Abstract
Objectives:
To observe the efficiency and safety of direct and indirect three arch vessels’ cannulation for bilateral antegrade cerebral perfusion during total arch replacement.
Methods:
Between 2002 and 2014, 130 patients underwent total arch replacement with direct and 66 patients with indirect cannulation for antegrade cerebral perfusion under moderate hypothermia. Patients were assigned to the direct cannulation group based on the condition of the aortic arch vessels. Demographics and concomitant surgeries were similar in the two groups. Patient characteristics, surgical and haemodynamic measurements, and postoperative neurologic findings were observed.
Results:
Aortic cross-clamping (120 ± 42 vs 139 ± 65 min; p = 0.032) and cardiopulmonary bypass (178 ± 57 vs 206 ± 71 min; p = 0.025) times were lower in the direct cannulation group. There were no differences in overall hospital mortality (4% vs 6%; p = 0.87), selective antegrade cerebral perfusion time (91 ± 52 vs 100 ± 65 min; p = 0.185), and incidence of neurologic deficit (3% vs 9%; p = 0.07). Indirect cannulation led to more frequent re-exploration for bleeding (5% vs 13%; p = 0.0356). There was no difference in midterm survival (log-rank p = 0.103; mean follow-up times: 4.74 years (direct cannulation), 5.50 years (indirect cannulation)).
Conclusion:
Direct cannulation during mild systemic hypothermia yields excellent outcomes while reducing time and rate of reoperation for bleeding and can be used in total arch replacement without increasing morbidity and mortality.
Keywords
Introduction
Total arch replacement is technically challenging and requires complex circulatory management techniques to provide cerebral protection. Although postoperative results are important in all aspects of cardiac surgery, neurologic and mortality outcomes are especially significant in thoracic aortic surgery as these outcomes directly reflect both circulation management techniques and the aortic pathology. Recently, antegrade cerebral perfusion (ACP) with moderate hypothermic circulatory arrest has been proposed as a safe and effective cerebral circulatory management technique by a number of aortic surgeons. 1 Furthermore, this technique allows extending the aortic arch reconstruction time, which permits more complex aortic arch procedures.
The traditional practice at our institution was to provide proper circulatory flow during total arch replacement via ascending aorta or peripheral vessel (axillary or femoral) cannulation in combination with arch trifurcation with indirect cannulation during ACP. Such cannulation may potentially lead to adverse consequences, including the need for an additional incision, nerve injury, localised dissection, and malperfusion. 2
To avoid an additional incision and to streamline operations requiring brief periods of circulatory arrest, we sought to identify a method that would allow proper circulatory flow and good visibility during anastomosis of the distal arch and arch vessels without embolic phenomena. Since 2002, we have performed total arch replacement using direct cannulation of the brachiocephalic artery (BCA), left common carotid artery (LCCA), and left subclavian artery (LSA), referred to as ‘direct cannulation of the arch vessels’, with bilateral ACP (direct cannulation–antegrade cerebral perfusion (DC-ACP)).
Methods
This study is a retrospective single-centre observational study of a series of patients who underwent total arch replacement using ACP. We have observed two different strategies for ACP during aortic arch repair: cannulation of the ascending aorta or peripheral (axillary or femoral) cannulation in combination with indirect cannulation through the ostia of the supra-aortic vessels during ACP (indirect cannulation–antegrade cerebral perfusion (IC-ACP)) and DC-ACP.
From January 2002 to April 2014, 524 patients underwent aortic arch replacement (209: ascending aorta and hemiarch replacement, 315: total arch replacement). In this study, two perfusion techniques were compared in 196 patients who underwent total arch replacement using bilateral ACP.
Patients were assigned to either DC-ACP or IC-ACP preoperatively based on findings of enhanced computed tomography (CT). DC-ACP was selected when the three arch vessels were not calcified and were not located deeply. Direct cannulation method was unsuitable when the subclavian artery has recessed the case and from the body more than 6 cm that they have hidden in the common carotid artery (Figures 1 and 2).
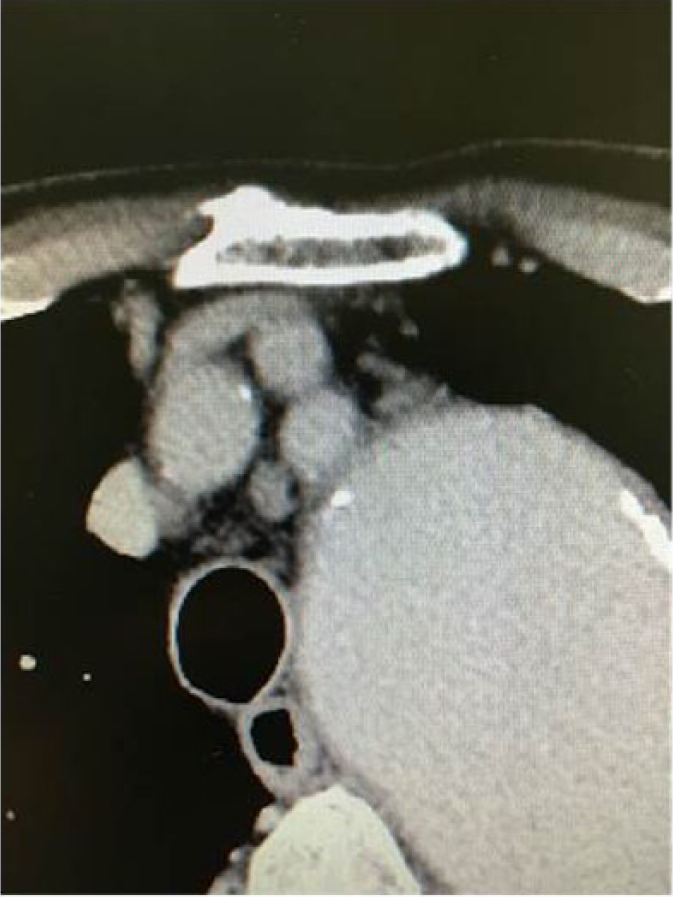
Patients were assigned to either DC-ACP or IC-ACP preoperatively based on findings of enhanced computed tomography (CT). DC-ACP was selected when the three arch vessels were not calcified and were not located deeply.
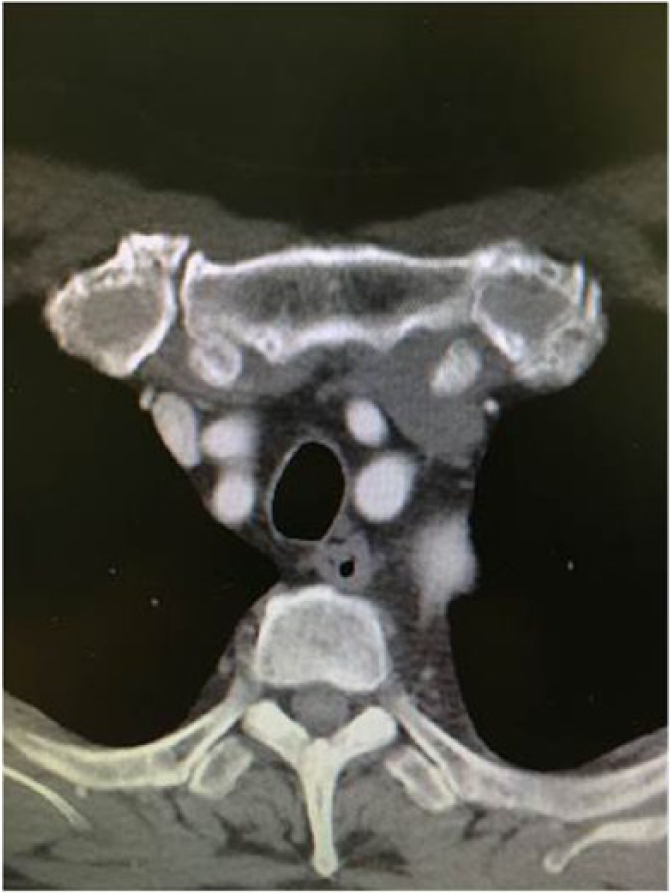
Direct cannulation method was unsuitable when the subclavian artery has recessed the case and from the body more than 6 cm that they have hidden in the common carotid artery.
We observed the outcomes of 130 patients who underwent total arch replacement using DC-ACP and 66 patients who underwent the same procedure using IC-ACP, during which a balloon-tipped cannula was inserted from inside the aorta into the aortic arch vessels individually. Unilateral selective antegrade cerebral perfusion (SACP) or hemiarch replacement was performed in the remaining 328 patients who were not included in the present analysis.
The primary endpoint was in-hospital mortality following surgery, and the secondary endpoints were incidences of transient or permanent neurologic deficit, re-exploration for bleeding, new-onset postoperative renal failure (need of dialysis) and prolonged ventilation (more than 48 h). Temporary neurologic deficit was defined as a clinical neurologic dysfunction that occurred postoperatively without documented evidence of anatomic abnormality by CT or magnetic resonance imaging (MRI) and resolved before discharge. Permanent neurologic deficit was defined as a neurologic deficit that occurred postoperatively with structural abnormality detected by documented CT or MRI. The two groups were matched with regard to sex and the incidence of stroke, diabetes, hypertension, chronic obstructive pulmonary disease, and renal failure.
The research ethics committee of our institution approved this study, and patients provided consent prior to inclusion.
Surgical procedure
Transoesophageal echocardiography performed with the patient in the supine position was used to confirm cardiac and valve function. Temperature probes were placed for tympanic and rectal temperature monitoring. In all patients, median sternotomy was performed. The arterial cannulation site was selected based on preoperative CT and intraoperative epiaortic ultrasonography findings. We mobilised the ascending aorta and arch, isolated the innominate vein, exposed the proximal end, and gently dissected and mobilised the base of the arch arteries. If the ostium of the arch vessel was severely atherosclerotic, the cannulation site was extended distally from the diseased ostium. One purse-string suture using a 5-0 polypropylene thread was placed on the anterior walls of the BCA, LCCA, and LSA. After systemic heparinisation, we cannulated the arch arteries using a paediatric cannula (Figure 3). The diameter of Sorin A272 paediatric cannulae used in the DC-ACP group ranged from 9–10.5 Fr (LCCA and LSA) to ~12 Fr (BCA). A 9- to 10.5-Fr cannula typically provides a maximal flow of 1–1.5 L/min, whereas that of a 12-Fr cannula is 2 L/min, resulting in a total maximal flow of 4–5 L/min. The choice of cannula sizes was at the discretion of the surgeon. The total flow was maintained at 2.2 L/min/m2, and we checked the circuit internal pressure. The flow rate was adjusted to maintain the LCCA, right innominate artery, and LSA pressure within 30–50 mmHg.
DC-ACP technique. After systemic heparinisation, the diameter of Sorin A272 paediatric cannulae used in the DC-ACP group ranged from 9–10.5 Fr (LCCA and LSA) to ~12 Fr (BCA). A 9- to 10.5-Fr cannula typically provides a maximal flow of 1–1.5 L/min, whereas that of a 12-Fr cannula is 2 L/min, resulting in a total maximal flow of 4–5 L/min. The choice of cannula sizes was at the discretion of the surgeon. The total flow was maintained at 2.2 L/min/m2, and we checked the circuit internal pressure. The flow rate was adjusted to maintain the LCCA, right innominate artery, and LSA pressure within 30–50 mmHg. Cannulae were placed through purse-string sutures in the arterial wall.
Cannulae were placed through purse-string sutures in the arterial wall. The initial site of cannulation was the arch of the three vessels in the DC-ACP group. In the IC-ACP group, cannulation sites included the ascending aorta/arch (56%), femoral artery (17%), axillary artery (5%), femoral artery and axillary artery (6%), and ascending aorta and femoral artery (14%). Venous cannulae were inserted into the right atrium. A left ventricular vent cannula was inserted through the right superior pulmonary vein, and total flow was obtained from the three arch vessels. Systemic cooling was started immediately. Systemic cooling was considered adequate for circulatory arrest when the tympanic temperature reached 28°C. Each arch vessel was then cross-clamped, and the aortic arch was opened. This cross-clamping procedure distinguishes the ACP procedure from systemic cardiopulmonary bypass (CPB) (Figure 4).

Cross-clamping procedure of arch vessels. Systemic cooling was considered adequate for circulatory arrest when the tympanic temperature reached 28°C. Each arch vessel was then cross-clamped, and the aortic arch was opened. This cross-clamping procedure distinguishes the ACP procedure from systemic cardiopulmonary bypass.
In the case of deep LSA, in situ cannulation into the LSA without decompression of the aorta using CBP may be extremely difficult. Moreover, the proximal area of the LSA is very often atherosclerotically changed, so for the deep left subclavian case, we select the IC-ACP.
In the IC-ACP group, the aortic arch aneurysm was opened as described by Okita et al., 3 with simultaneous increase in the central venous pressure to 10 mmHg or brief periods of retrograde cerebral perfusion. The inside of the aortic arch was exposed. Next, SACP was initiated. A 14- or 16-Fr balloon-tipped cannula was inserted from inside the aorta into the BCA, and 12-Fr cannulae were positioned in the LCCA and LSA. Self-inflating serrated-balloon-tipped cannulae (Fuji Systems, Tokyo, Japan) were used routinely. Avoiding cerebral embolism was particularly important in selective cerebral perfusion (SCP) cannulation. For the ostium of severely atherosclerotic arch vessels, the arteriotomy of the arch vessels was extended from the diseased ostium to a relatively clear distal part so that cannulae were placed in position under direct vision. Balloon tip pressure was maintained under 30–50 mmHg. In almost all cases, when the tympanic temperature reached 25°C after 10–20 min, the core temperature (rectum) remained at 30°C–32°C. Myocardial protection was ensured by antegrade infusion of cold blood cardioplegic solution directly into the coronary ostium. To prevent left recurrent nerve injury, direct nucleation of the proximal end of the descending aorta was performed in addition to dissection of the descending aorta after dividing the upper intercostal arteries. Distal anastomosis using a 4-0 monofilament suture with felt strip reinforcement was started at the 3-o’clock position and continued in the counter-clockwise direction. A flexible sucker was positioned inside the graft. First, once the distal anastomosis was complete, the aortic line was connected to the lateral limb of the graft, the lower body circulation was restarted through the lateral limb of the graft, and whole body rewarming was started. We performed LSA neck anastomosis using 5-0 monofilament sutures. Proximal anastomosis was performed using 4-0 monofilament sutures. Once the neo-sinotubular junction anastomosis was complete, de-airing the heart, the cross-clamp was removed. Coronary reperfusion was then started, and the heart was defibrillated if necessary. The BCA and LCCA anastomoses were performed after the cross-clamp was removed during the cardiac reperfusion.
Statistical analysis
Continuous data are presented as mean ± standard deviation. Categorical variables were analysed using chi-square or Fisher’s exact tests. Continuous variables were examined using the t-test or the Mann–Whitney U-test. Differences were considered significant at the level of p < 0.05. Kaplan–Meier curves were generated to compare the survival in the DC-ACP and IC-ACP groups. Wilcoxon and Kruskal–Wallis trials with log-rank tests were used for survival analysis. Data were analysed using JMP, version 12 (SAS Institute Inc., Cary, North Carolina).
Results
Baseline demographic characteristics of the patients are listed in Table 1. The mean age was 71.5 ± 15.3 years in the DC-ACP group and 69.3 ± 11.5 years in the IC-ACP group. Aneurysm (containing chronic type B dissection) was identified in 121 patients (93%) versus 59 (89%) and acute type A dissection in 9 (7%) versus 7 (11%). A shaggy aortic arch was present in 15 (12%) versus 10 (15%) patients. The average European System for Cardiac Operative Risk Evaluation (EuroSCORE II) scores did not differ significantly (2.57% vs 2.34%, p = 0.52).
Patients’ characteristics.
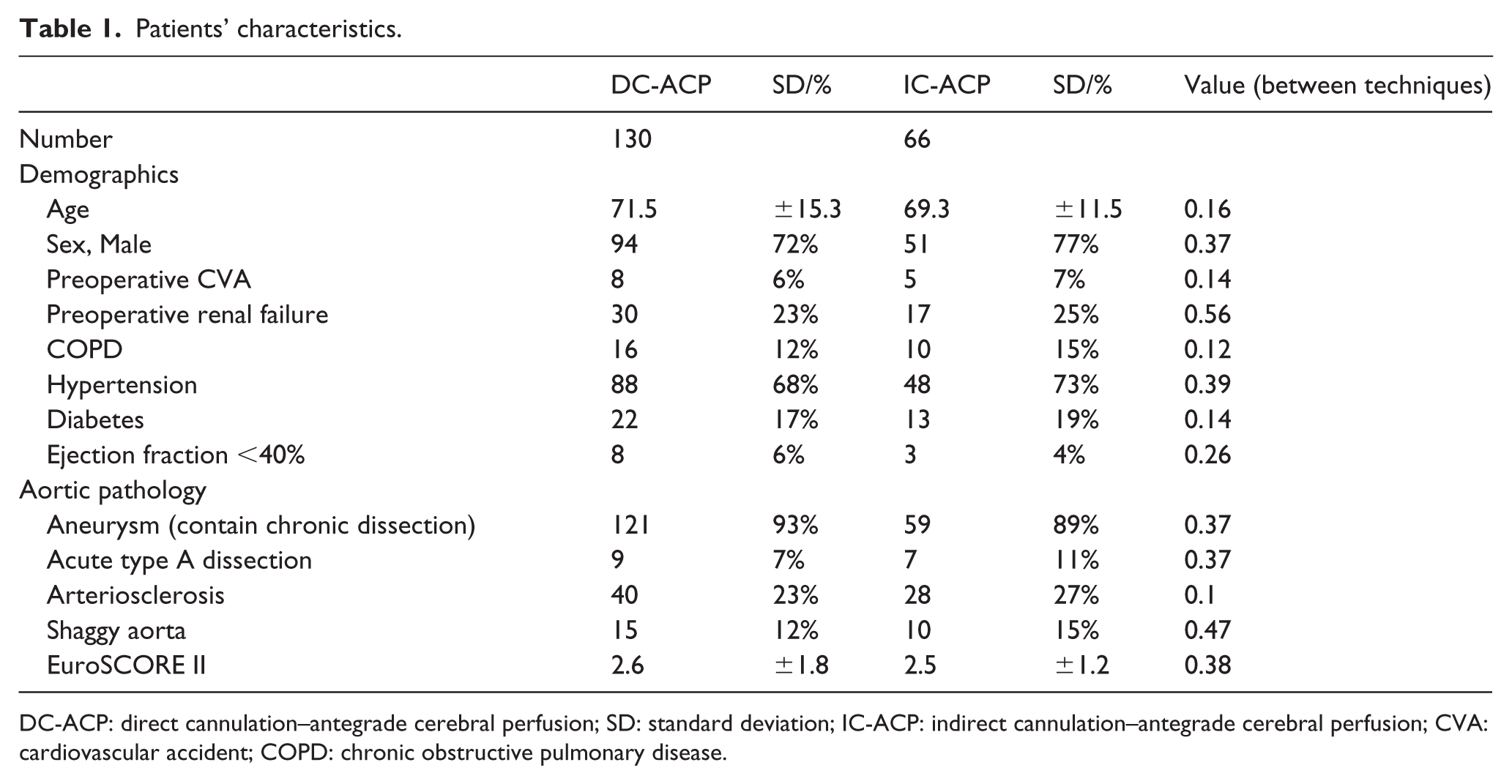
DC-ACP: direct cannulation–antegrade cerebral perfusion; SD: standard deviation; IC-ACP: indirect cannulation–antegrade cerebral perfusion; CVA: cardiovascular accident; COPD: chronic obstructive pulmonary disease.
Surgical procedures for patients undergoing total arch replacement using DC-ACP or IC-ACP and concomitant surgeries performed at the time of total arch replacement are summarised in Table 2. There was no significant difference between the groups in hospital mortality (DC-ACP, 5%; IC-ACP, 4%; p = 0.67). Average SACP times were similar between the groups (DC-ACP: 100 min; IC-ACP: 91 min; p = 0.597). Cross-clamping of the arch vessels is the distinguishing factor between the DC-ACP and IC-ACP procedures. In the DC-ACP group, there was no cerebral circulatory arrest; in contrast, the IC-ACP procedure involved a period of circulatory arrest during the insertion of the cannula from the internal aspect of the aorta.
Intraoperative variables.
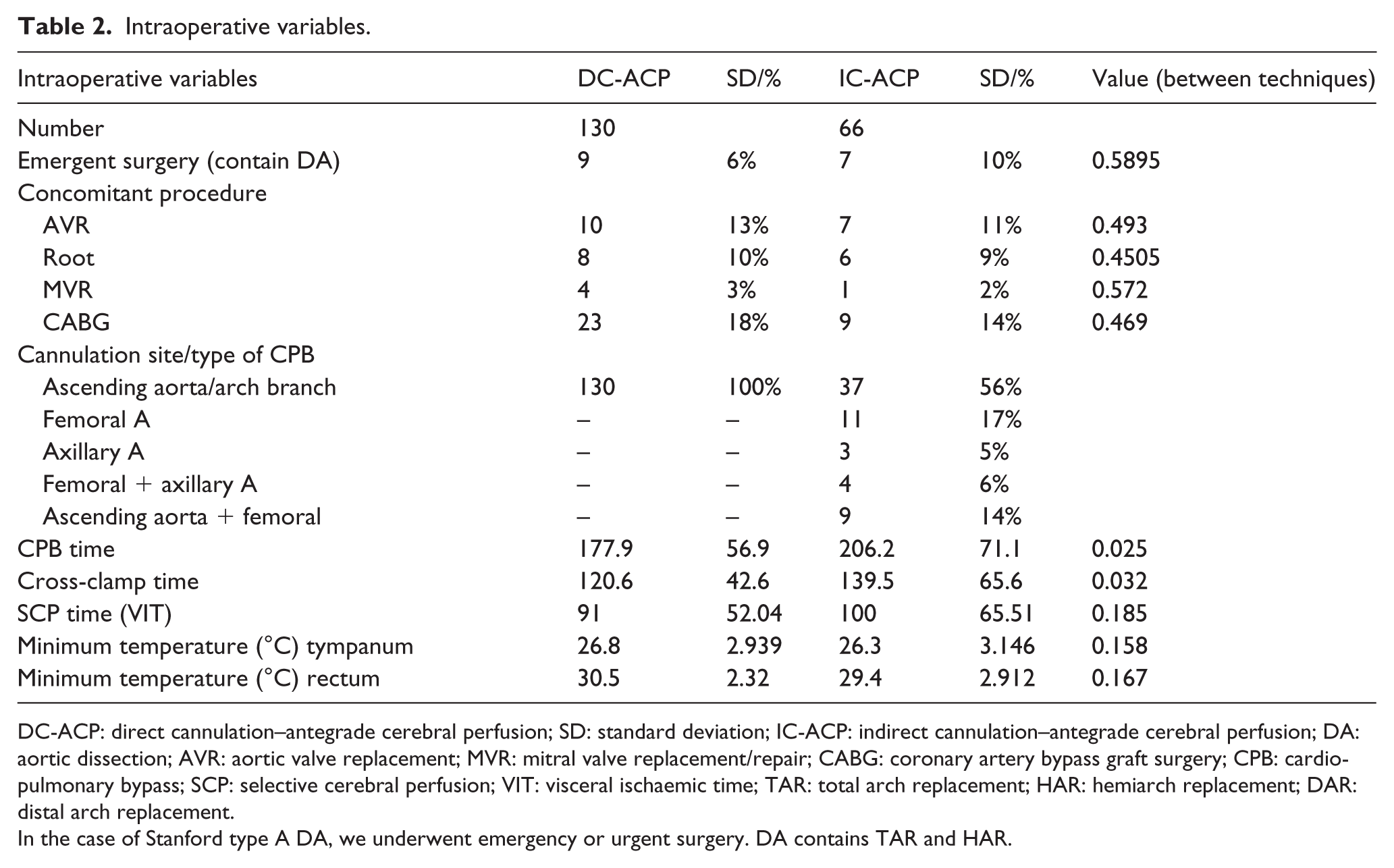
DC-ACP: direct cannulation–antegrade cerebral perfusion; SD: standard deviation; IC-ACP: indirect cannulation–antegrade cerebral perfusion; DA: aortic dissection; AVR: aortic valve replacement; MVR: mitral valve replacement/repair; CABG: coronary artery bypass graft surgery; CPB: cardiopulmonary bypass; SCP: selective cerebral perfusion; VIT: visceral ischaemic time; TAR: total arch replacement; HAR: hemiarch replacement; DAR: distal arch replacement.
In the case of Stanford type A DA, we underwent emergency or urgent surgery. DA contains TAR and HAR.
There were no significant differences in the minimum temperature of the bladder and tympanum between the patients in the two groups (Table 2). There were no significant differences in the in-hospital mortality (DC-ACP, 4%; IC-ACP, 6%;p = 0.88) and the incidence of transient neurologic deficit (DC-ACP, 2%; IC-ACP, 5%; p = 0.47), but permanent neurologic deficit tended to occur more frequently in the IC-ACP group (DC-ACP, 3%; IC-ACP 9%; p = 0.07). There were significant differences in the rates of re-exploration for bleeding (DC-ACP, 5%; SACP, 13%; p = 0.029) (Table 3). Additionally, aortic cross-clamping (120 ± 42 min vs 139 ± 65 min; p = 0.032) and CPB (178 ± 57 vs. 206 ± 71 min; p = 0.025) required more time in the IC-ACP group. The follow-up was complete for 97% of the patients. The mean follow-up time was 4.74 years (1.95–8.13 years) in the DC-ACP group and 5.50 years (2.03–8.56 years) in the IC-ACP group. Midterm survival was not influenced by the cannulation site (log-rank p = 0.103) (Figure 5). After direct cannulation, survival at 5 and 10 years was 92.25 ± 7.75% and 79.07 ± 9.43%, and after indirect cannulation, 86.43 ± 4.52% and 64.82 ± 13.61%, respectively.
Univariable analysis of operative outcome.
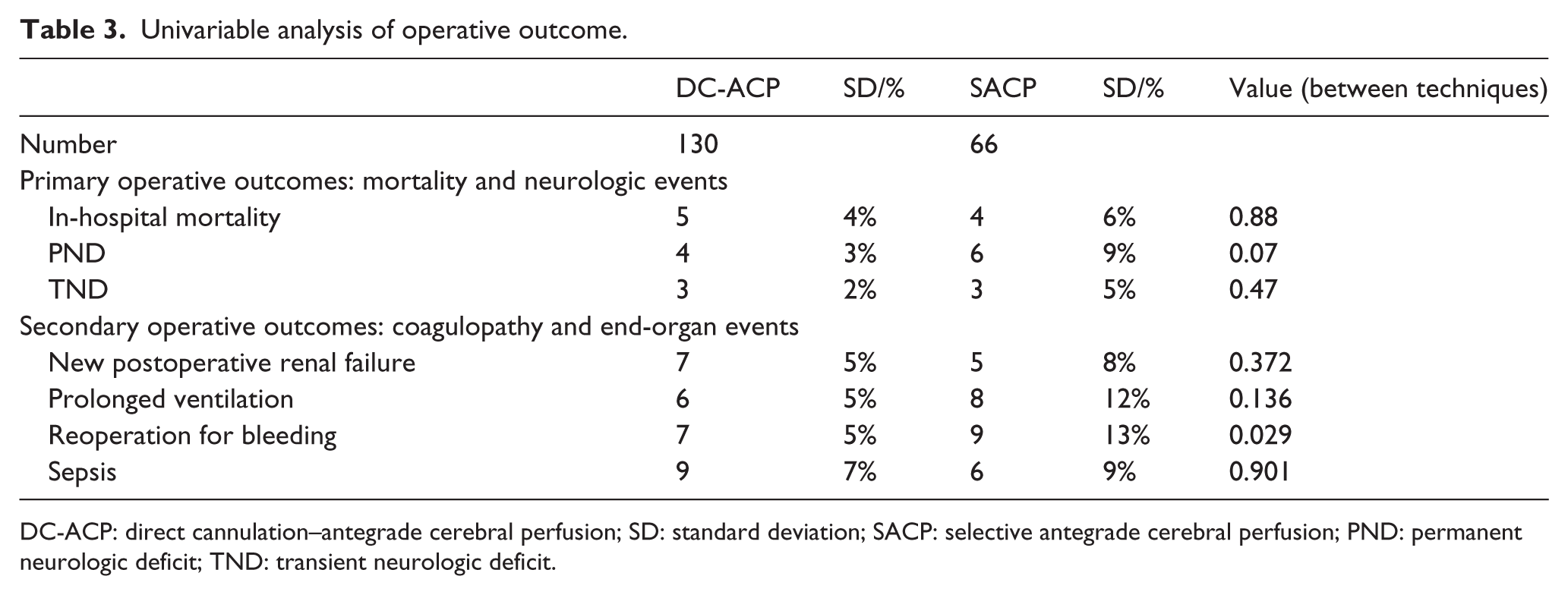
DC-ACP: direct cannulation–antegrade cerebral perfusion; SD: standard deviation; SACP: selective antegrade cerebral perfusion; PND: permanent neurologic deficit; TND: transient neurologic deficit.
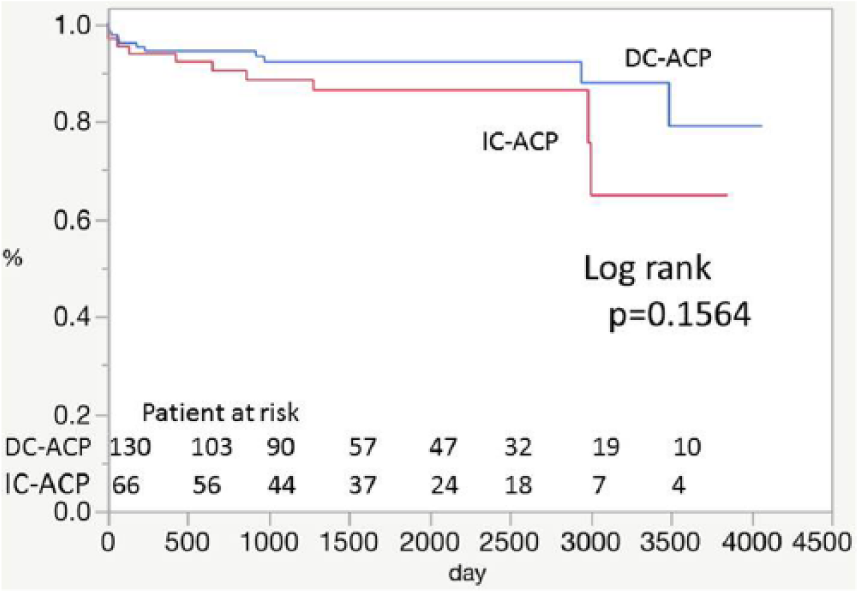
Kaplan–Meier curves were generated to compare the survival in the DC-ACP and IC-ACP groups. Follow-up was complete by 97% of patients. The mean follow-up time was 4.74 years (1.95–8.13 years) in the DC-ACP group and 5.50 years (2.03–8.56 years) in the IC-ACP group. Midterm survival was not influenced by the type of cannulation site (log-rank p = 0.1564).
Discussion
Cerebral protection strategies have evolved over the years and resulted in lower rates of neurologic complications during aortic arch surgery. Initially, deep hypothermic circulatory arrest (DHCA) was the primary method of neuro-protection, followed by retrograde cerebral perfusion to improve neuro-protection during DHCA. 4 Deep hypothermia causes many negative side effects, such as coagulopathy and organ dysfunction, and attempts have been made to avoid it. 5 To extend the safe period of cerebral protection, to avoid deep hypothermia, and to shorten the time of CPB, several techniques of ACP have been proposed. 6 The use of ACP makes deep hypothermia non-essential for neuro-protection, resulting in a growing tendency to increase the body temperature during circulatory arrest with selective brain perfusion. However, the clinical efficacy of mild-to-moderate hypothermia in ischaemic organ protection during circulatory arrest has not been thoroughly studied.
Urbanski et al. 7 evaluated the safety and efficiency of mild-to-moderate hypothermia for lower body protection during aortic arch surgery with circulatory arrest and ACP. Furthermore, studies comparing ACP and retrograde cerebral perfusion showed that ACP provides better cerebral protection during complex aortic procedures, which require additional time. 8 Okita et al. 9 demonstrated that ACP was associated with shorter intensive care unit stay and time on ventilation, while Halkos et al. 10 documented fewer renal and pulmonary complications. Thus, ACP is now considered a good method for the protection of cerebral circulation and has become the primary approach in the field of aortic surgery.
Most scholars agree that unilateral SCP can provide good cerebral protection if the integrity of the circle of Willis is preserved. 11 However, unilateral perfusion presents a potential risk of hypoperfusion in the left hemisphere, especially during mild-to-moderate hypothermia, creating controversy with regard to the safety of this procedure. The prevalence of incomplete circle of Willis has been reported to be 20%–30% in the normal population, 12 with elderly patients with hypoplastic or stenotic vertebral arteries being most commonly affected. Furthermore, the LSA often supplies collateral vessels to the spinal cord. To prevent spinal paraplegia, perfusion of the LSA is important. 13
Given that this study investigated SCP under mild hypothermia and that judging the integrity of the circle of Willis perioperatively is difficult, bilateral perfusion was employed to ensure the safety of arch replacement, which is especially important in total arch replacement. Some surgeons use cannulae inserted from inside of the arch during ACP.14,15 In the case of total arch replacement, these cannulae are typically obscured, which may interfere with the procedure and make it difficult to identify the orifice of the descending aorta. Furthermore, debris in the arch or descending aorta that are transferred into the cerebral blood vessels during whole body cooling with cannulation from inside the aorta or peripheral arteries (femoral or axillary) can lead to embolism because atheromatous and ulcerative lesions mainly exist at the orifice of the arch.14,16 In contrast, arterial inflow via the arch artery results in high-velocity retrograde blood flow into the aortic arch from the opening of the arch orifice, which may protect against embolic phenomena from the proximal aorta at the time of aortic manipulations or cross-clamping.
Accordingly, we performed direct cannulation of the right BCA, LCCA and LSA, referred to as ‘direct cannulation of the arch vessels’, with bilateral ACP. Our technique minimises manipulation of the arch arteries and dispenses with the need of sewing a branch graft to the arch arteries, thus reducing the risk of injury to the artery or adjacent anatomic structures such as the sympathetic chain and phrenic nerve. After initiation of circulatory arrest, individual clamps are applied to the BCA, LCCA and LSA. This minimises the likelihood of embolic events from the right and left carotid systems, as well as the posterior circulation via the LSA. Additionally, arch arteries, except for the LSA, are easily accessible in the neck and, when appropriate, can even be approached intrathoracically without an additional incision. If the LSA was located deeply and could not be manipulated, we did not perform its direct cannulation and only inserted a balloon tip cannula. This allowed routine cannulation of the arch arteries.
There is a potential risk of cerebral ‘luxury perfusion’ during CPB. Our strategy to prevent this over-perfusion is to reduce the arterial pressure during CPB to about 30–50 mmHg because cerebral blood flow is determined mainly by the arterial pressure and not the CPB flow rate. 17 Bilateral ACP was performed with a mean pressure in the arterial line of about 100 mmHg, and the flow rate was adjusted while maintaining the LCCA, right innominate artery, and LSA pressure within 30–50 mmHg. As a result, we observed no symptoms of cerebral over-perfusion. Postoperative cranial CT of 30 disease-free patients revealed no signs of cerebral oedema.
Similar to Garg et al. 18 and Vallabhajosyula et al., 19 we cannulated the arteries of the arch using paediatric cannulae from 9 Fr (LCCA and LSA) to ~12 Fr (BCA). Cannulae were used as a means of decreasing the risk of trauma to the arch vessels by minimising dissection and manipulation of the artery. In the study by Garg et al., 18 BCA cannulation was performed solely for ACP. In contrast, in our triple-artery approach, we performed cannulation for whole body perfusion and cooling. Hence, our method does not rely on cannulation of the ascending aorta or proximal arch and other peripheral arteries for CPB. Furthermore, we performed procedures only via median sternotomy rather than femoral or axillary artery cannulation, which is beneficial for extracorporeal circulation by reducing operative time. Additionally, since this method does not require circulatory arrest of the brain, the tympanic temperature should be maintained between 28°C and 32°C to ensure moderate hypothermia. This suppresses haemorrhagic diathesis resulting from extreme coagulation abnormality, which is a complication of DHCA. Vallabhajosyula et al. 19 argued that it is important to understand the time threshold for which systemic moderate hypothermia with ACP via only direct innominate artery cannulation with a small-sized cannula can be safely used before extending its use for more complex aortic arch reconstructions such as total arch replacement. In this regard, adequate circulatory flow could be obtained with our technique, which involved three arch vessels.
Axillary artery cannulation has become popular for treating acute type A aortic dissection or shaggy ascending aorta because it can provide antegrade blood flow in the aortic arch. We used DC-ACP during total arch replacement in nine patients. The three arch vessels were not dissected in these nine cases, which allowed selecting the cannulation site. DC-ACP method is not suitable if the arch vessels are dissected, and we opted for axillary or femoral cannulation instead.
There were no significant differences in the incidences of neurologic deficit, new-onset renal failure, prolonged ventilation, and sepsis. However, the rate of permanent neurologic deficit was slightly higher in the SACP group (DC-ACP, 3%; SACP, 8%; p = 0.08), even though DC-ACP requires clamping of the vessels, which many would argue means more manipulation and trauma than when balloon-tipped catheters are used without clamping. There was no significant difference in the SCP time, which may explain the similar rates of transient neurological deficit. Clinical reports suggest an association between temporary neurologic dysfunction after aortic surgery and global cerebral ischaemia due to the interruption of native cerebral flow. A correlation exists between the incidence of temporary neurologic dysfunction and the duration of cerebral perfusion management. 16
In the DC-ACP group, permanent neurologic deficit occurred in four patients (3%). No decrease in regional cerebral oxygen saturation or iatrogenic dissection of the arch vessels was observed in either group. Direct innominate artery cannulation was previously related to stroke rates of 0%–4.2%,13,20 which is similar to our results. Milewski et al. 16 suggested that most permanent neurologic injuries after aortic surgery occur secondary to embolic events rather than global ischaemia. We thought that IC-ACP group had occurred more embolic event rather than DC-ACP group when balloon-tipped catheters insert the arch vessels. In addition, permanent neurologic deficit correlates with the adverse sequelae of atherosclerotic aortic pathology and is less dependent on the cerebral protection technique utilised. However, a trend towards a longer CPB and cross-clamping time in the IC-ACP procedure was noted, necessitating the development of methodologies able to reduce the operative time in IC-ACP.
In addition to significantly decreasing intraoperative time, the use of DC-ACP reduced the rate of reoperation for bleeding. This is likely related to the more severe consumptive and functional coagulopathy associated with relatively longer CPB times. Moreover, moderate hypothermia provides greater protection from platelet dysfunction and coagulopathy. Midterm survival was not influenced by the type of cannulation. Further studies should involve careful long-term follow-up, with early detection of graft failure and dissection as priorities.
Our study has several limitations. First, this was a retrospective, nonrandomised, single-centre study. Future randomised studies with detailed neurocognitive assessment will be important for validating the utility of the DC-ACP strategy. Second, there could have been considerable selection bias, with our preferences and severity of arch pathology having played important roles in the choice of cannulation method. Finally, our proposed perfusion strategy require manipulating arch vessels and may thus be unsuitable in emergent cases of acute aortic dissection or rupture in patients with unstable preoperative haemodynamics.
In conclusion, DC-ACP during mild systemic hypothermia produced excellent outcomes. Moreover, DC-ACP significantly reduced the operative time and rate of reoperation for bleeding. DC-ACP is therefore a CPB method suitable for use in total arch replacement without increasing patient morbidity and mortality.
Footnotes
Acknowledgements
The authors thank Dr Touitsu Hirayama for advice on experimental design and for developing this surgical procedure. Dr Uesugi, Dr Ideta, Dr Oshitomi, and cardiovascular team members have been greatly tolerant and supportive of this procedure and my study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
