Abstract
During the COVID-19 pandemic, which triggered a worldwide emergency call to pursue new drugs and devices to treat and cure the disease, vaccines began to be developed mid-2020, as a promising weapon and shield. Fiocruz, a Brazilian government science and technology institute, signed a public procurement for innovation contract with the foreign pharmaceutical company AstraZeneca to purchase, at that time, vaccines still under development. It was the first public procurement for innovation contract in the Brazilian public health system (SUS). This paper analyzes how the Brazilian public procurement contracts for the COVID-19 vaccine dealt with technology and institutional risks and uncertainties. It also aims to describe how such agreements (and the legal apparatus behind them) relate to the Brazilian system of innovation and to the theoretical approach of contracting for innovation. A key question is: how does the public procurement for innovation vaccine contract signed by Fiocruz deal with the intrinsic nature of uncertainty that characterizes innovation, and what lessons and challenges can be drawn?
Introduction
Public health sector policies cover areas of healthcare, science, and technology that demand innovation (Proksch et al. 2019; Trindade 2008). The problems of healthcare are complex, systemic, and urgent (also referred to as “wicked” problems), thus requiring insights and actions from different perspectives (Kattel et al. 2018; Mazzucato 2017). Less obvious, nonetheless, these problems are also highly challenging in terms of legal conception, structuring, and implementation.
In Brazil, the health sector is anchored in the public health system (Sistema Único de Saúde (SUS), the Brazilian equivalent to the National Health Service in the United Kingdom), a large and universal coverage policy arrangement that provides healthcare to citizens, often through large-scale public procurement and public–private partnerships (PDPs, Laplane 2020; Silva and Elias 2019; Sundfeld and Souza 2013; Varrichio 2017).
During the COVID-19 pandemic, which triggered a worldwide emergency call to pursue new drugs and devices to treat and cure the disease, vaccines began to be developed mid-2020, as a promising weapon and shield (Azoulay and Jones 2020; Da Silva et al. 2020). In 2020, the Brazilian government, through two public science and technology institutes, Instituto Butantan and Fundação Oswaldo Cruz (Fiocruz), signed contracts with foreign pharmaceutical companies whose scope included urgent purchase of vaccines—at that time, still under development (Rauen 2020). The Fiocruz COVID-19 vaccine contract was a new type of instrument for public procurement for innovation (PPI), referred to as “technology order agreements.” The Butantan COVID-19 vaccine contract, on the other hand, is a research and development agreement, a kind of collaborative contract that is not a typical public procurement contract, and, consequently, it is not examined in this paper. 1
We analyze how the Brazilian government Fiocruz technology procurement contract for COVID-19 vaccines dealt with technology and institutional risks and uncertainties, and we also describe how such an agreement (and the legal apparatus behind it) relates to the Brazilian innovation system and, more broadly, to the literature on contracting for innovation. Our more specific goal is to identify the roles—both constraining and facilitating—played by the law (and legal actors, processes, and institutions, broadly speaking) in fostering technology acquisition and proposing a distinguished methodological, sociolegal approach to examine how public procurement can foster innovation.
PPI has a key role as an innovation policy measure (Aschhoff and Sofka 2009; Edler and Georghiou 2007; Edquist et al. 2015). During the last decades, the Brazilian legal apparatus for PPI grew and was strengthened. Nonetheless, it is still underused, and, in part, there is a disconnection between public procurement and the innovation of public policy goals and priorities (Foss and Bonacelli 2022). The lack of resources to the existing apparatus concerning legal tools to foster innovation has also something to do with the Brazilian legal culture, prone to formalism, with little room for flexibility, experimentation, and uncertainty (being the latter a key and well-known feature of innovative processes).
A well-known assumption from which we depart is that building organizational (or governmental) capabilities to implement it is of primary importance for realizing development objectives (Andrews, Pritchett, and Woolcock 2017; Enriquez and Centeno 2012, among others). This means that good policies–proposals, plans, goals, and their respective institutional arrangements are not enough if countries effectively lack implementation capacities. Less obvious, we also assume that law and state actions are, in multiple and intricate ways, mutually constitutive and that if it is possible to observe and understand the roles of law in public policy, it would also be possible to improve or perfect them from a legal and institutional perspective (Coutinho 2013). In addition, for development strategies (this being the case of innovation policies) to be successful, it is essential that the law embedded in policy implementation processes play certain roles that ultimately require a nontrivial degree of state capacity, such as safeguarding flexibility, stimulating orchestration, framing synergy, and ensuring legitimacy (Trubek, Coutinho, and Schapiro 2014).
In terms of perceived weaknesses, the Brazilian subsystem of innovation policy and regulation has been described as having problematic overlapping objectives, including dealing with competition for and nonstrategic use of resources, discontinuity of investments, programs, and excessive bureaucracy in auditing of innovation policies, including procurement (Mazzucato and Penna 2016). In view of this, some questions emerge: how has the legal framework for contracting science, technology, and innovation—namely, procurement contracts designed by the Brazilian government—been mobilized in the critical pandemic context to acquire COVID-19 vaccines in Brazil? What role has procurement law played in this context? To what extent do vaccine contracts deal with the intrinsic nature of uncertainty that characterizes innovation, and what lessons can be drawn when it comes to state capacity?
This paper comprises five sections, including this introduction. The second section describes the theoretical approach of contracting for innovation and its effects on risks and uncertainties vis-à-vis the concept of public procurement as an innovation demand policy tool. The third section comprises our research methods based on a document analysis and data collected through interviews with managers and lawyers from Fiocruz. The fourth section presents a sociolegal analysis of the contracts signed by Fiocruz to acquire COVID-19 vaccines. In the fifth section, we present our conclusions and implications for further investigation of this research agenda.
Contracting for innovation and the state capacity
A theoretical approach distinguishing contracts for innovation from the traditional contractual framework arose in legal scholarship during the 2000s (Gilson, Sabel, and Scott 2009; Jennejohn 2011). The main argument is that conventional contract law does not adequately deal with the radical uncertainties typical of the innovative process (Lessa 2011). As a result, the theoretical approach of the contracts for innovation calls, among other things, for a more holistic understanding of the innovative process through legal lenses.
According to Gilson, Sabel, and Scott (2009), the option to vertically integrate activities instead of contracting, a model associated with the conventional industrial organizational theory to mitigate opportunism, is considered an alternative to contracting. Indeed, vertical integration has been a pillar of the theory of the firm since the 1980s, since it prevents strategic behavior. Examples of vertical integration between producers and the acquisition of suppliers date from the US automobile industry in the early decades of the 1920s. Nonetheless, practically speaking, vertical integration has not worked as a solution to innovation when it comes to replacing traditional contracting. This is so because, among other reasons, firms with mutually dependent investments would rather wait for the other firm to invest before getting more favorable terms, which leads to the holdup problem. Integrated firms, in addition, are under the same control, which is not necessarily the case in innovation activities.
There are many arguments favoring the vertical integration model, as this model may mitigate holdups and opportunistic risks. Another mechanism in favor of vertical integration is modularity, which means separating production into stages or modules, and this organization, in turn, can reduce investment. The modularity mechanism, however, can lead to a situation in which a firm avoids taking risks and maintains its production based on no longer competitive technologies, which has been called a “modularity trap” (Chesbrough and Kusunoki 2004). In fact, since the 1980s, several firms opted for disintegration, and peculiar contracts have been used as tools to codesign and collaborate with other firms (Teece 1992; Vonortas 1998). The Japanese automobile industry in the 1990s, based on subcontracting from producers and suppliers, was a turning point away from the integration model to new forms of contracting (Gilson, Sabel, and Scott 2009). It is well known that in science and technology-based industries, there are multiple examples of collaborative platforms among firms (e.g. development of airplanes, mobile phones, etc.).
A new institutional economics transaction cost approach paved the way to understanding a firm's dilemma of integration or contracting (Wiliamsson 1985). Due to the recognition of bounded rationality and opportunistic behavior, contracts could mitigate risks and uncertainties, becoming, at least, the less costly option (Coase 1988). Nevertheless, transaction cost economics does not address technological uncertainty as a turning point in the contractual analysis. Aiming to fill this gap, Gilson, Sabel, and Scott (2009), Jennejohn (2011), Hart (2009), and Lessa (2011) theorized about contracting for innovation. Given the challenges of modulating uncertainties associated with the innovative process in the traditional contract theory, Gilson, Sabel, and Scott 2009 (GSS, hereinafter) proposed the “contracting for innovation” approach as follows: “contracting for innovation supports iterative collaboration between firms by interweaving explicit and implicit terms that respond to the uncertainty inherent in the innovation process: The inability of the parties to specify ex ante the nature of the product to be produced or its performance characteristics means that the terms of performance will be determined by the very governance process the contract creates” (Gilson, Sabel, and Scott 2009, 4)
As one can see, contracts for innovation are more closely related to collaborative contracts, and one of their striking features is that they are not governed by top-down mechanisms or command and control elements that typically incorporate the principal-agent approach. They are rather a codesign iterative collaborative agreement with information sharing in a transparent manner between the parties. Jennejohn (2011) argues that these contracts create a framework for learning and “not for bureaucracies.” Information-sharing clauses and mechanisms for collaboration, codesign, and learning are, in other words, key elements in contracts for innovation.
Relational governance, in this context, is taken as a viable tool for understanding collaboration through contracts (Grandori 2006). The relational contracts theory is based on informal cooperation and the noncontractual social and network bonds to support collaboration reducing opportunism. In general, a relational contract involves long-term transactions embedded in complex relations, which requires understanding and recognition of enveloping relations (Macneil 2000). However, Sabel and Zeitlin (2004) argued that the pure relational contracting theory does not explain the complex contracting innovation framework.
While modular and relational collaborations do not fully attend to the disintegration process, GSS suggest that what they call iterative collaboration can connect firms. Iterative collaboration—the active interaction of the parties based on reiterated actions and information sharing—is closer to a cooperative mechanism designed to facilitate learning, which is not necessarily enforceable and does not obey hierarchical ordering. The iterative codesign, as a continuous and reiterated method, is commonly observed in research and development agreements as a first step of cooperation between the contracting parties, they explain.
To provide empirical evidence for the contracting for innovation theoretical framework, GSS examined three sorts of contractual relationships. The first, the Deere–Stanadyne agreement, relates to the supply and purchase of John Deere products and the collaborative process of new product development—this second goal without any enforceable obligations. The second was the Apple–SCI agreement comprising legally enforceable obligations to manufacture and supply a product and collaborate on continual improvements in the product as an unspecific and legally unenforceable obligation. The third case, the Warner–Lambert–Ligand contract, refers to creating a single class of pharmaceutical products, considering this sector's regulatory requirements.
GSS highlight three main common features in all of these contracts. The first one is that the scope of the contract cannot be entirely ex ante-specified. In the second place, neither party has the capacity to develop or specify the contract output (product or service) requiring codevelopment. The third aspect is that the developing process must be iterative. Summing up, the parties need to communicate and cooperate to achieve the contract goal since the demanding one cannot fully specify what she wants to contract. There is also a need to continuously follow up on the research and development steps. Thus, contracting for innovation is about dealing with uncertainty—which differs from risk, as Knight explained—as the former is characterized by a situation in which fewer variables are known ex ante, thus constraining calculability (Knight 1964, 44).
Considering the contract incompleteness and the challenges of contracting under continuous uncertainty, the literature on contracting for innovation deepened the analysis of the contractual patterns. In short, as a feature, a governance structure established in the contract's design shall adjust constraints during the contract execution. Accordingly, close collaboration between the contracting parties, which enables transparency and full disclosure of information, is the mechanism to eliminate opportunism. Based on case studies and literature-identified contractual patterns, as shown in Figure 1, we analyzed the Fiocruz COVID-19 vaccine procurement contract.
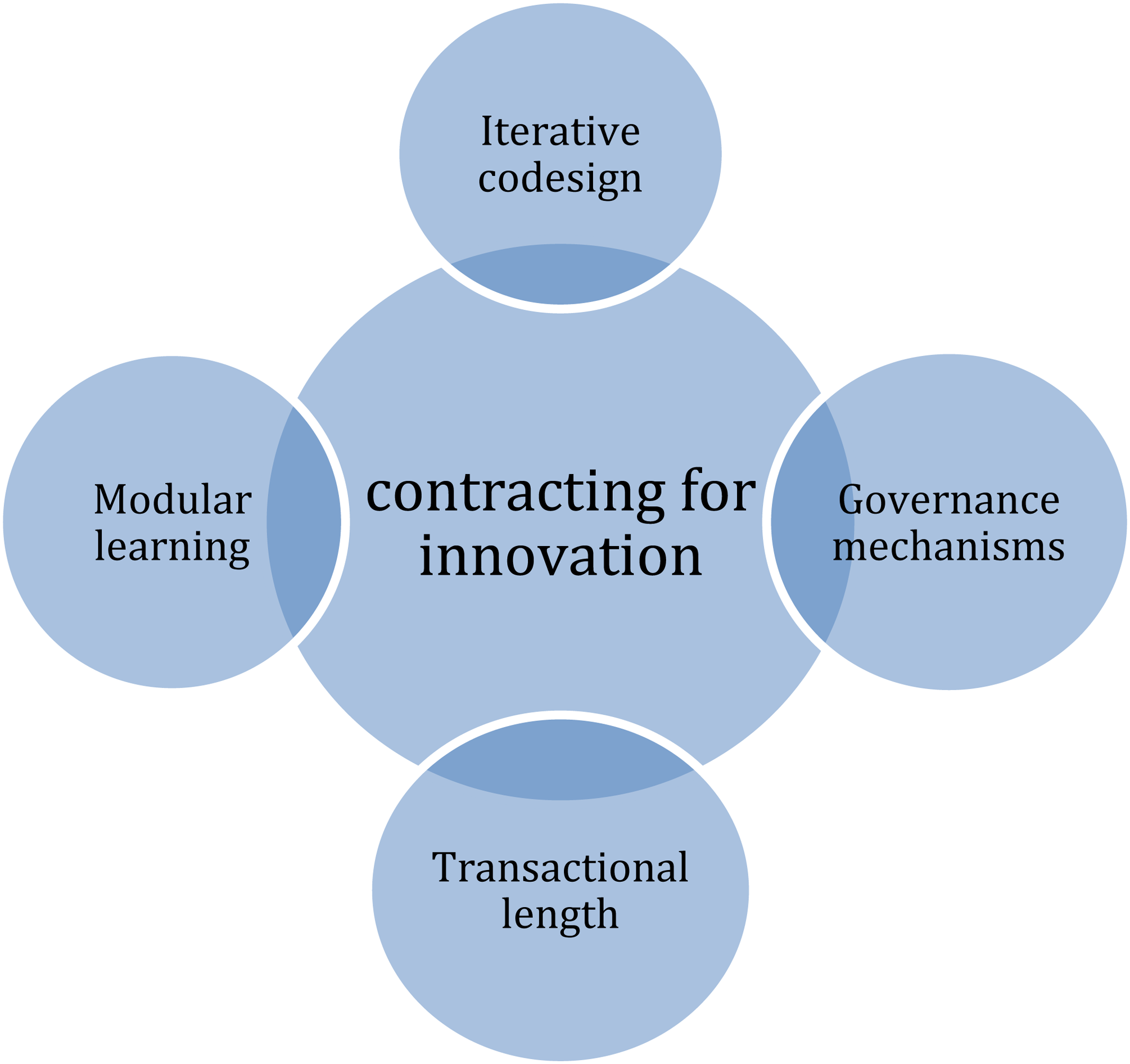
Contractual patterns in contracting for innovation.
Figure 1 sums up the main contractual patterns examined by the contracting for innovation theoretical approach, introduced by Gilson, Sabel, and Scott (2009). Iterative codesign, as mentioned before, presupposes sharing of information between parties in a transparent manner. The governance mechanism aims to deter opportunistic behavior, establishing conditions, for instance, to appoint a referee to solve disputes during the ongoing contract or shifting costs between parties. The transactional length refers to the contractual duration, as engaging in a long-term collaboration essentially differs from short-term collaboration. Finally, drawing on the organizational modularity method, “modular learning” is a mechanism to organize and decompose the contract into modules or subunits, making learning through the agreement easier and more manageable. These contractual patterns are pillars for contracting for innovation literature and will be further analyzed through this paper.
Another way to interpret contracts is through their explicit and implicit elements. The latter is legally binding and enforceable (e.g. price and payment conditions and termination clause) by a third party, such as a court. On the other hand, the implicit contract is hard to monitor by a third party, becoming a self-enforcing measure (e.g. a precontractual negotiation or a performance score of suppliers after the contract execution, reputational effect). In general, implicit and explicit contracts are braided, and their incentives and conditions vary according to the specific agreement.
Despite the emerging literature that identifies contracts for innovation as a distinguished category, the empirical basis of this argument is limited to interfirm contracts governed by corporate law. Complex and innovative as it may be, it lacks reference and consideration of the state's role in the contracting for innovation theoretical framework. This gap can be seen as a paradox when contrasted with the National Systems of Innovation approach, which emphasizes the government's duty to promote science and technological development (Lundvall 1992; Nelson 1993). Moreover, the recovered mission-oriented policy rationale reveals the relevance of public procurement and a broad range of public–private partnerships to induce innovation and economic development. COVID-19 vaccine procurement contracts in Brazil shed light on the intricate public and private interactions and motivate this paper's analysis.
PPI as an innovation policy mechanism
The technology order agreement is a public procurement legal tool for innovation foreseen by the 2004 Brazilian Innovation Law, but it is still underused. The Fiocruz COVID-19 vaccine contract is the first actual test of this tool in the health sector. Despite the limitations of examining ongoing contracts (due to confidential information limitations), we consider it a valuable example of bridging the existing gap between contracting for innovation and the National Systems of Innovation theoretical approaches. In this section, we present the PPI as a demand-side innovation policy tool that has been used in COVID-19 vaccine purchases.
In early 2000, a theoretical approach focused on understanding the state’s role in stimulating economic development through innovation policies developed in Europe (Aschhoff and Sofka 2009; Edler and Georghiou 2007; Edquist and Hommen 1999; Edquist et al. 2000, 2015). The first diagnosis was that the innovation policies, until that time, did not promote expected technology upgrades, despite continuous R&D expenditures. Lundvall (2007) called this diagnosis an innovation paradox in Europe. One response to this paradox came with the demand-side innovation policy theory.
The demand-side policy constitutes a theoretical approach aiming to understand the role of the innovation policy and how it impacts the creation and diffusion of innovation (Edler 2009; Suhonen et al. 2019; Uyarra and Flanagan 2009; Uyarra et al. 2017). According to Edler et al. (2016), demand-side public measures can stimulate innovation, generation, and diffusion, increasing demand for innovation and the articulation of the innovation system institutions through the use of state purchase power, regulation, and other means. In other words, this approach differs from the traditional innovation policy interventions primarily derived from the supply side (e.g. fiscal incentives, grants, support for R&D, and others) (OECD 2011). Nevertheless, the classification of demand and supply-side mechanisms is not tight, and combination and interaction of these mechanisms are becoming increasingly evident (Edler and Yeow 2016; Uyarra and Flanagan 2009; Uyarra et al. 2017).
Considering the rationale of innovation policies, the systemic approach inspired the demand-side policy studies. The systemic view, linked to the National Systems of Innovation concept, opened new perspectives to understand how science, technology, and innovation produce socioeconomic growth. Hence, the literature on innovation systems aims to examine all economic, social, political, institutional, and organizational factors related to innovation creation, diffusion, and use (Edquist 1997).
The use of state purchasing power is typically classified as a demand-side policy mechanism. Public procurement accounts for 12% of the gross national product (GNP) of the Organization for Economic Co-operation and Development (OECD) countries (OECD 2019). Although the public procurement spending relevance and its use by different governments worldwide, using state power to purchase and contract innovative products and services is not widespread in the policy portfolio. PPI implementation fails in disconnection with the local demand, showing that the one-size-fits-all model is not proper for implementing this innovation policy tool, as Uyarra and Flanagan (2009) warned. In addition, we argue that legal and institutional constraints affect PPI design and implementation, deterring public actions and interventions, as we explained in the following Brazilian case analysis.
It is worth mentioning that public procurement is usually set based on the lowest price criterion. However, PPI opens a new perspective for government procurement; since the innovative product or service is not on the market, comparing price and technical specifications are not proper criteria. In this sense, the contractor can procure goods and services targeting social, economic, innovation, and environmental goals through public organizations (i.e. through new technical solutions, products, and services) or expect it through spillover effects (i.e. creation of new markets, generation and diffusion of innovation, and support to strategic missions).
Research methods
We have analyzed data regarding the procurement contract signed by Fiocruz with AstraZeneca. Fiocruz disclosed partially content of the memorandum of understanding, the technology order agreement, and the technology transfer agreement signed with pharmaceutical laboratory (Fiocruz 2020, 2021). The documental analysis was based on these partially–publicly accessible documents, webinars, and further publications regarding the Fiocruz procurement contract negotiation (Medeiros et al. 2022). Although Fiocruz had created channels of communication about the procurement contract, in addition to the press and other scientific disclosures, the detailed analysis of contractual terms and conditions was restricted due to limitations of confidentiality agreed between the parties.
In addition to the documental analysis, we conducted semistructured interviews (through videoconference) with six managers and lawyers from Fiocruz comprising the following three topics
2
:
understanding the connection of the respondent with the vaccine procurement actions carried out by Fiocruz, understanding how implementing the technological order provides a way to innovate in public procurement, and understanding how PPI can lead to a technological absorption leap.
The interviews aimed to identify feasible connections between the respondent activities concerning the procurement contract negotiation and the contractual patterns that the contracting for innovation theory sets out. The fact that we did not interview the AstraZeneca staff directly involved with this vaccine contract was offset by the fact that Fiocruz, as a contractor and public-owned foundation, was the one who proposed the contractual terms and conditions. Additionally, the access to additional information about the negotiation, for instance, via webinars and informal conversations with public attorneys and technical staff of other Brazilian government institutions, enabled us to unveil how this public procurement contract embedded the contracting for innovation patterns.
The COVID-19 vaccine procurement contract in Brazil
This section provides a glimpse of the Brazilian PPI system. The main focus is understanding the origin and characteristics of the technology order agreement, the PPI contract used by Fiocruz to acquire COVID-19 vaccines. Besides this, through document analysis and information gathered in the interviews we conducted, we propose categories of analysis of the Fiocruz COVID-19 vaccine contract based on the contracting for innovation patterns, i.e. modular learning, iterative codesign, governance mechanisms, and transactional length.
Overview of the technology order agreement
The contract signed by Fiocruz with the biopharmaceutical AstraZeneca was a technology order agreement, one example of a PPI contract in the Brazilian legal framework. This legal instrument—based on complex institutional arrangements of governance and monitoring, foreseen by the Innovation Law (Law 10.793/2004)—suits for contracting research and development and innovation activities with technological risk “to solve a specific technical problem or obtain an innovative product, service or process” (article 20). In other words, the purpose of the technological order is to contract a solution for a specific technical problem or obtain an innovative product, service, or process whose developments will involve technological risk (uncertainty assumed when contracting innovation, given that the innovative process may not lead to the expected result) (Brasil 2004).
The state, through federal, state, and city-level entities, can contract companies, individually or in a consortium, or scientific, technological, and innovation institutions (ICTs) (public entities or nonprofit associations or foundations governed by public or private law), which are focused on research activities and recognized technological capacity in the sector in which they operate. The technology order agreement regulates the state's purchasing power in a situation of substantial uncertainty regarding the object to be contracted. For this reason, it could be used to purchase, for example, COVID-19 vaccines that were still in development during the procurement contract negotiation. Nonetheless, it is unknown whether the research and development efforts will actually result in an effective vaccine or how long it will take for the contract object to be executed. Neither is it possible to specify ex ante the resources needed to place the order. In this context, the technology order agreement was specifically designed to deal with uncertainties, severe doubts, and the possibility of many failures amid some success stories (Foss et al. 2021; Rauen and Barbosa 2019). It is also worth noting that the use of this instrument to purchase vaccines by Fiocruz did not prevent the Brazilian government from parallel negotiations for acquiring other vaccines in the international market (Rauen 2020).
The Brazilian government procurement system is an overprescriptive set of norms and regulations with limited opportunities for new and flexible arrangements. The Brazilian Public Procurement Law (Law 8.666/1993), enacted in the 1990s with the scope of organizing the government contracting system at that time, was already weakened by fraud and corruption scandals (Foss and Bonacelli 2022). A new Public Procurement Law (Law 14.133/2021) came into force in April 2021 but did not affect technology procurement contracts directly. Still, it established new modalities of public–private contracting (e.g. competitive dialogue) and new innovation-friendly conditions for the already set bidding mechanisms (e.g. prize contest allowing the assignment of the intellectual property rights to the individual not exclusively for the contracting party, the state entity). Due to this background, contracting with the Brazilian government at federal, state, and city levels is complex, surrounded by uncertainties and risks. Notwithstanding the general procurement system, the technology order dispenses bidding, although it obeys a specific proceeding established by law (Decree 9.283/2018).
Technology order agreements are foreseen in the Brazilian Innovation Law and related laws, but they are still a new and underused tool. Therefore, understanding the technology order proceeding steps is crucial, and Figure 2 aims to explain this PPI journey.
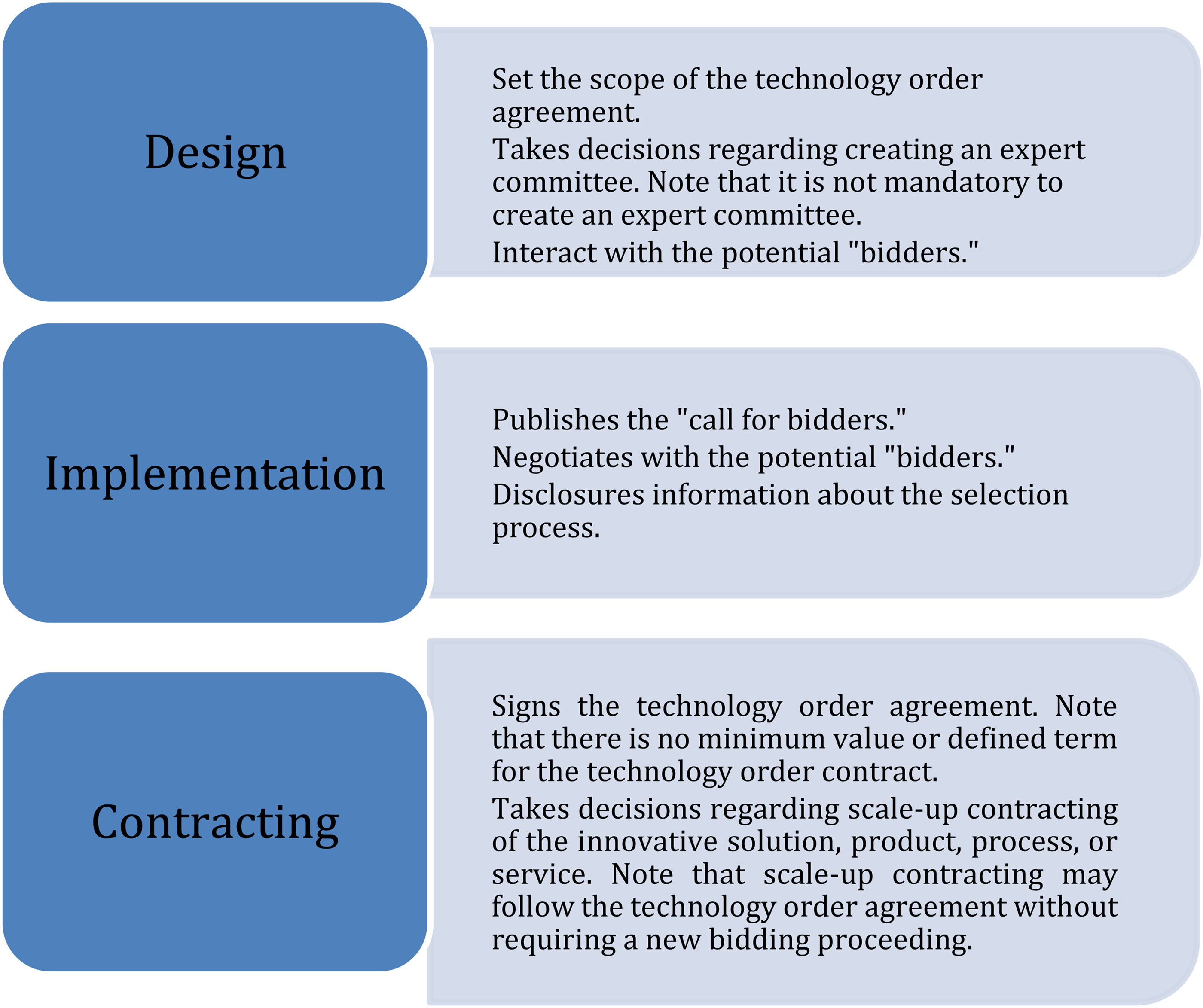
Technology order agreement steps.
Figure 2 shows the steps of the technology order agreement, but not all the steps are followed in the signed contracts. Nevertheless, the systematization of the proceeding steps and references from the PPI instruments supports their use dissemination. 3 Given the specific conditions that characterize the cases of technology order agreements, they do not replace the procurement modalities used by Brazilian public entities. Quite the contrary, technological order contracts allow the state contracting entity to bargain risk and rewards in procurement since it enables contract R&D efforts, which will not necessarily result in a new product, service, or process (Rauen and Barbosa 2019). In 2013, a systematic review of the Brazilian S&T legal framework modified the Innovation Law, the science, technology, and innovation act enacted in 2016 (Law13.243/2016), changing several science, technology, and innovation laws and reinforcing the technology procurement contract, such as the PPI instrument. However, the Innovation Law demanded further regulation, which came into force only in 2018, based on Decree 9.283.
Technology order agreements are public–private procurement instruments for performing R&D activities involving technological risk, solving specific technological problems or challenges, or obtaining an innovative product, service, or process (Brasil 2018). Technological risk, therefore, is a constitutive part of the object of the technology order contract. According to Decree 9.283/2018, it is defined as the “possibility of failure to develop a solution, resulting from a process in which the outcome is uncertain due to insufficient technical-scientific knowledge at the time the decision is taken to perform the action” (Brasil 2018). Technological risk is required to design and implement technology order agreements, although it is well known that the innovative process involves risks and uncertainty.
Risks and uncertainties permeate the innovation process, and the contracts assume a key role in these factors’ mitigation. As the literature on contracting for innovation points out, several innovative firms choose to contract instead of integrating. In essence, contracts are legal tools that aim to mitigate risks, agreeing with the parties’ will to achieve a specific goal (Roppo 1988). Nevertheless, contracts demand enforcement, and the state is in charge of this task. Enforcement assures greater security for organizations and individuals to enter contracts since it creates mechanisms to deal in case of noncompliance of obligations by one of the parties, misconduct or deviation, or case of anticipated contract termination, among other possibilities. In short, enforcement ensures the predictability of contracting, which is crucial to mitigate risks and uncertainties (Dequech 2000).
One mechanism to measure the risks and maturity level of the technology during the design of a project and contract is the technology readiness level (TRL). The TRL scale came from the space sector, created by the National Aeronautics and Space Administration (NASA) as a benchmark to measure technology maturity. Due to its origin in the space sector, the scale has limitations for implementing in other sectors (for instance, in the software sector, as argued by Foss et al. 2021). Still, Brazilian public entities currently use TRLs to support their decisions to buy or contract innovative products and services (e.g. the Brazilian Space Agency (AEB) technology order contract and the Fiocruz COVID-19 vaccine contract) (AEB Brazilian Space Agency 2021).
Learning and experimentation from the health innovation procurement
Considering the technology order agreement as an example of contracting for innovation, in this section, we discuss our main findings regarding our primary data (documents and interviews) and the contracting for innovation patterns. In the first place, the COVID-19 vaccine contract is a relevant experimentation in a significant health public procurement market. However, using state power to acquire products and services is not new in the Brazilian public health system.
The COVID-19 vaccine contract signed by Fiocruz with the pharmaceutical company AstraZeneca was the first technology order agreement entered into by the Brazilian health public system. Notwithstanding the incipient use of PPI, the Brazilian public health system (SUS) is quite experienced with the public procurement system. The expenditures from public and private health sectors in Brazil in 2019 were 9.6% of the gross domestic product (GDP). SUS has been continuously struggling, although it attends to ∼70% of the Brazilian population and supports around 90% of the expenditures with prevention (including vaccines), promotion, and health surveillance (Brasil 2022).
Public procurement is substantial for the Brazilian public health system. Varrichio (2017) shows an increasing expense with procuring medical, dental, and veterinary equipment. For instance, the expenditure on the procurement of health equipment grew from R$6 billion in 2009 to R$16 billion in 2015 (Brazilian reais). Likewise, spending on procurement of medicine has also been increasing, reaching R$9.1 billion in 2015 (Brazilian reais) concerning public–private partnership purchases (Varrichio 2017, 186).
The National Immunization Program (PNI), which was created in 1973, is a public health service offered to all Brazilians, regardless of whether or not the user has a private health plan (Brasil/Ministry of Health 2003). For a significant part of the population, the prevention and surveillance public platform is the only route to access the health system (Brasil 2022). The public laboratories, Fiocruz and Butantan, have key roles in the Brazilian public health system, and, concerning vaccine production, not taking the COVID-19 vaccine into account, they are one of the 15 top vaccine producers in the world, according to the Global Vaccine Market Report (WHO 2022).
Another public procurement instrument for innovation is the public–private partnership with technology transfer (PDP). A PDP is a public procurement mechanism connected with public policy goals initially designed in 2008 within the scope of the Executive Group of the Industrial Health Complex (GECIS), although PDP became a policy tool only in 2012 (Varrichio 2017). The scope of the PDP mechanism was to use state purchasing power with technology transfer, allowing the domestic production of strategic inputs (including pharmaceutical products, such as medicines and vaccines) for the Brazilian public health system.
A PDP is entered into by a foreign pharmaceutical company, the technology owner, a public laboratory responsible for absorbing the technology and manufacturing the product at the end of the PDP technology internalization phase, and a public laboratory or a private company, national developer, and domestic producer of the active pharmaceutical ingredient (API) or critical technological component (Brasil 2014). The public procurement contract, the object of the PDP, is based on the selection of proposals from a list of strategic products published by the Ministry of Health.
Although there are approximations of the PDP model to the framework of PPI, as Pimentel (2018) points out, the PDP does not replace the technological order since the latter aims to acquire a product, process, or technical solution still under development, while the former seeks to rationalize the use of state purchasing power and provide autonomy for the local production.
Turning now to examine the Fiocruz COVID-19 vaccine contract, we refer to contractual patterns identified by Gilson, Sabel, and Scott (2009): governance mechanism, iterative codesign, modular learning, and transactional length.
This section analysis comprises the information disclosed from the technology order and technology transfer contracts signed by Fiocruz, Bio-Manguinhos, and AstraZeneca. As mentioned before, those agreements are partially publicly available due to confidential restrictions. References to answers we obtained in the interviews are provided below.
Governance mechanisms
The technology order agreement between Fiocruz, Bio-Manguinhos, and AstraZeneca foresees the provision of internal monitoring mechanisms and inspection of the execution of the contract through a “steering committee,” a sort of board of directors appointed by both parties (clause 6). The constitution of the steering committee is not the same as the “technical committee of experts” since two representatives of each party (Fiocruz and AstraZeneca) create the former, and the latter is formed by the contractor staff in the first steps of the technology order negotiations as Figure 2 shows.
This contractual provision of a “steering committee” indicates a sophisticated governance arrangement, whereby the parties monitor the contract implementation—and the execution—of the procurement in practical terms. It is worth noting that such a contractual governance structure transcends the usual bureaucratic mechanisms of public contracts in Brazil. However, one respondent pointed out that this type of governance mechanism is typical in health technology transfer agreements entered by Fiocruz. Three respondents said that governance mechanisms were key to the technology order contract, mentioning even actions taken by Fiocruz before the contract signature. Two respondents emphasized that an internal committee conducted a foresight study concerning the potential COVID-19 vaccines that could be incorporated into the public laboratory's immunization production infrastructure in the shortest possible time.
One projected scenario was in case of an earlier contractual termination due to unsuccessful results in phase 3 of the vaccine trial (technological risk, section 9.15 of the technology order agreement). One key manager respondent made it clear that this was risky contracting (referring to innovation uncertainties), having the foreign pharmaceutical laboratory the right to receive its payments regardless of the delivery. The payment agreement was adjusted to a fixed price per dose, which became the lowest COVID-19 vaccine price negotiated by the Brazilian government (Medeiros et al. 2022).
The dispute resolution clause is consistent with the risks and uncertainties of the contract and assumes the search for an amicable solution between the parties, anyhow, supposing the parties do not agree on a consensual solution. In that case, they may proceed for dispute resolution in the Federal Court of Rio de Janeiro, where Fiocruz and Bio-Manguinhos Institute headquarters are.
Transactional length
Public laboratories, such as Fiocruz and Butantan, depend on APIs from other countries to produce vaccines and medicines (Paranhos and Perin 2021). Delays in importing APIs and consequent constraints on domestic production seriously harm the vaccination program. In the COVID-19 context, these delays and noncompliance duties were feared. Contractual mechanisms partly deal with such risks by stipulating penalties and liabilities to the infringing party. However, contracting innovative products adds intricate layers of uncertainties to the traditional contract theory. An example: during the contract negotiation, the contracted object is not previously known because the innovative product, process, or technical solution is still being developed. Considering the risks and uncertainties of the innovative process and the political tensions that sadly hampered vaccine contract negotiations (Massard, Shadlen, and Bastos 2021), the transaction length of the Fiocruz PPI—from the negotiations to the signature and delivery of the vaccines to the population—was atypical (Cossenza et al. 2022).
After the World Health Organization decreed the pandemic in March 2020, Fiocruz and AstraZeneca signed the Memorandum of Understanding on 31 July 2020 and on 9 September 2020 signed the technology order contract. 4 In less than 1 year after the pandemic began, the Fiocruz vaccine (the same occurred for the Butantan vaccine) was already submitted for emergency approval by the Brazilian Health Regulatory Agency (Anvisa) (Cossenza et al. 2022).
Obtaining regulatory approval in a such short period was also a significant achievement following the Fiocruz COVID-19 vaccine contract. Before the emergency approval by the Brazilian regulatory agency, the research and development (R&D) phase had the support of the Medicines and Healthcare product Regulatory Agency (MHRA) from the United Kingdom (Maia et al. 2022, 150). During the technology order contract negotiation, two respondents stressed that regulatory matters are strictly followed in the biohealth sector, and Fiocruz has a technical staff that is used to dealing with these issues. Therefore, their previous technical training and preparedness were crucial for promptly handling technical and regulatory requirements during the contract negotiation. Regulatory affairs and limitations and protection of intellectual property rights (including the sublicense agreement by Oxford University, the patent owner to Fiocruz) were in the spotlight; meanwhile, the technology order agreement and the technology transfer agreement were negotiated.
Modular learning
Fiocruz has a long history in negotiating complex and intricate public–private agreements with domestic and foreign pharmaceutical companies. This background added to a skilled workforce that met the challenge and performed their activities; despite disagreements with the Jair Bolsonaro administration, which did not lead this negotiation and made erratic decisions in the fight against the COVID-19 pandemic, they were decisive in putting a new PPI contract into practice.
The contract between Fiocruz and AstraZeneca establishes the technology transfer for vaccine production because the final product packaging and preparation occurred at the Bio-Manguinhos laboratory. The scope of the PPI includes the scale-up (development of the process on an industrial scale) of the production of the API by Fiocruz or a third party, in addition to the supply of the API, via import, for the production of 100.4 million doses of the vaccine (Fiocruz 2021). The decision to segregate the technology transfer agreement from the PPI contract was strategic. One respondent emphasized that the first goal was to obtain the API to guarantee the domestic production of the vaccines and then enter the technology transfer agreement, following terms and conditions already negotiated by Fiocruz.
The technology transfer agreement, signed on 1 June 2021 and partially released by Fiocruz, also has provisions connected with the aforementioned contractual patterns. 5 This contract scope included licensing the API or the drug substance of the vaccine from AstraZeneca to Fiocruz. The technology transfer was carried out in stages, initially involving the API filling and later its production internalization. It contains governance mechanisms similar to the technology order contract, enabling Fiocruz and AstraZeneca to review, discuss, propose, and approve strategies related to the supply of the licensed product, manufacturing, regulatory plans, and even the donation of the licensed product. The parties agree to appoint representatives to join the joint steering committee, as already laid down in the technology order agreement. Also, the parties agreed upon ex post provisions regarding the supply of the licensed product, confidentiality, and further developments and sublicensing. An essential condition for transferring technology was the Bio-Manguinhos infrastructure, where the vaccine was manufactured. Technology transfer will not be accomplished without this infrastructure, which requires public investment.
Iterative codesign
Based on the interviews with Fiocruz staff, all respondents mentioned how challenging it was to procure a COVID-19 vaccine and enter into a new kind of procurement during the most significant deadly contagious disease emergency in recent human history. The first draft and the negotiation premises were set by Fiocruz, the contracting party, but all respondents emphasized that these fast-track contract negotiations were deeply and intensely discussed between the parties. Additionally, beyond the contractual parties, one respondent mentioned that an expert committee and public lawyers worked together with the Fiocruz staff.
As mentioned before, the approach of the technological risk as an inherent part of the PPI is a distinct feature and was regulated in the contract signed by Fiocruz in case of contract termination. In other words, the contract expressly stated that the contractual object's development was uncertain and involved technological risk since the clinical trials might not achieve the expected results (and the vaccine development might have failed). In this sense, if the delivery of the APIs were not possible due to an event that constituted an obstacle (the materialization of a specific technological risk), the contract could be terminated upon duly grounded justification and agreed upon by the parties.
Additionally, measures adopted by Fiocruz for disclosing information on the progress of negotiations, as well as partial disclosure of signed contracts, ensured transparency. At the same time, such measures sought to enable some degree of contracting monitoring by the media, society, and control government bodies.
Another aspect relevant from the design legal perspective mentioned by one respondent was the intellectual property negotiation. The University of Oxford is the intellectual property owner (patent rights) of the vaccine ChAdOx1 nCoV-19. AstraZeneca signed a license agreement with the University of Oxford to produce the vaccine. Fiocruz became the sublicensee, to which AstraZeneca transferred the API technology for domestic production. As a sublicensee, Fiocruz became responsible for third-party rights infringement and other matters that should be addressed in the PPI contract and the technology transfer agreement, taking additional risks compared to contracts celebrated for the technology transfer of finished products. Therefore, one respondent highlighted intellectual property rights as a hot topic during the technology order agreement negotiation. Ultimately, these risks and concerns on intellectual property clauses lead to a process of learning and state capacity development in the Brazilian public laboratory.
Conclusions
This paper is a first attempt to review, discuss, and reconcile the theoretical frameworks of contracting for innovation with PPI in a broad sense. As stated above, there is a gap in the contracting for innovation literature regarding the state's role in these agreements. This is a paradoxical gap, we believe, since science, technology, and innovation studies have clearly appointed the crucial role to the state in the national innovation systems, and we understand contracting for innovation as a framework that goes beyond the segmentation of public and private law. Besides identifying this gap, this article aims to discuss the possible adherence of the contracting for innovation theoretical framework to an emblematic case: procuring a COVID-19 vaccine in a developing country.
The actions of the Brazilian state in response to the COVID-19 pandemic, notably in the vaccine acquisition, were admittedly uncoordinated and, to put it crudely, disastrous. Nevertheless, Fiocruz, a Brazilian public laboratory, staffed by career public servants supported by federal attorneys and other PPI experts, negotiated and contracted COVID-19 vaccines in 2020. The technology order agreement and the technology transfer agreement signed by Fiocruz with the pharmaceutical AstraZeneca have similarities with the contracting for innovation patterns and typical clauses. In short, we noted that the Brazilian state, through Fiocruz staff, along with the science, technology, and innovation laws and regulations, played key roles in COVID-19 vaccine contracting. The respondents stressed how public laboratory preparedness was the key to address the COVID-19 vaccine demand—from their deep prospection phase on vaccine candidates, throughout contract negotiation and local production.
This paper’s findings show that the innovation legislation in force in Brazil was mobilized to design and draft sophisticated types of public–private agreements—the technology order agreements discussed here—that allowed for urgent acquisition of vaccines during the pandemic despite the very concerning fact that the pandemic-denying federal government proved to be not only incompetent but also inexplicably resistant to pursue a mass vaccination effort in the country. Bureaucrats and lawyers, not politicians, were the ones who managed to commission and buy vaccines in Brazil, being the case of technology order agreements—an innovation-related tool—an important example of (legal) state capacity for development.
Returning to our research question, we conclude that the PPI vaccine contract signed by Fiocruz contained adequate provisions to mitigate the uncertainty involved in this endeavor. This conclusion is closely related to the contractual patterns introduced by the contracting for innovation theory. For example, we highlight the strategic decision to negotiate the technology transfer of a vaccine still under development and its subsequent incorporation into the Brazilian laboratory's technology portfolio. Lessons in implementing a PPI contract during a pandemic can inspire innovation policy missions within, but not limited to, the health system.
Footnotes
Acknowledgments
The authors acknowledge support by the São Paulo Research Foundation (FAPESP) in connection to the Thematic Project “Uncertainty Management in Innovation Activities” at the University of Sao Paulo (USP) (Grants 2019/16147-7 and 2015/26662-5). FAPESP is not responsible for the contents of this paper. The remaining mistakes and misconceptions are solely the responsibility of the authors. The authors gratefully thank the researchers Fernanda Costa Lino Noia, João Lucas Sacchi de Oliveira, and Letícia Nunes de Oliveira for their support with bibliographic research and interviews. The authors thank Professor Elize Massard da Fonseca for her suggestions and comments in the previous draft of this paper. Jim Hesson edited the manuscript (AcademicEnglishSolutions.com).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fundação de Amparo à Pesquisa do Estado de São Paulo (grant numbers 2015/26662-5, 2019/16147-7).
