Abstract
A 56-year-old man was readmitted to our hospital because of cardiac silhouette enlargement. One month before admission, he suffered cardiac tamponade due to an oozing rupture of the left ventricle 4 days after an acute posterior myocardial infarction and was salvaged by pericardial drainage. Upon readmission, computed tomography revealed a giant pseudoaneurysm (105 × 85 × 75 mm3) in the posterior wall of the left ventricle. The patient underwent aneurysmal repair successfully.
Introduction
Left ventricular (LV) pseudoaneurysm is a rare but fatal complication of acute myocardial infarction (MI), which occurs in less than 0.1% of all MI patients. 1 The mean maximum diameter of pseudoaneurysms has been reported to be 4.7–6.0 cm,2,3 and giant pseudoaneurysms exceeding 10 cm in size are very rare. We here report a rare case of a giant LV aneurysm, 10.5 cm in maximum diameter, developing after MI.
Case
A 56-year-old man was admitted to another hospital with a 2-day history of chest pain. He suffered shock due to cardiac tamponade on Day 5 of hospitalization and was transferred to our hospital after salvage by pericardial drainage. Electrocardiogram showed negative T waves in I, II, III, aVF, and V1–V4. Echocardiography showed wall thinning and reduced wall motion of the posterior wall. Coronary computed tomographic angiography revealed an occlusion of the obtuse marginal branch of the left circumflex artery. Accordingly, the patient was diagnosed with cardiac rupture caused by acute posterior MI. Coronary intervention for the occluded obtuse marginal branch was not performed because there was no residual myocardial viability, and the patient was treated medically. His condition improved, and he was discharged from the hospital in good health on Day 19 of hospitalization. Echocardiography before discharge showed wall thinning and small aneurysmal formation at the posterior wall (Figure 1(a)).
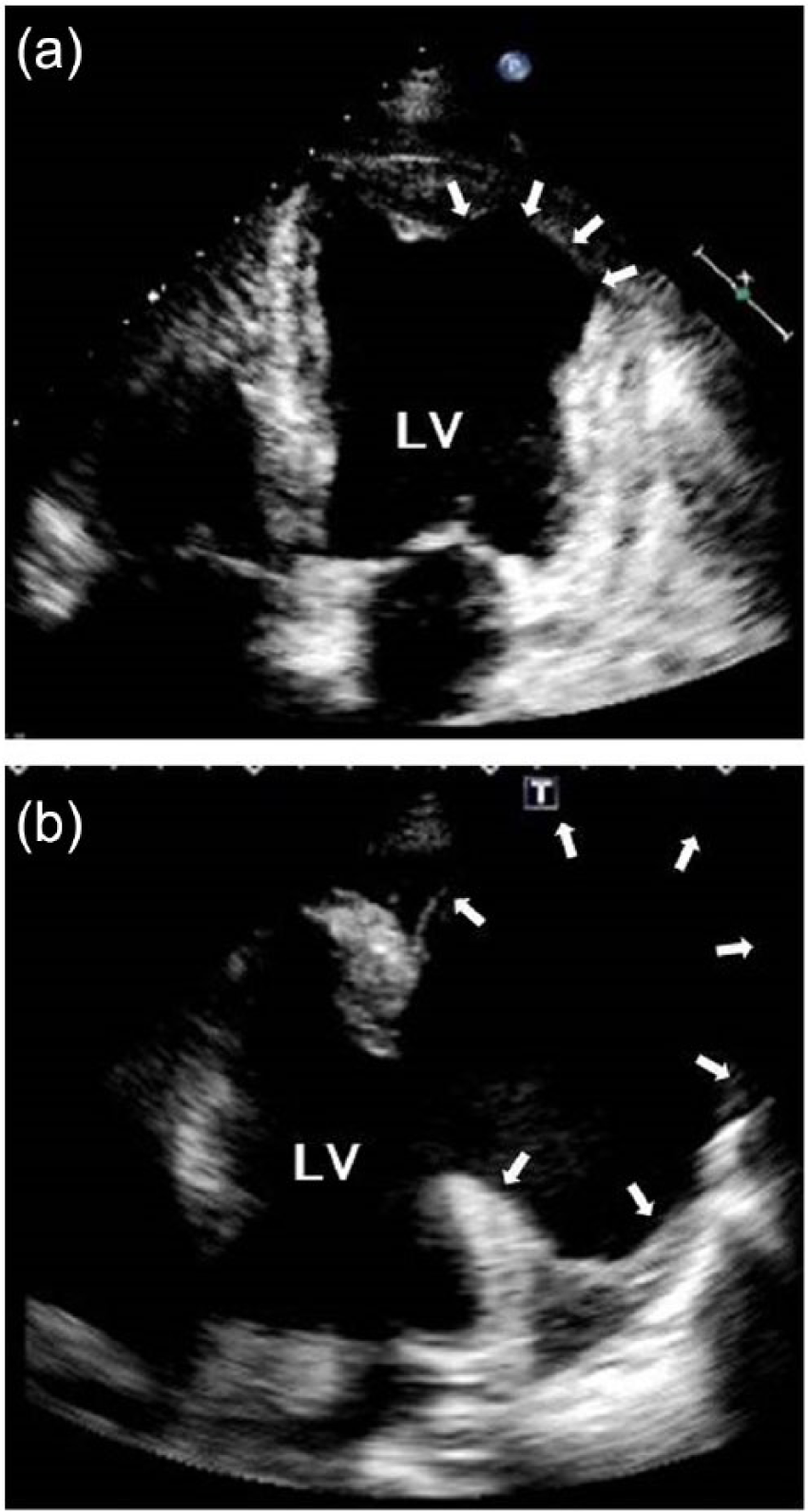
(a) Pre-discharge echocardiography of the first hospitalization revealed wall thinning and dyskinetic wall motion at the posterior wall of the LV (arrows) and (b) echocardiography at the second admission shows a giant aneurysmal formation at the posterior wall (arrows).
The patient was followed-up in our outpatient department 1 month after discharge. Although he had no clinical symptoms, repeated chest radiography revealed enlarged cardiac silhouette. Hence, echocardiography was performed promptly that revealed a large aneurysm at the posterior wall of the LV (Figure 1(b)). Computed tomography demonstrated a giant pseudoaneurysm (105 mm × 85 mm × 75 mm) in the posterior wall of the LV, with a large orifice (40 mm × 36 mm; Figure 2). Consequently, he was readmitted, and urgent circular endoaneurysmorrhaphy/simple patch repair was performed.
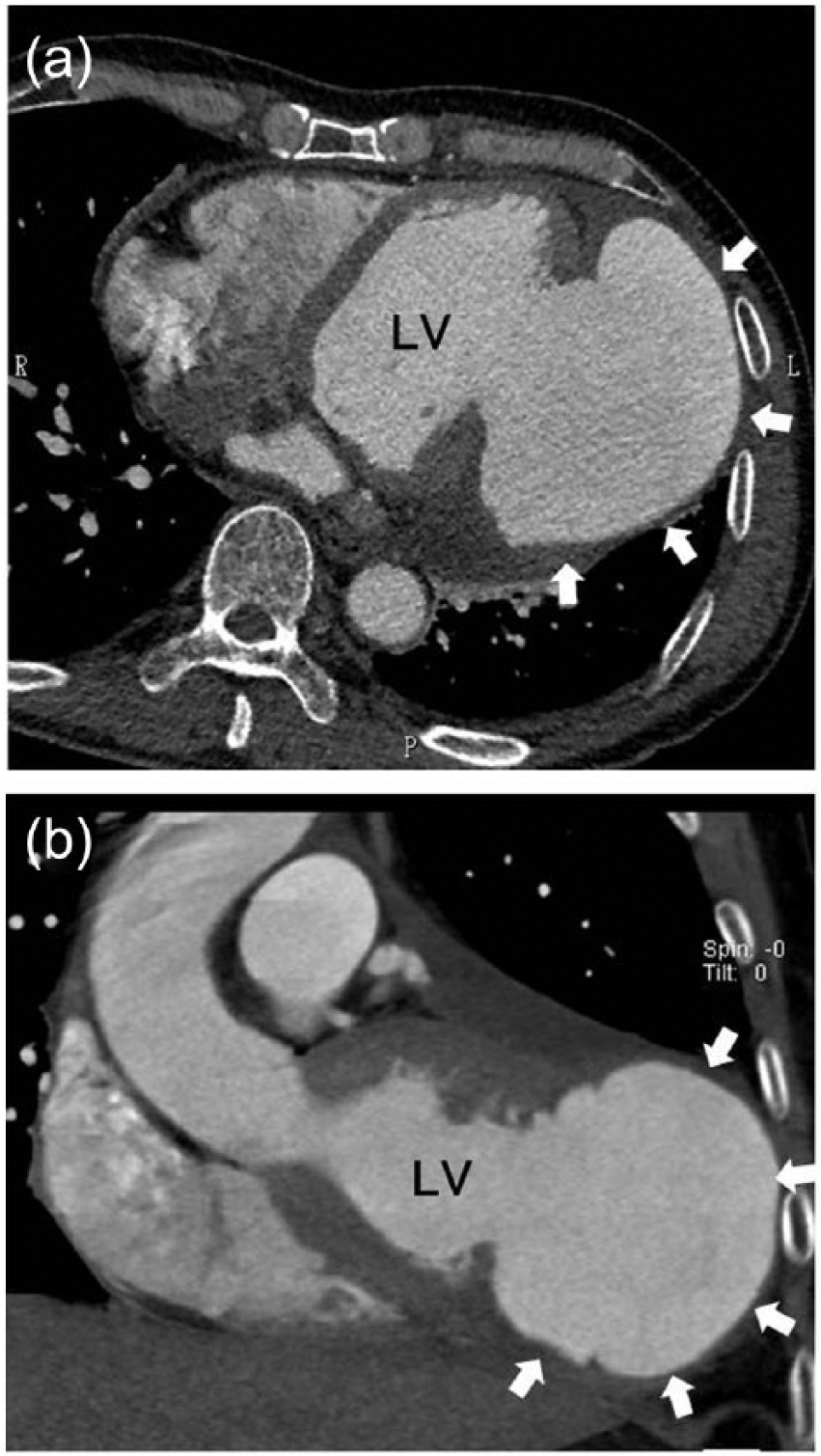
(a) Computed tomography demonstrating a left ventricular pseudoaneurysm lager than the LV volume (arrows). (b) Computed tomography at the second admission.
Surgery was performed through a median sternotomy. The patient was operated under mild hypothermic cardiac arrest and standard cardiopulmonary bypass. The posterior wall of the LV was densely adhered to the pericardium. Cold blood cardioplegic solution was administered in both antegrade and retrograde manners. Under cardioplegic arrest, the pseudoaneurysm was opened. The rim of the aneurysm wall was clear and measured 4 cm in diameter. The defect was closed using a polytetrafluoroethylene (PTFE) patch and interrupted pledgeted mattress sutures (2-0 polyether suture; Figure 3). The patient was weaned from cardiopulmonary bypass without difficulty and had an uneventful postoperative course. Left ventricular ejection fraction (LVEF) evaluated using echocardiography (the biplane modified Simpson’s method) was not significantly different before and after surgery (55% vs 56%, respectively). Postoperative echocardiography revealed that the orifice closed with a PTFE patch led to an akinetic segment. Global LV function was maintained. Figure 4 shows postoperative computed tomography.
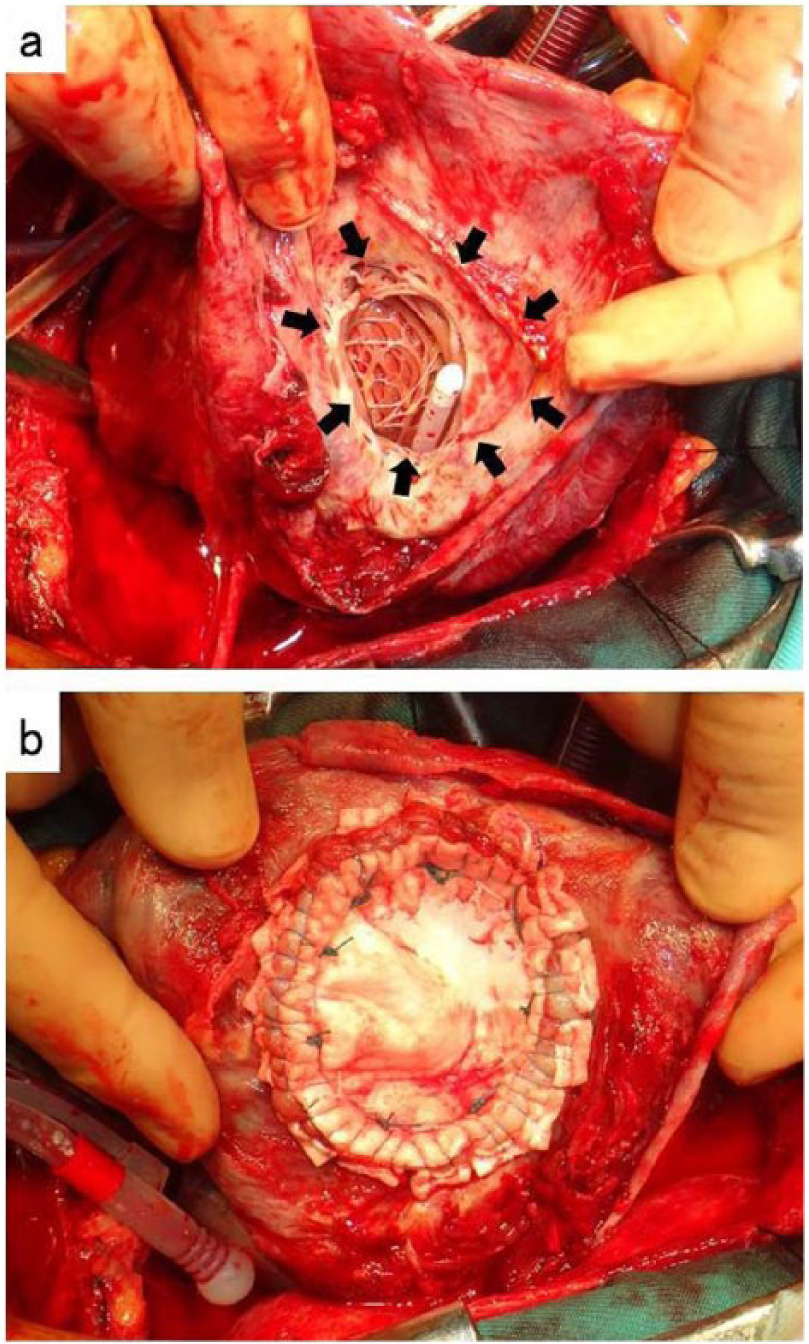
Operative views: (a) a large orifice was identified in the left ventricle cavity (arrows) and (b) the orifice was closed with a polytetrafluoroethylene patch.
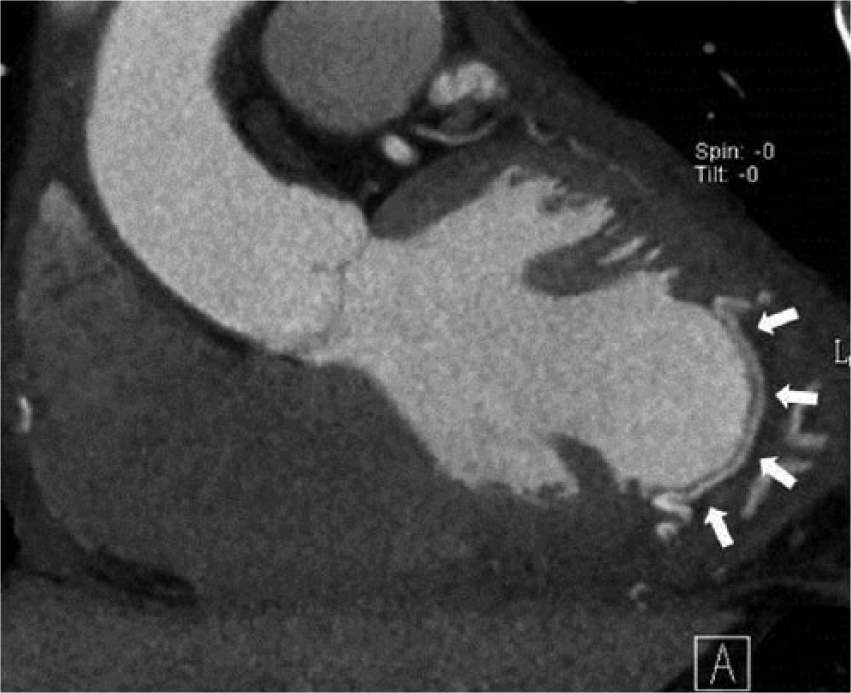
Postoperative computed tomography demonstrating the polytetrafluoroethylene patch (arrows).
The pathological evaluation revealed that the whole layer of the aneurysmal wall was replaced by fibrous tissue, and the lesion was diagnosed as a pseudoaneurysm. The patient was discharged on postoperative Day 18. At the latest follow-up (1 year after discharge), the patient was doing well.
Discussion
LV pseudoaneurysms are characterized by a contained cardiac rupture within the surrounding tissue. Although they are rare complications of acute MI and cardiac surgery, pseudoaneurysms are potentially fatal, because they are associated with high risks of rupture, thromboembolism, and ventricular arrhythmia. 1 The mortality risk of surgically untreated pseudoaneurysms is approximately 48%, 4 indicating that immediate surgical treatment is essential. However, the definitive diagnosis is sometimes difficult because routine examinations, including the symptoms, signs, electrocardiogram, and radiographic findings, are not always specific. 3 Eren et al. reported that LV pseudoaneurysms can be categorized as acute when discovered within 2 weeks of MI and as chronic when discovered more than 2 weeks after the event. 2 The definitive diagnosis is sometimes delayed, and chronic lesions are commonly discovered during investigations of symptoms such as cardiac failure, angina pectoris, and arrhythmia.2–7 Even though our case presented huge pseudoaneurysmal formation, the diagnosis was delayed (diagnosed 2 months after the onset of acute MI). To prevent catastrophic events, earlier diagnosis based on thorough echographical follow-up examinations is necessary, especially for patients with a history of hemopericardium after acute MI.
Regarding the aneurysmal size, previous reports have indicated that the mean maximum diameter of pseudoaneurysms ranges between 4.7 and 6.0 cm.2,3 On the other hand, reports of giant LV pseudoaneurysms are rare. In this case, the pseudoaneurysm exceeded 10 cm in maximum diameter. According to Laplace’s law, the larger the aneurysm, the higher the risk of rupture. Furthermore, pseudoaneurysms are more prone to rupture than true aneurysms, because they show defects in the myocardial continuity. Accordingly, prompt diagnosis and surgical intervention are recommended for this life-threatening complication.
Congenital LV diverticulum is important for differential diagnosis. 8 Ventricular diverticulum cases are classified into two types: muscular and fibrous. Muscular-type ventricular diverticulum involves all layers of the ventricular wall and can contract synchronously with the ventricle; therefore, this type can be identified relatively easily. On the other hand, fibrous-type ventricular diverticulum contains fibrous tissue that has partially replaced the myocardium and thus cannot contract. Fibrous-type ventricular diverticula are frequently found in the submitral location, and coronary angiography shows normal coronary anatomy. The patient described in this case had a recent history of MI, and formation of the aneurysm coincided with an obstructed coronary area away from the submitral lesion. Furthermore, pathological evaluation revealed that the whole layer of the aneurysmal wall was replaced by fibrous tissue. These results support that this aneurysm occurred as a late consequence of MI.
Although surgical intervention, including aneurysmal resection and patch repair, has traditionally been the standard treatment for LV pseudoaneurysms, recent advances in catheter intervention have enabled percutaneous LV pseudoaneurysm closure. Singh et al. 9 reported a case of percutaneous aneurysm closure using direct chest wall entry into the pseudoaneurysm, and Kumar et al. 10 treated LV pseudoaneurysms with an Amplatzer septal occluder device and/or percutaneous coil embolization. Although the indications should be strictly restricted based on the evaluations of the neck size and relative anatomy, particularly of the coronaries and valves, percutaneous trans-catheter closure may have potential benefits as an alternative treatment for high-risk patients.
In conclusion, we treated a giant LV pseudoaneurysm exceeding 10 cm in size following acute MI. To prevent sudden death, early diagnosis based on thorough and regular echographic follow-up is necessary.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
