Abstract
Background
Natalizumab is effective for relapsing MS but may trigger anti-natalizumab antibodies (ANZ) that neutralise drug activity and increase reactions. Trials report persistent antibodies in ∼6% and overall prevalence between 4% and 14%, but real-world burden and optimal testing remain uncertain. We assessed ANZ prevalence, timing, and correlates, comparing systematic with indication-driven testing.
Methods
Single-centre retrospective cohort of natalizumab-treated patients with ≥1 ANZ test. Testing followed routine care: systematic screening at months 6/12/18 or testing triggered by suspected inefficacy or infusion reactions. Clinical/MRI data were abstracted; time to positivity was used in Kaplan–Meier; predictors were assessed using multivariable logistic regression.
Results
Among 182 patients (348 tests), ANZ were detected in nine (4.9%; four persistent and five transient), all within the first 12 infusions. Positivity differed by indication (p = 0.005): suspected inefficacy 10.5% (4/38), systematic screening 1.6% (5/304), post-reaction testing 0% (0/6). Within suspected inefficacy, MRI activity was more strongly associated with positivity than symptoms alone (p = 0.04). In multivariable models, suspected inefficacy was the only independent predictor; adding indication improved discrimination (AUC 0.74 vs. 0.56).
Conclusions
In routine care, ANZ positivity is uncommon and occurs early. Indication-driven testing – especially when prompted by new MRI lesions – yields greater diagnostic value than routine screening of clinically stable patients, supporting a selective, context-driven approach to ANZ monitoring.
Keywords
Introduction
Natalizumab is a recombinant humanised monoclonal antibody targeting the α4 subunit of α4β1 integrin (VLA-4), a cell adhesion molecule on activated lymphocytes. By binding α4-integrin, it blocks interaction with VCAM-1 and MadCAM-1 on the endothelium, preventing immune cell transmigration across the blood–brain barrier into the CNS.1,2 This mechanism limits CNS inflammation in relapsing multiple sclerosis, leading to marked reductions in relapses and MRI activity, as shown in phase III trials (AFFIRM and SENTINEL)3,4 that demonstrated significant decreases in relapse rates, lesion burden, and disability progression versus placebo or interferon beta-1a.
Despite its proven efficacy, natalizumab is associated with a recognised risk of immunogenicity. The development of anti-natalizumab antibodies (ANZ), a subset of anti-drug antibodies (ADA), can lower circulating drug levels, neutralise its biological effects, and provoke infusion-related hypersensitivity reactions ranging from mild cutaneous symptoms to severe systemic manifestations.5,6 Anti-natalizumab antibodies may be transient or persistent, with only persistent antibodies consistently associated with loss of efficacy. When persistent, ANZ virtually abolish natalizumab efficacy. Evidence from the AFFIRM and SENTINEL trials showed that ∼6% of people with MS developed persistent antibodies and experienced higher relapse rates, faster disability progression, and greater MRI activity compared with antibody-negative individuals. 5 In addition, case reports describing ANZ-associated tumefactive demyelinating lesions (TDLs) and severe rebound activity7,8 further highlight the clinical relevance of these antibodies. Taken together, these findings underscore the need for timely ANZ detection, as their presence may compromise treatment efficacy, mandate a switch to alternative high-efficacy therapies, and increase the risk of disease reactivation.
While the incidence of ANZ formation has been well characterised in clinical trials, with reported prevalence between 4.5% and 14.1%,9–11 real-world data on their frequency, persistence, and clinical impact remain limited. The optimal ANZ monitoring strategy remains debated due to the low prevalence of persistent antibodies, the uncertain benefit of systematic screening, and the associated economic implications. Most centres restrict testing to subjects with suspected loss of efficacy or infusion-related reactions. However, following a dramatic case of severe disease activity after 10 infusions of natalizumab, despite no clinical or radiological activity at 6 months, 7 our centre adopted a systematic screening protocol. The diagnostic yield and clinical value of these different approaches are not yet well established. This study, therefore, aims to define the prevalence, timing, and predictors of ANZ positivity in a large real-world MS cohort treated with natalizumab, and to compare the diagnostic performance of systematic versus indication-driven testing strategies.
Methods
Study design and population
We retrospectively included all people with MS receiving natalizumab at Pitié-Salpêtrière Hospital, Paris, between January 2018 and January 2024, who underwent at least one ANZ test. Inclusion criteria were (i) MS diagnosis according to the 2017 McDonald criteria, 12 (ii) age ≥18 years, and (iii) current natalizumab treatment at time of testing. Subjects were excluded if data on test indication or date was missing. Out of 446 tests retrieved, 348 were included, corresponding to 182 individuals with MS. Tests performed solely to confirm prior positivity were excluded from prevalence calculations.
Testing strategy and data collection
ANZ testing was performed systematically at months 6, 12, and 18 of natalizumab treatment, and additionally if treatment failure or infusion reactions were suspected. Although this schedule was part of routine care, not all patients underwent testing at all three time points due to treatment discontinuation, missed visits, or unavailable testing. Anti-natalizumab antibodies were identified by means of a validated bridging enzyme immunoassay involving a competitive procedure for confirmation. Samples were processed in screening wells and in confirmatory wells after incubation with excess natalizumab to separate specific anti-drug antibodies from non-specific binding. Only antibodies whose interaction was blocked by co-incubation with excess natalizumab were considered to be ANZ. This procedure, although not including a functional cell-based neutralisation assay, selects for specific anti-natalizumab antibodies with competitive binding potential.9,10 Positive ANZ results were systematically repeated after one month in order to differentiate persistent from transient antibody responses. We extracted demographic and clinical data, including Expanded Disability Status Scale (EDSS), disease duration, MS phenotype, number of infusions, recent infections, and MRI activity. ANZ were classified as transient (single positive result not confirmed on repeat testing) or persistent (confirmed on repeat testing).
All people with MS included in this protocol are part of the OFSEP cohort 13 and provide written informed consent. This study is recorded in the general registry of data processing at AP-HP (Pitié-Salpêtrière Hospital) under the MR-004 framework (registration number 20250303093206).
Statistical analysis
All statistical analyses and data visualisations were conducted using R software (version 4.4.1). Descriptive statistics were used to characterise the study population, with mean age, disease duration (DD), and EDSS calculated and compared according to the ANZ test result (positive vs. negative). Continuous variables were compared using the Wilcoxon rank-sum test, while categorical variables (e.g. sex, MS phenotype, and test indication) were assessed using chi-squared tests.
Time-to-event analyses were conducted to assess the temporal dynamics of ANZ emergence. Kaplan-Meier survival curves were constructed using the number of natalizumab infusions as the time scale to reflect cumulative drug exposure, with extended or delayed intervals inherently accounted for, and Cox proportional hazards regression was applied to evaluate associations between baseline variables and time to first ANZ-positive result.
To identify independent predictors of ANZ positivity, we fitted multivariable logistic regression models including age, sex, disease duration, MS clinical phenotype, EDSS at testing, and test indication (clinical context). Because some people with MS contributed multiple tests, we accounted for within-patient correlation using cluster-robust standard errors at the patient level. Discrimination was evaluated with 10-fold cross-validated ROC analysis with patient-level grouping. We compared a full model (including test indication) with a reduced model (excluding it) to assess the added predictive value of clinical context.
Data availability
The dataset underlying this study contains sensitive clinical information collected at Pitié-Salpêtrière Hospital and is subject to institutional and GDPR restrictions. De-identified individual-level data and the analysis code are available from the corresponding author on reasonable request, contingent on approval by the local data-access committee.
Results
Between 2018 and 2024, 271 patients were identified in our clinical database as having received natalizumab, of whom 182 underwent at least one ANZ test. The mean age was 34 ± 9 years, and 78% of subjects were female. Most participants (97%) had relapsing–remitting MS (RRMS), while six had secondary progressive MS with relapses. The median EDSS was 1.5 (range 0–8), and the median disease duration was 8.1 years (range 0.3–33). In total, 348 tests were performed, with a mean of 1.96 tests per patient. Of these, 144 (41.3%) were obtained at or before the sixth infusion of natalizumab, 109 (31.3%) between the sixth and 12th infusions, 62 (18.3%) between the 12th and 18th infusions, and only 31 (9.1%) after the 18th infusion. Among the 182 subjects included, 114 underwent at least two tests, and 50 had ANZ assessed at all three timepoints (sixth, 12th, and 18th infusions).
Prevalence, timing, and clinical context of ANZ positivity
Testing was conducted systematically in 304 cases (87%), in suspected treatment failure in 38 cases (11%), and following infusion reactions in six cases (2%).
ANZ were detected in nine of 182 subjects (4.9%). Four (2.2%) had persistent positivity, and five (2.7%) were transiently positive. Notably, all positive tests were performed within the first 12 infusions. Kaplan-Meier analysis showed that all ANZ-positive cases occurred early in treatment, typically within the first 12 infusions and never thereafter in our cohort (Figure 1A and B). Cox regression identified no significant predictors of earlier positivity, and survival curves confirmed early clustering of immunogenicity events.

Temporal dynamics of anti-natalizumab antibody testing and seropositivity by number of infusions. (A) Stacked histogram of the number of natalizumab infusions at the time of ANZ testing (0–24 infusions shown; tests >24 not displayed, n = 23). Bars are stratified by result. Testing is concentrated early in treatment (≈3–12 infusions), and most ANZ-positive results also occur in this window; positives beyond ∼10 infusions are uncommon. (B) Kaplan–Meier curve of the probability of remaining ANZ-negative over infusions (S(t)). The curve shows small drops during the early infusions (≈4–8) and is largely flat thereafter. Shaded area = 95% CI. The table reports the number of people with MS at risk at each infusion mark, decreasing as fewer subjects reach later infusion counts.
Among patients tested for suspected inefficacy, ANZ-positive individuals had a numerically shorter disease duration (7.20 ± 7.6 vs. mean 9.00 ± 10.9) and were slightly older (35.40 ± 10.2 vs. 33.70 ± 9.10), although these differences did not reach statistical significance (p > 0.5). EDSS distribution was comparable across groups (p > 0.05).
Clinical, radiological, and predictive factors
ANZ positivity differed significantly by testing indication, with a positivity rate of 10.5% (4/38) among patients tested for suspected inefficacy, 1.6% (5/304) during systematic screening, and 0% (0/6) following infusion reactions (p = 0.005). Restricting to subjects tested for suspected inefficacy (one index episode per patient), ANZ positivity was higher when testing was prompted by new MRI lesions than by clinical symptoms alone (e.g. motor worsening, paraesthesia, visual acuity decline, and neuropathic pain) (25% [3/12] vs. 4% [1/23]; Fisher's exact p = 0.04; Figure 2).
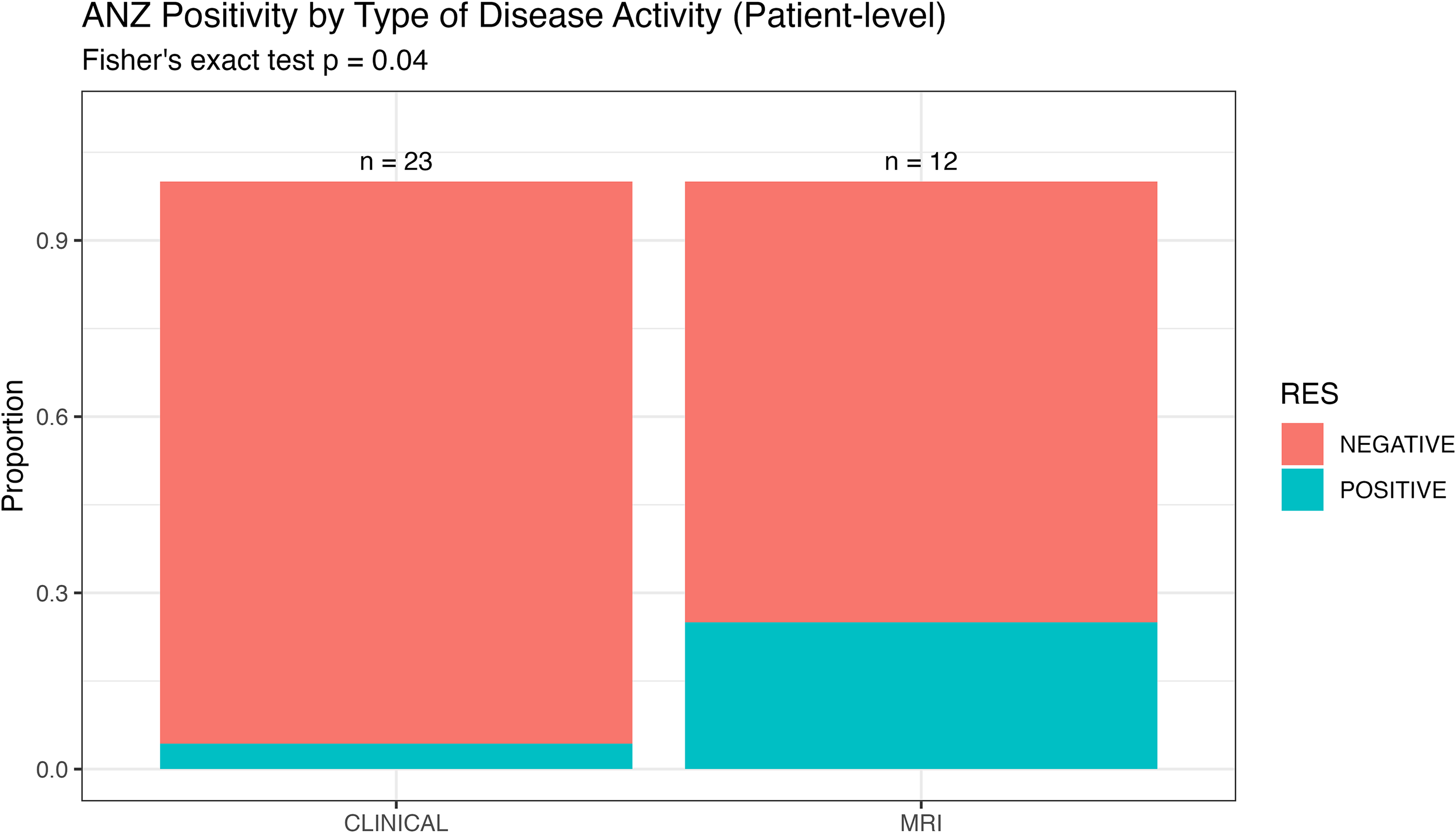
Anti-natalizumab antibody (ANZ) positivity by type of disease activity prompting testing (patient-level). Stacked bar plot showing the proportion of ANZ-positive and ANZ-negative results among subjects tested for suspected natalizumab inefficacy, using one index episode per patient. Sample sizes are shown above bars (clinical symptoms alone, n = 23; new MRI lesions, n = 12). ANZ positivity was higher when testing was prompted by new MRI lesions than by clinical symptoms alone (25% [3/12] vs. 4% [1/23]; Fisher's exact p = 0.04), suggesting that radiological activity may be more strongly associated with immunogenicity to natalizumab than clinical relapses.
Systematic screening and exploratory analyses
Among the 304 systematic tests, five were positive (1.6%), with three confirmed as persistently positive, unlike the 10.5% positivity rate in the case of suspected inefficacy. These three subjects had no clinical or radiological signs of disease activity but were switched to an alternative high-efficacy therapy (anti-CD20). Thus, the number of systematic tests needed to identify one persistently ANZ-positive patient was ∼100 (0.98% of tests leading to treatment change).
Multivariable logistic regression revealed that only suspected inefficacy was independently associated with ANZ positivity (adjusted OR 5.53, 95% CI 1.13–26.95; p = 0.034), whereas age, sex, MS type, disease duration, and EDSS were not significant. In patient-grouped cross-validation, inclusion of test reason improved model performance: AUC increased from 0.56 (reduced model) to 0.74, supporting clinical context in testing decisions. These predictive models were specified at the test level, with patient-level clustering to account for repeated measurements.
Discussion
In this large real-world cohort of natalizumab-treated people with MS, anti-natalizumab antibodies (ANZ) were rare, with a prevalence of 4.9%, a rate comparable to other real-world studies,9,14 and lower than reported in clinical trials. 5 Differences in reported ANZ prevalence across studies may partly reflect methodological heterogeneity, including differences in assay sensitivity and the ability to detect low-titer or neutralising antibodies. Crucially, ANZ were more frequently detected in cases with clinical or radiological signs of disease activity, in line with findings from Ciano-Petersen et al. 15 MRI activity emerged as a particularly strong correlate of ANZ positivity, supporting the role of imaging as a surveillance tool during natalizumab therapy.
Most ANZ-positive cases occurred within the first year of treatment, consistent with previous work indicating that immunisation against natalizumab typically emerges early.11,16 This timing aligns with immunological models in which early-phase inflammation may favour ADA generation.17,18 The clinical impact of systematic screening was modest: in our cohort, only 3/182 patients (1.6%) were identified through routine testing and switched to an alternative high-efficacy therapy – changes that may have prevented future relapses. However, the significance of persistent anti-natalizumab antibody positivity in the absence of clinical or radiological relapse remains unclear, and potential assay limitations, such as low sensitivity/specificity or low-titer, may complicate interpretation. Given the low yield and resource implications, our data argue against systematic ANZ screening. We suggest an indication-driven approach, reserving testing for clinical or MRI suspicion of therapeutic failure (or infusion reactions), with repeat assays only when warranted by the clinical course. Although infusion reactions have been linked to ANZ in clinical trials,5,19 we did not observe ANZ positivity in people with MS with infusion symptoms, probably due to the mild nature of reactions in our cohort. One notable case involved a negative test following an early allergic reaction, but subsequent positivity, possibly due to immune complex interference with the assay at the early phase – a phenomenon also suggested by Vennegoor et al. 6
Despite evaluating a wide range of variables, no clinical phenotype was clearly predisposed to ANZ formation. This aligns with prior evidence suggesting that host immunological factors may outweigh clinical markers in driving ADA formation. Although EDSS, disease duration, and age were evaluated as candidate predictors, they did not differ significantly between ANZ-positive and ANZ-negative individuals and provided only limited discriminatory information in regression analyses, insufficient to support risk-based screening models. MRI activity remains the most realistic predictor.
The retrospective design of this study, including only a limited number of ANZ-positive people with MS, limits the generalisability of findings. Further, potential selection bias related to test indication needs to be considered, as ANZ testing was partly driven by clinical or radiological suspicion and did not apply uniformly to all treated patients. Potential confounders also include variability in follow-up and adherence to the MRI protocol. Nevertheless, its large cohort and integration of clinical, radiological, and biological data enhance its relevance.
Taken together, these findings suggest that anti-natalizumab antibodies represent a rare and early immunogenic event in real-world clinical practice, with limited clinical yield from routine or indefinite screening. The significantly higher detection rate when screening is prompted by clinical or MRI signs of disease activity supports the usefulness of the ANZ test for confirmatory purposes in the context of suspected therapeutic failure, rather than as a surveillance strategy. Although antibody testing is often triggered by overt disease activity, the identification of ANZ at this stage remains clinically meaningful, as it provides a clear biological explanation for treatment inefficacy and directly informs timely therapeutic switching, with the potential to limit further inflammatory damage. On this basis, our data support an indication-driven approach to ANZ testing, guided by clinical and/or radiological criteria or infusion reactions, rather than systematic screening.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
