be supported if ROM could be controlled and enhanced with MyoPro® use, even if muscles remain too weak to provide enough volitional power to overcome gravity or lift objects. Furthermore, a focus on bimanual tasks in stroke patients has been shown to provide a significant and often untapped therapeutic benefit. 7 , 26 Given that functional improvements in the upper limb following stroke is still limited, 27 , 28 it is an interesting and intriguing opportunity to see if introducing a myoelectric EWHO at an early stage of acute post-stroke care can provide stable support and improve ROM and neuromuscular activation.
Many participants were able to hold weights typical of everyday objects against gravity using the MyoPro® over time, without training. Heavier weight was generally more difficult for subjects to maintain for longer periods, likely resulting from a need to maintain consistent muscle signal while balancing the greater mechanical torque created by increasing weight. Additional device tuning might reduce the need for this increased effort but would also likely change sensitivity and reliability; optimal functionality of the device must be determined for each patient based on their current presentation and goals for use. Most functional tasks require less than two minutes of extended holding, which was seen to be generally achievable in Biceps Mode and moderately more difficult in Dual Mode. This supports use of the device to perform some real-world tasks, such as bimanual holding of items or carrying a bag with the affected arm while ambulating.
Study limitations
The current study did not incorporate patient reports or surveys of device usability. Although this was not in the scope of the study, patient perspectives are critical for further device development to ensure the needs of the user are being met; future studies should consider questionnaires such as the System Usability Scale, QUEST 2.0, or Usefulness, Satisfaction, and Ease of use (USE) for in-depth usability evaluations of these or similar devices. Having multiple versions of this myoelectric EWHO (custom and adjustable) introduced additional variability into the testing, but was necessary to improve recruitment for this limited study population. Efforts were made to minimize the potential differences by only testing movement of the elbow joint, which was the same across all devices. Nevertheless, all device models utilized wrist-hand control to stabilize the forearm and position the wrist. Future studies should evaluate the functionality and efficacy of the wrist-hand assistance using consistent device models. Another limitation is that we did not collect separate electromyography signals to determine muscle activation patterns, and thus cannot evaluate the impact of the device on muscle synergies and resulting movement patterns; this would be an important consideration for future work. Additionally, we did not collect measures of these participants’ motor impairment (other than spasticity), which likely affects performance with the device. Participants were also not evaluated while holding weights without the device, which would be necessary to understand the device contribution to payload assistance and hold duration. Finally, the small sample size may not have been sufficient to capture variance among the larger target population. To determine whether results would generalize to other current or potential users, future work should consider evaluating the impact of level of impairment and amount of device experience on the users’ performance.
Conclusions
This study demonstrates the feasibility of a myoelectric EWHO as an assistive device for the upper extremity following stroke. The device was able to support the affected limb, and no significant adverse events were recorded during this study, including an absence of joint hyperextension or mediolateral instability. All participants were able to control the device in at least one of the three operational modes, and the device improved AROM of the elbow and allowed users to lift and hold weight. Given that stroke increasingly affects younger individuals with long life expectancies, it is critical to encourage the use of advanced orthotic technology to enhance motor performance while next generation clinical and pharmaceutical interventions are developed. Further work is required to investigate the additional benefits of training with the device, defining the characteristics of optimal users, customizing the devices for diverse patient populations, and evaluating of the devices during daily use and ADLs in long-term home trials. 19
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Partial funding and devices used during the study were provided by Myomo, Inc. through a grant to the Shirley Ryan AbilityLab and the Max Nader Lab. The funding sources had no role in the study design, data collection, data analysis, data interpretation, writing of the report, or decision to submit the paper for publication. The authors have no financial benefit to report.
Guarantor
AJ.
Contributorship
AJ conceived and designed the research protocol. SHL, CM, JA, KT and LK performed data collection. SHL, CM conducted data analysis. CM and MK performed the statistical analysis. AJ interpreted results. All authors participated in drafting the manuscript. All authors read and approved the final manuscript.
Authors’ note
Authors claim this report as original work and no part this manuscript was been presented or published elsewhere.
Device status
The MyoPro® (Motion E, Motion W and Motion G), from Myomo Inc., is registered for indicated usage in the US and is registered with the FDA as a Class II (510 (k) exempt) device (Biofeedback Device).
Acknowledgements
The authors would like to thank the participants for their participation and the clinical groups at Shirley Ryan AbilityLab and Geauga Rehabilitation Engineering Orthotics and Prosthetics for support during recruitment and testing procedures.
