Abstract
A 70-year-old man underwent mitral valve replacement using a porcine bioprosthesis for infective endocarditis. Transthoracic echocardiography revealed thickened leaflets with increased transprosthetic pressure gradient and new regurgitation, 6 months after surgery. He was followed up medically with the diagnosis of early valve degeneration. He ultimately presented with worsening orthopnea 13 months after surgery. Transesophageal echocardiography revealed an immobile round mass attached to the ventricular aspect of the leaflets, which hampered valve opening, suggesting bioprosthetic valve thrombosis. Anticoagulation with heparin and warfarin were effective and transthoracic echocardiography showed normal transprosthetic pressure gradient and disappearance of regurgitation 2 months later.
Introduction
Bioprosthetic valve thrombosis (BPVT) is generally uncommon compared with mechanical valve thrombosis.1,2 The clinical and echocardiographic diagnosis remains challenging, and the optimal management also remains unclear due to a lack of any randomized trial comparing surgery and medical therapy. 3 Herein, we report a rare case of early BPVT of a third-generation stented xenograft, Epic porcine valve (St Jude Medical, St Paul, MN, USA), in the mitral position developing 6 months after replacement that was successfully treated with anticoagulants.
Case report
A 70-year-old man with infective endocarditis underwent radical debridement of vegetation and complete resection of infected or destroyed leaflets, followed by mitral valve replacement using a St Jude Medical Epic porcine bioprosthesis. Routine transthoracic echocardiography (TTE) 2 weeks after surgery showed normal leaflet motion of the bioprosthesis with a mean transprosthetic pressure gradient (mTPPG) of 4 mmHg and no regurgitation (Figure 1(a)). Anticoagulation with warfarin was continued for 3 months postoperatively. However, regular TTE 6 months after surgery revealed irregular and inflexible leaflets with an increased mTPPG of 9 mmHg and new regurgitation (Figure 1(b) and (c)). The patient was followed medically, not surgically, with the possible diagnosis of early valve deterioration because he had exhibited neither heart failure nor recurrent infection. Ultimately, he developed heart failure with worsening orthopnea 13 months postoperatively. TTE revealed progressive thickening of the leaflets with an increased mTPPG of 15 mmHg (Figure 1(d)). Transesophageal echocardiography (TEE) revealed immobile non-pedunculated masses measuring 10 mm in maximum diameter that adhered to the ventricular aspect of the non-thickened and flexible leaflets (Figure 1(e)). The masses prevented the wide opening of the leaflets, resulting in severe mitral stenosis. These TEE findings led us to suspect BPVT. As the patient was hemodynamically stable and the thrombus was immobile and not large, anticoagulation was initiated using heparin and warfarin. Heparin was stopped when the international normalizing ratio exceeded 2.0, and warfarin has been continued. TTE showed a marked reduction in the leaflet thickness with a decreased mTPPG of 3 mmHg and disappearance of the regurgitation 2 months later, which suggested that most of the thrombus had resolved echographically (Figure 1(f)). The patient had no thromboembolic events and no further heart failure during the anticoagulation.
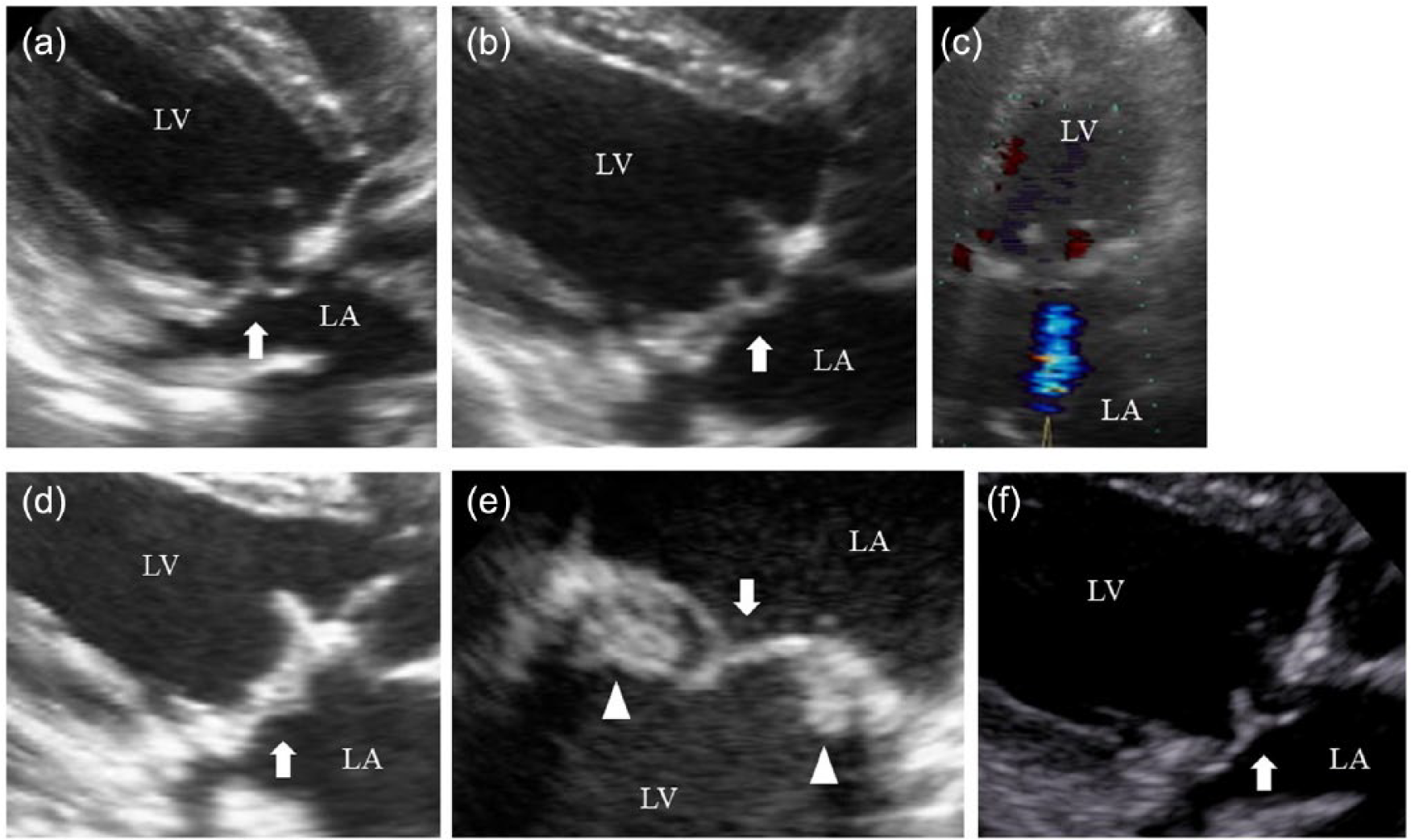
(a)–(d) and (f) Transthoracic and (e) transesophageal echocardiographic view of a mitral bioprosthesis (white arrow): (a) 2-week postoperative image showing normal configuration, (b) and (c) 6-month postoperative image showing slightly thickened leaflets and emerging regurgitation, (d) 13-month postoperative image showing progressive thickening of the leaflets, (e) transesophageal echocardiographic view 13 months after surgery showing ball-like masses attached to the ventricular side of the leaflets (white arrowheads) and normal thickness of the leaflets (white arrow), and (f) 2-month post-anticoagulation image showing disappearance of the leaflet thickness.
Discussion
An algorithm for a systematic approach to BPVT has recently been proposed. 4 This algorithm is characterized by providing adequate education to the cardiologists and surgeons regarding BPVT as the first step in the accurate diagnosis and treatment. Aggressive sharing of the experience and the knowledge of BPVT can promote early awareness of BPVT. 5 Pislaru et al. 4 emphasized that the detection of BPVT has been increasing steadily in association with the growing awareness of BPVT. In our case, we had no experience of BPVT before we encountered this case. Therefore, the diagnosis was delayed by half a year after the first detection of a bioprosthetic abnormality.
In the algorithm, increase in TPPG to greater than 50% when compared to the baseline and thickened leaflets with newly developed regurgitation are key findings of TTE for suspected prosthetic valve dysfunction including BPVT. TEE provides detailed visualization of the leaflets and abnormal structures on the prosthesis. The images of prosthesis by TEE sometimes appear extremely different from those obtained from TTE. In our case, the TTE showed diffuse thickness of the leaflets without any ball-like structure on the leaflet, which made us first assume very early valve degeneration, not valve thrombosis. In contrast, the TEE revealed normal thickness of the leaflets with ball-like structures on the leaflet, which could easily lead to the diagnosis of BPVT. However, even the TEE indicates the possible diagnosis of BPVT. This diagnosis can be changed to the definite diagnosis when complete resolution of the thrombus is achieved by the medical treatments. 6
Regarding the treatment strategies for prosthetic valve thrombosis, the 2014 American Heart Association/American College of Cardiology (AHA/ACC) valvular heart disease guidelines recommend medical therapy for patients with recent onset (<14 days), a small thrombus, and New York Heart Association (NYHA) I to II. 7 Surgical intervention is preferred for the patients with NYHA class III or IV, or with a mobile or large thrombus. 4 In our case, we selected medical therapy with anticoagulant using heparin and warfarin because the masses were immobile and borderline in size (10 mm), and the hemodynamic condition was quite stable after treatment for heart failure. Although the anticoagulation was started half a year after detection by TTE, the thrombus disappeared and TPPG decreased to normal. This finding suggests that anticoagulation performed within several months after the onset may not necessarily lead to failure of thrombus resolution, but can result in good outcome without explantation of the bioprosthesis.5,8
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
