Abstract
Background:
Pulmonary arteriovenous malformations have been associated with clinical situations that put life at risk or cause physical disability. When the clinical picture is evident, percutaneous occlusion is recommended.
Methods:
We describe the use of an Amplatzer vascular plug in a female patient for the treatment of an idiopathic pulmonary arteriovenous malformation associated with syncope and transient ischemic attacks.
Results:
The patient was successfully treated via percutaneous intervention without complications. At 6 months follow-up, there was an absence of the pulmonary arteriovenous malformation confirmed via angiotomography and a full reintegration into her daily activities with no-clinical eventualities.
Conclusion:
The use of an Amplatzer vascular plug should be considered for the treatment of idiopathic pulmonary arteriovenous malformation. In experienced centers the procedure can be done safely with excellent clinical outcomes.
Introduction
Pulmonary arteriovenous malformations (AVMs) are branches from the pulmonary artery that connect directly to the pulmonary veins disregarding capillaries, resulting in right-to-left shunts. Most of the cases are associated with hereditary hemorrhagic telangiectasia (HHT) or Rendu–Osler–Weber syndrome. 1 There is another group of cases where no obvious cause is found, thus classifying them as idiopathic. We report a case of a patient affected by a large idiopathic AVM on the left lung.
Case presentation
We present a 44-year-old female patient who at the age of 18 years had two episodes of syncope classified as “disautonomy”; later, the patient had four ischemic attacks (2001, 2004, 2006, and 2008) which led to dysarthria as a result, no etiology was found.
The patient was referred to our hospital unit in June 2012 due to transient ischemic attack characterized by right-sided hemiparesis. A transthoracic and transesophagic echocardiogram were performed with the following findings: right atrium 35 × 38 mm, left atrium 45 × 34 mm, left ventricle end-diastolic and end-systolic diameter (46 and 30 mm, respectively), left ventricle ejection fraction of 70%, right ventricular telediastolic diameter of 30 mm, tricuspid annulus motion of 20 mm, pulmonary valve diameter of 19 mm, pulmonary artery trunk diameter of 18 mm, right pulmonary branch diameter of 12 mm, left pulmonary branch diameter of 11 mm, tricuspid valve structurally normal, pulmonary artery peak systolic pressure of 22 mmHg, and lipomatous hypertrophy of the interatrial septum with patent foramen ovale (PFO).
During her hospitalization, hypercoagulable disorders and other causes of stroke were ruled out, leaving the PFO as the only etiology possibility, so it was decided then to close the defect percutaneously.
For closure, left and right femoral venous access (10Fr and 8Fr, respectively) were obtained. Intracardiac echocardiography (ECI) was positioned at the junction of the inferior vena cava and the right atrium until an ideal image for evaluating the interatrial septum was displayed.
In Figure 1, saline solution microbubbles (NaCl 0.9%) were injected to the right atrium. Note that the bubbles do not cross through the foramen in spite of Valsalva maneuver. Three to five cardiac cycles later, the microbubbles fill the right atrium coming from the pulmonary vein. In Figure 2, we show Doppler imaging where the integrity of the interatrial septum is observed.
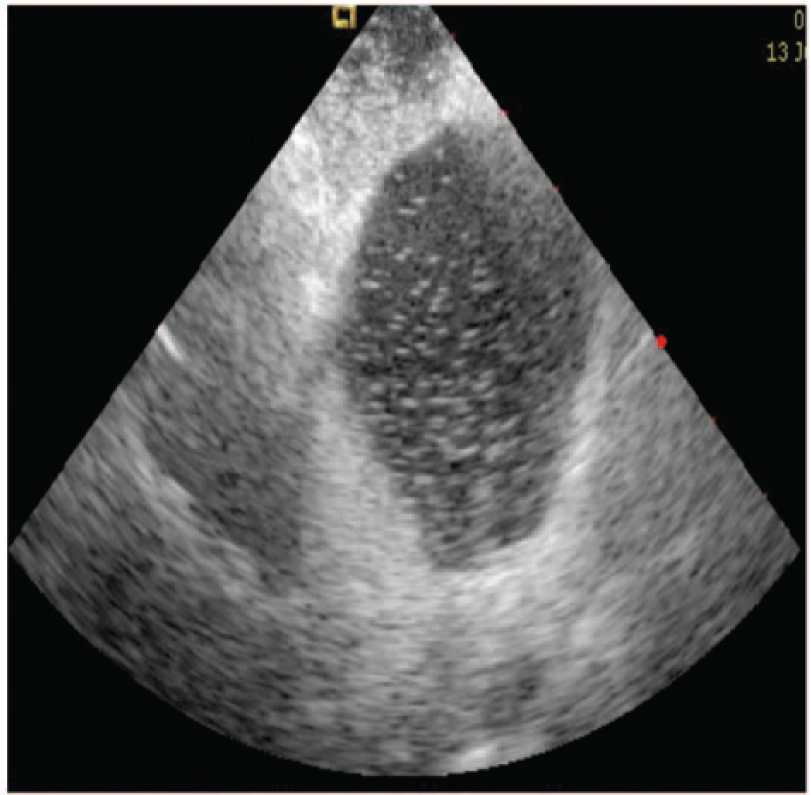
Microbubbles filling the left atrium from the pulmonary vein.
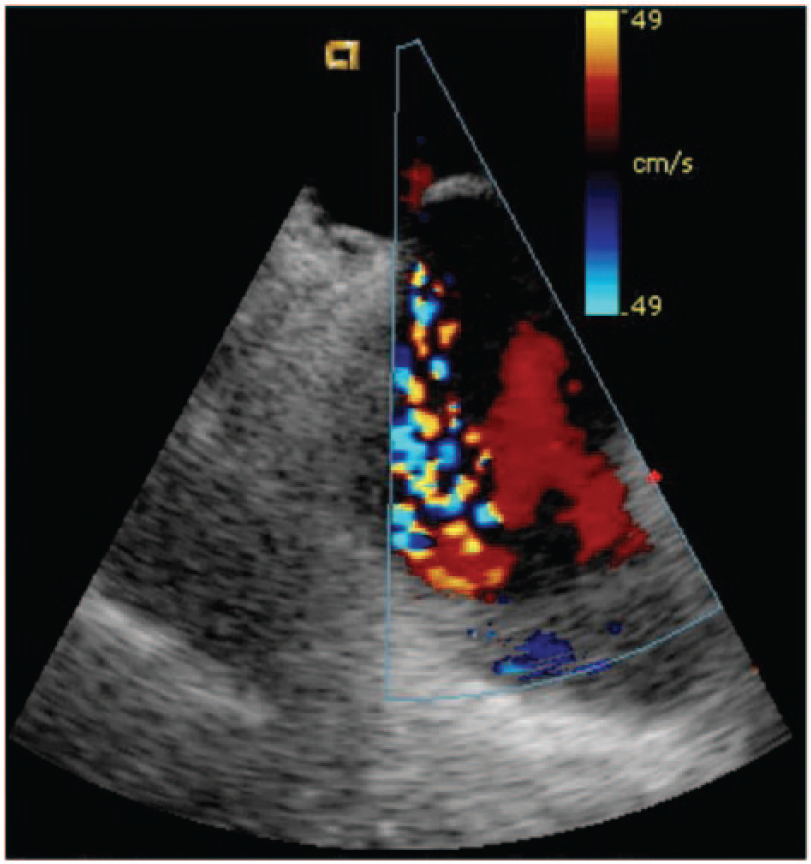
Intracardiac Doppler intracardiac showing interatrium septum integrity.
In the presence of the findings, selective and non-selective (Figure 3) pulmonary angiographies were performed. A large AVM was noted dependent to the left branch of the pulmonary artery that connects to the inferior pulmonary vein draining to the right atrium. No pulmonary hypertension was found. Aortic saturation was 92%.
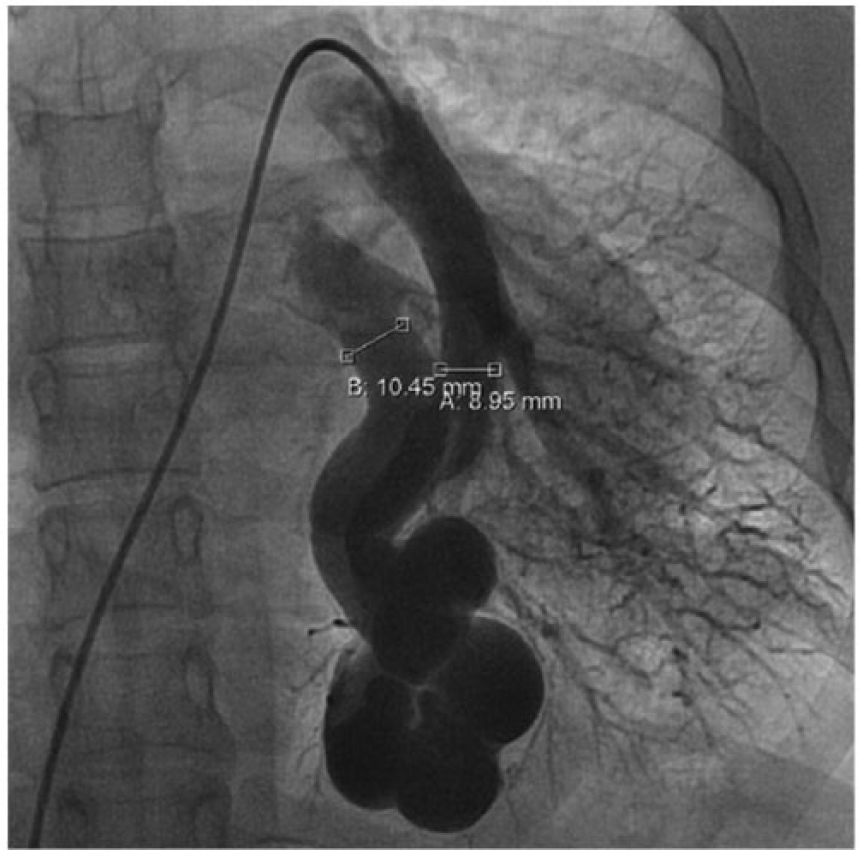
Selective angiography of the left branch of the pulmonary artery.
The diagnostic protocol was completed with a 64-slice computerized tomography (CT) scan with intravenous contrast and three-dimensional reconstruction where the angiographic findings were confirmed. In addition, the two left pulmonary veins are attached and reaching the right atrium as a single branch (Figure 4).
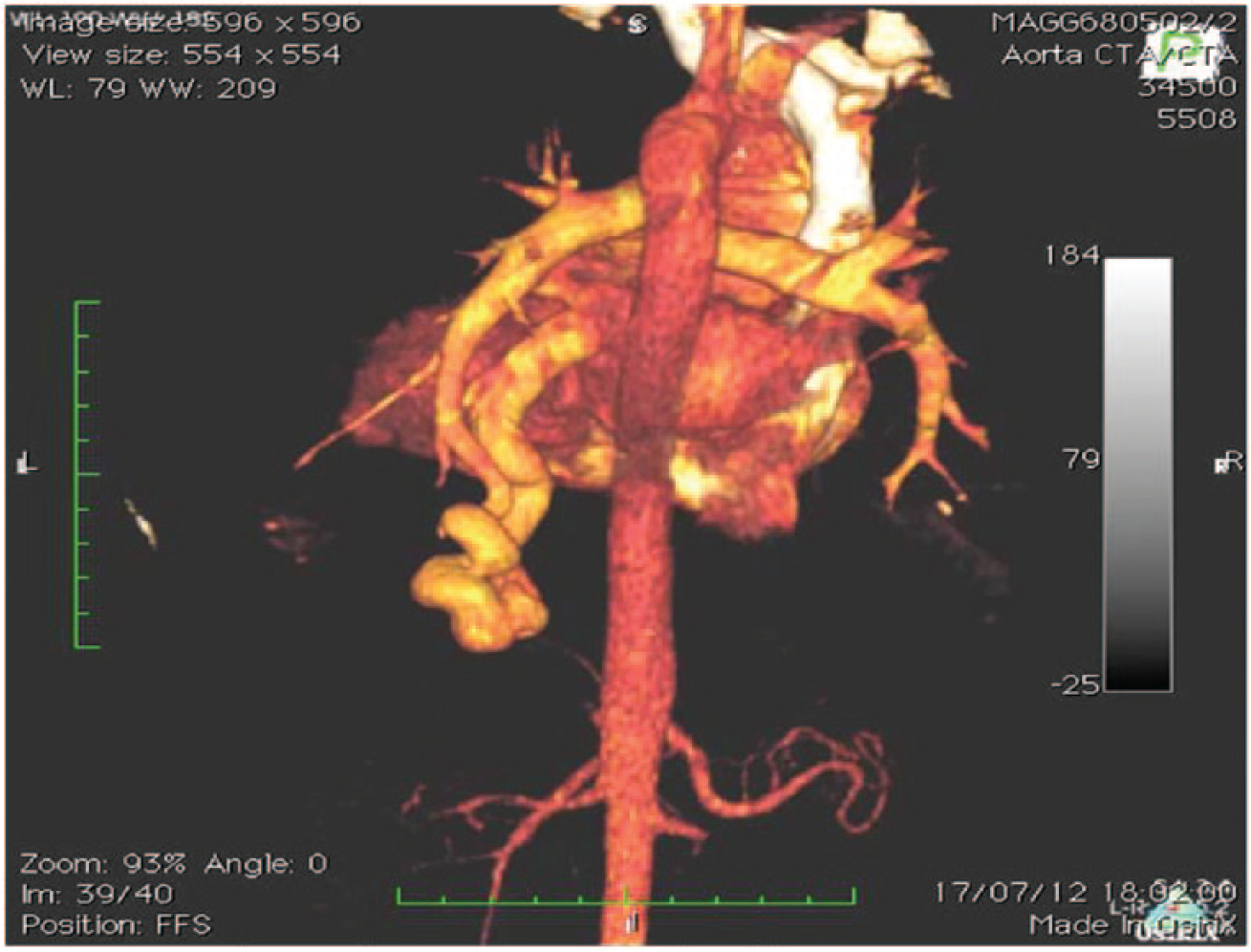
Cardiac angiotomography anterior view.
With all these data, we decided to use a vascular plug for occlusive purposes of the fistula percutaneously, the venous return was confirmed, the patient had no discomfort, was well and that the sack had three nutrient arteries but it was possible to encompass the three using just one device.
After a successful test using the Swan–Ganz catheter, an Amplatzer vascular plug type II of 14 mm (St. Jude Medical, MN, USA) was placed. This device is a nitinol-made autoexpandable net cylinder. Before releasing it, we confirmed its correct position and complete malformation absence, thus deciding its release and ending the procedure. No complications were observed (Figure 5).
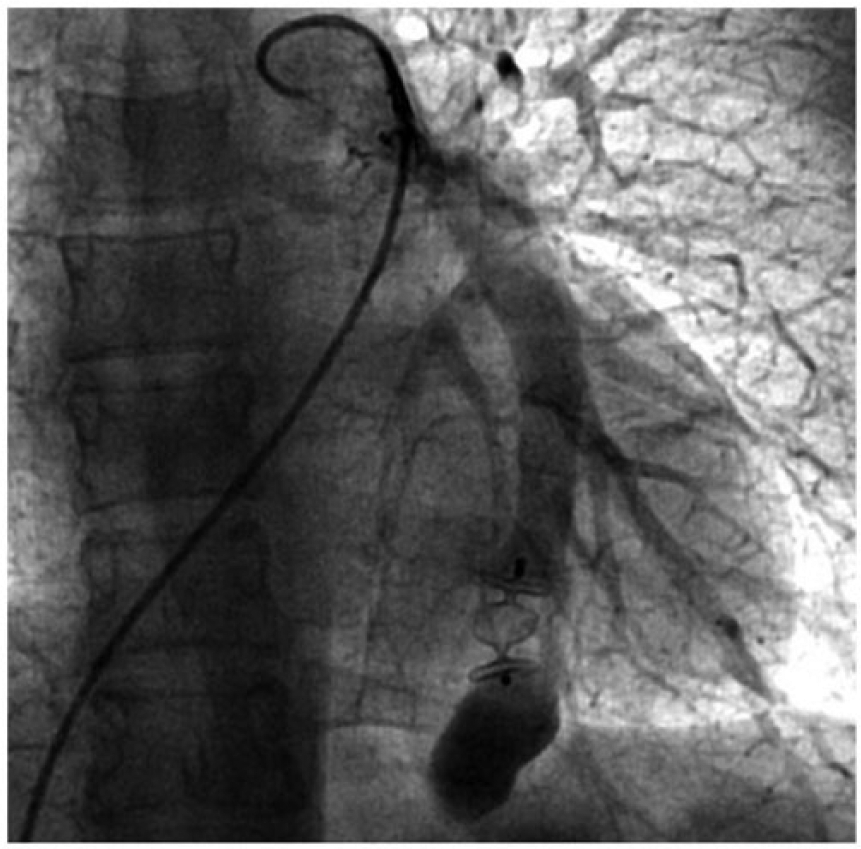
Angiographic control after device positioning. No contrast agent can be seen flowing through it.
The patient was discharged to her home 24 h later under coumarin therapy adjusted to an international normalized ratio (INR) value between 2.5 and 3.5.
After a 6-month follow-up, the patient had a functional class I according to New York Heart Association (NYHA), and no new strokes, no syncopes, and no diathesis were registered. The patient was fully integrated to her normal life. Control angiotomography did not reveal both the device and the fistula.
Discussion
AVMs are thinned-wall vascular spaces with one or more nutrient arteries, reaching several centimeters in diameter and with the probability to make right-to-left shunts. It is important to take into consideration that there are no capillaries associated with the malformation which favors paradoxical embolization. 2
Pulmonary veins are developed from the endodermic venous plexus, while pulmonary arteries are from the sixth aortic arch. During fetal period, minute arteriovenous fistulas are developed. Vascular septa among arterial and venous flow will form the vascular bed. According to some researchers, malformations originate from incomplete development of these septa, which prevents a connection between arteries and veins, all this during fetal development.3,4
HHT of Rendu–Osler–Weber syndrome account for 80 from 90% of the cases. It is an autosomal dominant disease, affecting two genes: 9q33-34 which synthesizes endoglin and 12q11-12 which synthesize activin-like kinase (AKL-1). Both act as transmembrane receptor for transforming growth factor (TGF-β), a protein that regulates cellular proliferation and differentiation, as well as migration and extracellular matrix formation. Other genes involved are 18q21.1 which synthesizes SMAD4 (mothers against decapentaplegic, drosophila, homolog) and causes juvenile gastrointestinal polyposis.1,5,6
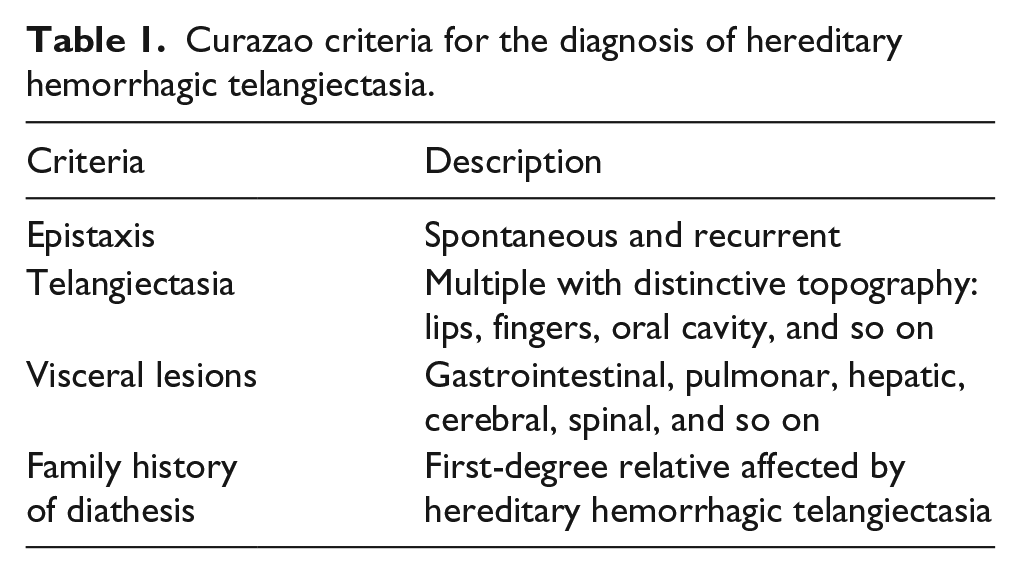
The genetic background of this disease is yet to be fully discovered, so that in 2000, for diagnostic and research purposes, a consensus was reached to standardize the diagnosis, called Curazao Criteria, where three or more criteria were met a definitive diagnosis is made; possible or suspicious case if two criteria were met and unlikely if only one criteria was met ( Table 1 ). 7
Curazao criteria for the diagnosis of hereditary hemorrhagic telangiectasia.
In 7% of the patients, pulmonary AVM arises as a sole finding, without fulfilling Curazao criteria for HHT, thus the case is classified as idiopathic. It is very important to interrogate the patients regarding history of recurrent epistaxis, since it is the most frequent sign and it is said that by the age of 45 years, 90% of the patients have had nose bleeding. 8
In our case, no genetic studies were done to rule out HHT. Nevertheless, it does not fulfill Curazao criteria with no positive family history, so we considered the case as an idiopathic pulmonary AVM, present in only 0.028% of the general population. 9
Pulmonary AVMs have been associated with life risk or cause physical disability such as stroke, transient ischemic attack, massive hemoptysis, spontaneous hemothorax, exercise dyspnea, and cyanosis. 10 As we mentioned earlier, the direct pass of all the systemic venous flow to the left auricle disregarding capillaries, favors paradoxical embolism. In our case, the patient had positive history for six strokes. Considering that single pulmonary malformations are similar to those found in HHT, it is possible to use the same criteria when treating them. The “Inter-national Guidelines for the Diagnosis and Management of Hereditary Haemorrhagic Telangiectasia” states that percutaneous embolization of the fistula should be considered when any of the aforementioned events are present if the nutrient artery is 3 mm or more in diameter.11,12 Both conditions were met in our patient, so malformation embolization was performed.
It is important to have in mind that, in this type of malformations the use of liquid embolizing agents and particle-based ones are contraindicated due to the lack of capillary bed. 3 Traditionally, coil-type devices have been used inside the nutrient artery. If a complex malformation is present, by this we mean that the sack receives flow for several nutrient arteries, each and one of them is embolized. The drawback of these devices are as follows: paradoxical embolization in up to 4% of cases, malformation revascularization in up to 22% of cases, the number of devices needed for the procedure (mean 4), and the time of fluoroscopy exposure.13–15 Since 1997, with the development of detachable coils, it was possible to embolize the aneurismatic sack en lieu of the nutrient artery which diminished malformation revascularization rate. However, it is necessary to consider some other complications such as sack ruptura, thrombi embolization during packing up, coil migration blocking venous drainage, and coil radial force of “0.018” which is smaller than that of the nutrient artery (“0.035”). 16
Another treatment option is a device called “plug,” a net of nitinol. There are four different types of nitinol. The one we used was type II which has a broad range of size (3–22 mm), its embolization time is shorter, and it has more contact area to the vessel and excellent radial force, thus the risk of embolization is less. This device is easy to release and usually only one is needed to close the vascular malformation which reduces procedure time.17,18
Nowadays, there are several case series where this plug was used to embolize an arterious malformation. Success is achieved in approximately 97% of the cases. It is important to assess the angulation, tortuosity and feeder vessels size, as these parameters may hinder proper delivery of the occluder device. It is recommended that the device is 30%–50% bigger than the vessel involved. These publications do not refer major complications during or after the procedure. Among the minor complications presented were pleuritic chest pain in 14% of patients which resolved with oral analgesics. No device migration was reported. In all, 4% of these malformations may relapse; so in order to decrease this phenomenon, it is necessary to place the plug no more than 1 cm apart from the sack neck.19,20
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
