Abstract
Background
In relapsing–remitting multiple sclerosis (RRMS), smouldering inflammation at the rims of chronic active lesions is increasingly recognised as a key driver of disease progression. Paramagnetic rim lesions (PRLs), detected using susceptibility-weighted imaging, have emerged as a potential biomarker of this chronic inflammatory activity. However, their clinical relevance and relationship to lesion-specific features such as size and age remain poorly understood.
Objective
To investigate the association between PRL presence and clinical/radiological measures of disease progression, and to explore how PRLs relate to lesion size and age.
Methods
A retrospective study of 60 RRMS patients with over 5 years of magnetic resonance imaging data was conducted using susceptibility-weighted angiography. Lesions larger than 100 mm3 were analysed.
Results
PRLs were present in 48% of patients and represented 13% of lesions. PRLs were significantly larger and more structurally damaged, with volume correlating with EDSS change and brain atrophy. All lesions formed within 5 years of imaging were PRLs. This finding was validated in two independent international cohorts. Moreover, the proportion of rim-positive lesions decreased as lesion age increased.
Conclusion
PRLs are closely linked to lesion age and early development, supporting their role as a dynamic biomarker of lesion activity in multiple sclerosis.
Keywords
Introduction
Among the various pathological features of multiple sclerosis (MS), smouldering inflammation – particularly at the rims of chronic active lesions – has emerged as a crucial driver of disease progression. This process leads to slow but persistent axonal injury, contributing to progressive neurodegeneration, loss of brain tissue and disability progression.1–4
Despite its importance, smouldering inflammation remains an unmet therapeutic target for people living with MS, underscoring the need for effective biomarkers to monitor and understand this phenomenon.
Paramagnetic rim lesions (PRLs), visualised on magnetic resonance imaging (MRI) using susceptibility/T2*-weighted sequences, have been identified as one of the most pathologically validated markers for smouldering inflammation.5–8
While the relationship between PRL presence and clinical and radiological measures of disease progression has been widely reported in recent years,9–19 the strength of this association remains unclear. Additionally, the temporal dynamics of PRLs remain uncertain. Although PRLs have been observed to arise at the time of lesion formation, 6 the reported proportion of new lesions developing a paramagnetic rim varies considerably.2,9,20–24
The objective of this exploratory study is to investigate the relationship between PRL presence and lesion-specific characteristics, focusing on lesion size and age, while also examining the associations of PRLs with clinical and radiological measures of disease progression.
Materials and methods
The primary study was approved by the University of Sydney Human Research Ethics Committees and followed the tenets of the Declaration of Helsinki. Written informed consent was obtained from all participants.
Subjects and study design
Consecutive patients with established relapsing–remitting multiple sclerosis (RRMS; diagnosed according to the revised McDonald 2010 criteria 25 ) who were enrolled in the ‘Mechanism of Axonal Degeneration in MS’ study, had reached at least 60 months of follow-up, underwent annual MRI on the same 3T MRI system and had susceptibility-weighted angiography (SWAN) imaging at the last timepoint (referred to as SWAN timepoint) were included (Figure 1).
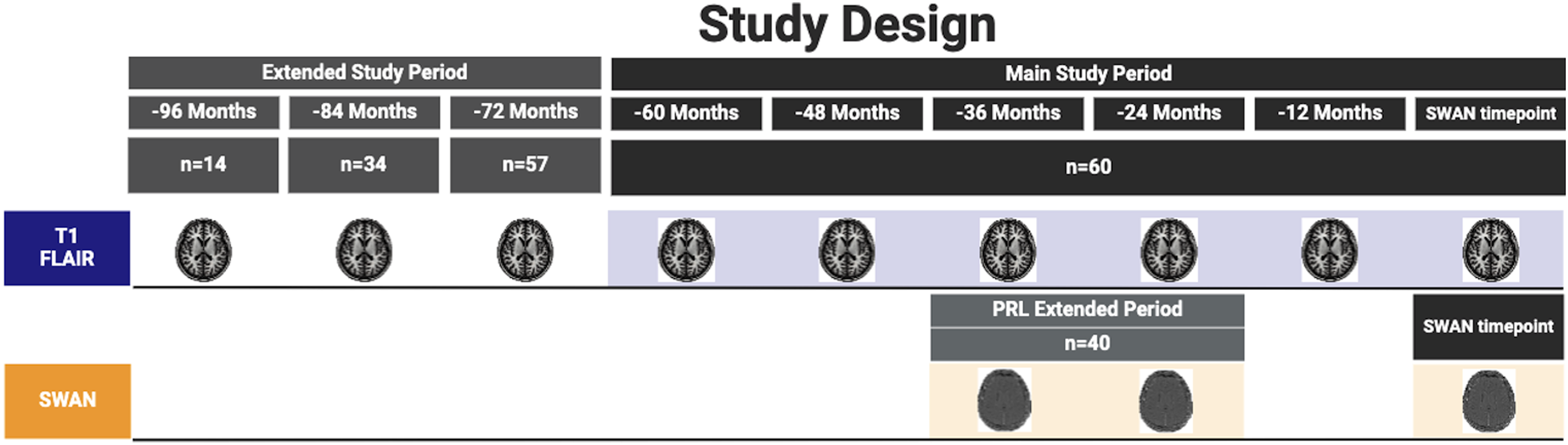
Schematic representation of the study design. This figure provides an overview of the study's structure, including key timepoints, imaging intervals and assessment phases used to track PRL changes over the extended study period. PRL: paramagnetic rim lesion.
The SWAN timepoint was used for all cross-sectional analyses; the data obtained within the 60 months prior (referred to as the main study period) was used for the longitudinal analysis.
The extended study period (up to 96 months prior to the SWAN timepoint), with a reduced number of patients, was used for a sub-study analysis.
MRI protocol for Sydney cohort
MRI was performed using a 3T GE Discovery MR750 scanner (GE Medical Systems, Milwaukee, WI). The following MRI sequences were acquired: sagittal 3D T1-weighted, 3D T2-FLAIR, diffusion-weighted imaging and SWAN protocol using a 3D gradient-echo sequence. Specific acquisition parameters and MRI image processing are presented in the Supplemental materials.
Lesion analysis
White matter lesions were segmented for each timepoint of each patient using an advanced artificial intelligence (AI)-based lesion segmentation algorithm (iQ-Solutions™ MS Report, Sydney Neuroimaging Analysis Centre, Sydney, Australia). For this analysis, only lesions exceeding a volume of 100 mm3 were included to maintain consistency and ensure the paramagnetic rim could be clearly visualised on phase imaging.
This threshold was chosen to minimise partial volume effects and dipolar artefacts, and to allow confident classification as rim-positive or definite non-rim. It is also consistent with North American Imaging in MS (NAIMS) guidance, which advises caution when assessing rims in lesions with a core diameter below ∼3 mm (∼65 mm3); a 100 mm3 threshold corresponds roughly to lesions with the core diameter ≥3.5 mm, where rim assessment is more reliable.
The degree of tissue damage within these chronic lesions was quantified using T1 hypointensity and mean diffusivity (MD), as described in previous studies 26 and specified in the Supplemental materials.
PRL analysis
For each patient, PRLs were visually identified on the phase images at SWAN timepoint scan according to criteria proposed in the 2022 NAIMS Consensus Statement on Imaging Chronic Active Lesions (CAL). 6 The senior author (AK) assessed the presence of rim lesions. The assessment was performed blinded to clinical information and other imaging results.
Volumetric brain analysis
Total brain, white matter, grey matter and ventricular volume27,28 were analysed using AssemblyNet, an AI brain segmentation tool, on T1 images co-registered to anterior commissure-posterior commissure space. 29 All volumes were scaled based on Montreal Neurological Institute template registration to account for head size variability and as such, the measures at the SWAN timepoint are reported as a percentage of total intracranial volume (% of TIV). For longitudinal analysis, the annualised percentage change during the main study period (i.e. 5 years) was calculated for each MRI measure to assess atrophy.
Disease-modifying therapy
Disease-modifying therapy categories for the main cohort were grouped into three categories: no treatment, moderate-efficacy treatment (e.g. interferon, glatiramer acetate, teriflunomide, fingolimod and dimethyl fumarate) and high-efficacy treatment (e.g. natalizumab, alemtuzumab and ocrelizumab).30,31
Validation datasets
To validate the relationship between the presence of a paramagnetic rim and lesion age, a consecutive cohort of RRMS patients from Charité – Universitaetsmedizin Berlin in Germany and RRMS patients and clinically isolated syndrome (CIS) from Vall d’Hebron University Hospital in Barcelona, Spain were included. Each patient underwent at least three consecutive MRI scans. Similar to the main study, PRLs were assessed at the final timepoint. The Berlin cohort was approved by the Ethics Committee of Charité – Universitätsmedizin Berlin (EA1/362/20, EA1/163/12), and the Spanish cohort was approved by the Clinical Research Ethics Committee at Vall d’Hebron University Hospital (PR(AG) 302/2018). All participants provided written informed consent for research and publication. Detailed information on the study protocols from the two cohorts has been published previously 32 and imaging protocols are listed in Supplemental Table 1.
Statistics
Continuous variables were analysed using parametric or non-parametric tests based on their distribution. Group comparisons utilised appropriate tests for categorical, ordinal and continuous variables. Correlations were assessed using Spearman's rank correlation. For intra-subject lesion comparisons, paired t-tests were used to assess mean differences between PRLs and non-PRLs within individual patients. As this was an exploratory study, no adjustment for multiple comparisons was performed, with p < 0.05 considered statistically significant. Detailed statistical methodology is provided in the Supplemental materials.
Results
The main study cohort (Sydney) included 60 consecutive MS patients enrolled in a longitudinal observational study, all of whom had reached at least 60 months of follow-up and had one SWAN scan at the latest follow-up.
At the SWAN timepoint, the average age of the cohort was 46.9 ± 9.9 years, with 44 females (73%) and 16 males. The patients had a mean disease duration of 13.8 ± 6.0 years, and the median Expanded Disability Status Score (EDSS) was 1.0 (interquartile range: 2). Disease-modifying Therapy (DMT) categories for the main cohort are described in Table 1.
Demographic and volumetric data between PRL+ and PRL− subjects. Significant values in bold.

PRL+: paramagnetic rim lesion-positive; PRL−: paramagnetic rim lesion-negative; SWAN: susceptibility-weighted angiography; IQR: interquartile range; % of TIV: percentage of total intracranial volume.
Of all the patients, 29 (48%) exhibited at least one PRL. In total, 80 out of 607 lesions were identified as PRLs, representing 13% of the total lesion count.
Demographic and volumetric comparisons between PRL-positive and PRL-negative subjects are presented in Table 1, showing significant differences in age and brain volume metrics. The associations between PRL metrics (count and volume) and various disease progression parameters are summarised in Table 2, highlighting significant correlations with brain atrophy measures.
Correlation between PRL count or PRL volume and demographic and radiological measures. Significant values in bold.
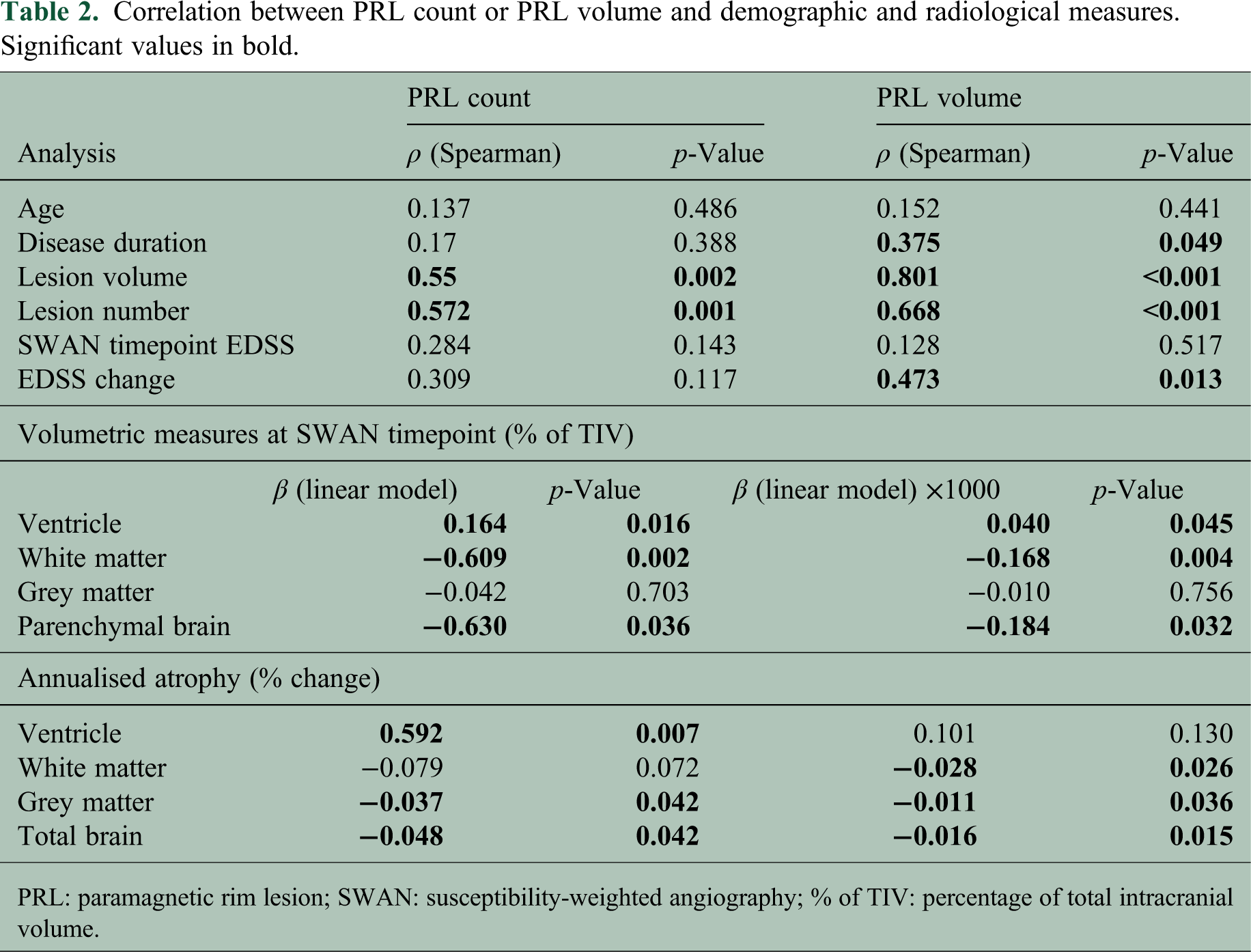
PRL: paramagnetic rim lesion; SWAN: susceptibility-weighted angiography; % of TIV: percentage of total intracranial volume.
Relationship between PRL and lesion size
The relationship between PRLs and non-PRLs across various metrics is shown in Table 3.
Lesion comparison between PRL and non-PRL for entire cohort and PRL+ patients only. Significant values in bold.

PRL: paramagnetic rim lesion; PRL+: paramagnetic rim lesion-positive; MD: mean diffusivity.
The combined analysis of all 607 lesions (80 rim-positive and 527 rim-negative lesions) across the entire cohort of 60 patients revealed that PRL had a significantly larger average volume (p < 0.0001). Additionally, the degree of tissue damage was more severe in PRL, as indicated by T1 intensity values and MD measurements (p < 0.0001 for both).
These differences persisted in intra-subject comparisons among 29 patients who had both lesion types (80 PRL, 262 non-PRL), with PRL exhibiting significantly larger volumes and more severe tissue damage (p < 0.0001 for all comparisons). Subject-level paired t-tests further confirmed these findings, with significant differences observed for lesion volume (p = 0.03), T1 intensity (p = 0.0001) and MD (p = 0.0001).
Relationship between PRL and lesion age
During the main study period of 60 months, 13 out of 60 patients (21.7%) developed 32 new lesions with volume above 100 mm3 within susceptibility visible areas (i.e. areas free of potential susceptibility distortion). Remarkably, all 32 of these new lesions exhibited a paramagnetic rim at the SWAN timepoint. Examples are shown in Figure 2.

Examples of new on-study lesions and rim at SWAN timepoint. Each row represents an individual patient. Column 1 (A, D, G, J): FLAIR image at baseline timepoints. Column 2 (B, E, H, K): FLAIR image of new lesion appearance at SWAN timepoint. Column 3 (C, F, I, L): Phase image at SWAN timepoint. SWAN: susceptibility-weighted angiography.
In five of these 13 patients, SWAN scans were available at the initial appearance of these lesions (for instance, at the ‘36 months’ timepoint). In every case, the paramagnetic rim was already visible at that early stage of lesion formation and remained detectable up to the SWAN timepoint.
Within the sub-group of 13 patients with new lesions, we observed distinct patterns of lesion development and rim presence. Two patients had only lesions that developed during the 5-year follow-up period with no lesions greater than 100 mm3 prior to the study period. Six patients had older lesions (>60 months) in addition to new lesions, but none of these older lesions displayed a paramagnetic rim. The remaining five patients exhibited both new lesions and older lesions that displayed paramagnetic rims.
Figure 3(A) illustrates the distribution of non-rim lesions (green), new on-study rim-positive lesions (red) and pre-study rim-positive lesions (blue) across these 13 patients (patient numbers 1–13 are indicated below each column). As shown in Figure 3(B), the age of on-study rim-positive lesions was distributed across the entire follow-up period.

Analysis of paramagnetic rim lesion (PRL) and non-PRL proportions and distribution in patients. (A) Distribution of non-PRL, old PRL (which appeared before the 60-month timepoint), and new on-study PRLs across 13 patients with new on-study lesions. (B) Distribution of the appearance timing of the 32 on-study lesions prior to the SWAN timepoint. (C) The proportion of lesions with a rim during the extended study period (n = number of lesions). (D) The proportion of patients with PRLs, grouped by lesion age categories (n = number of patients). SWAN: susceptibility-weighted angiography.
Some patients had conventional MRI data available from periods exceeding 60 months (72 to 96 months prior to the SWAN timepoint). In PRL-positive patients, rim-positive lesions became less common as lesions aged. While all lesions younger than 60 months (i.e. on-study lesions) had a paramagnetic rim, only 23% of lesions aged 72 months or more were rim-positive. This percentage decreased further to 13% for lesions at least 84 months old and 10% for lesions at least 96 months old (Figure 3(C)). A similar trend was observed at the patient level: all (i.e. 100%) patients with lesions less than 60 months old demonstrated PRL, while only 35% of patients with lesions older than 72 months had a PRL, decreasing to 29% at 84 months and 14% at 96 months (Figure 3(D)).
It should be noted that lesions labelled as ‘at least 72 months old’ include all lesions visible on scans 72 months prior to the SWAN timepoint, meaning some lesions are likely to be older than 72 months. Similar reasoning also applies to lesions at 84 and 96 months.
Rim decay over time
Out of the 60 patients, 40 had susceptibility imaging available at an additional timepoint, 24–36 months before the SWAN timepoint. Among these 40 patients, 24 had at least one PRL in the earlier scans, with a total of 61 PRLs identified. In the longitudinal analysis, 13 patients (with 16 lesions in total) showed a clear reduction or disappearance of the rim over time (Figure 4).

Example of rim decay. (A) Lesion at SWAN timepoint. (B) PRL on phase image 36 months prior to SWAN timepoint. (C) No clear rim at the SWAN timepoint. SWAN: susceptibility-weighted angiography; PRL: paramagnetic rim lesion.
Validation of association between the presence of paramagnetic rim and lesion age
To validate the relationship between the presence of a paramagnetic rim and lesion age, data from additional cohorts were used, including 100 RRMS patients from Charité – Universitätsmedizin Berlin, Germany, and 163 CIS and MS patients from Vall d’Hebron University Hospital, Barcelona, Spain. The average time of follow-up was 43 ± 23 months for the Berlin cohort and 43 ± 19 months for the Barcelona cohort.
Twelve patients from the Berlin cohort developed a total of 20 new lesions during the study; an example of a PRL is illustrated in Figure 5. In the Barcelona cohort, seven patients developed new lesions but only three patients had new lesions that met the size/location criteria for a total of five new, eligible lesions. Notably, all new lesions that met the size and location criteria from both the Berlin and Barcelona cohorts exhibited a paramagnetic rim on phase image, corroborating our main observation that paramagnetic rims consistently develop in newly formed lesions exceeding the designated volume threshold.
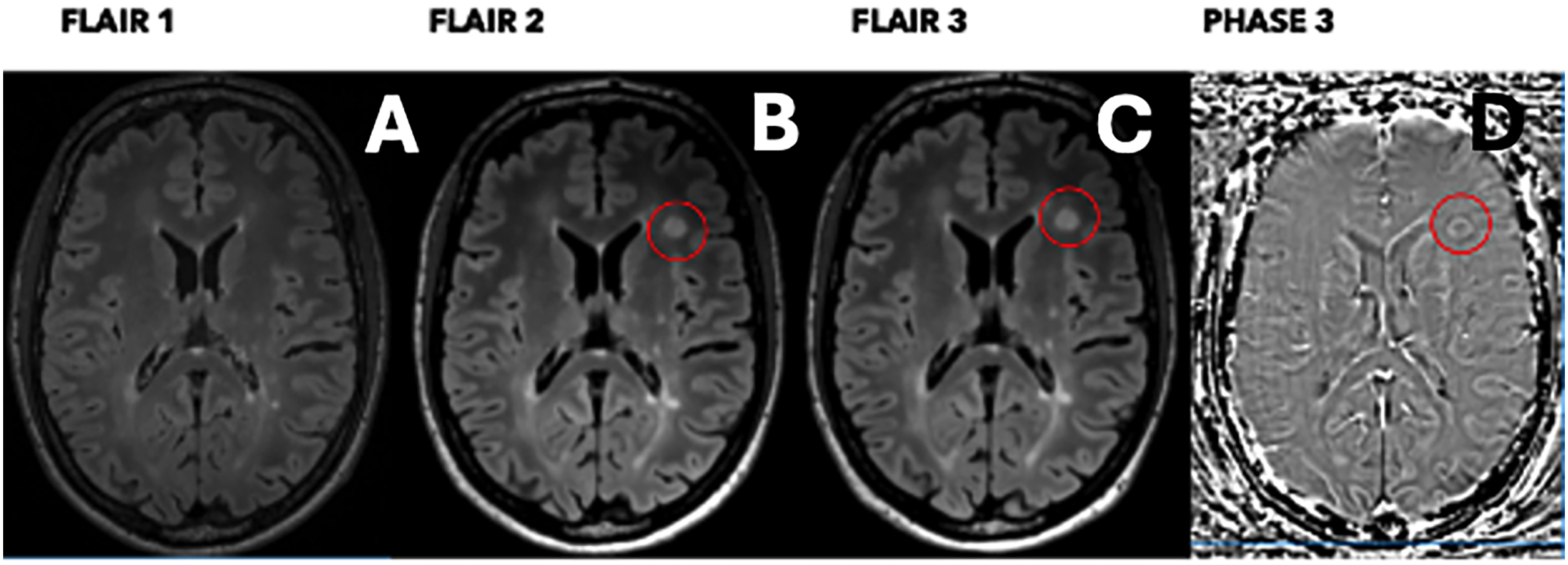
Example of on-study new lesion validation. FLAIR images at the (A) baseline timepoint, (B) intermediate timepoint and (C) last timepoint. Phase image (D) at the last timepoint. The circle highlights a new on-study lesion.
Discussion
In this exploratory study, we demonstrated a close relationship between the presence of a paramagnetic rim and lesion age. Despite the differences in study cohorts and imaging protocols, this finding was consistent across the entire dataset, with all recently (within 60 months) formed lesions exceeding 100 mm3 within paramagnetic visible areas exhibiting a paramagnetic rim – an observation validated in two independent international cohorts. Our results also revealed that the proportion of lesions displaying a paramagnetic rim decreased as lesion age increased. These findings suggest that the presence of a paramagnetic rim may be a relatively early feature of lesion formation, particularly in recently developed large lesions, rather than a marker that emerges solely during chronic inflammatory activity. While this does not exclude its role as a biomarker of chronic active lesions, our results highlight a strong temporal association with lesion age that may complement existing interpretations of PRL pathology.
Incidence of PRL
PRLs were present in 48% of patients, aligning with previous reports (35–63%).9,11,12,14,16–19,33,34 The prevalence of PRLs (13%; 80/607) was slightly higher than prior findings (4.5–11%),9,19,35,36 likely due to our exclusion of smaller (<100 mm3) lesions, which are less likely to have a paramagnetic rim.2,24 Our result also confirms a higher proportion of younger patients with PRLs, consistent with earlier studies.11,12,23
Relationship between PRL and disease progression
The presence of PRLs alone was not linked to disease progression, but PRL count and volume correlated with brain atrophy and EDSS change.
Prior studies show mixed results: some studies found a weak to moderate correlation between the presence11,34 or number15,18,33 of PRLs and progression in EDSS scores or brain atrophy, 17 while others noted that the volume of rim lesions is a more critical factor. 9 Conversely, others suggest non-rim lesion volume is a stronger predictor. 10 Despite variations due to small sample sizes, differing MRI techniques and identification methods, there is a general agreement that PRLs tend to correlate with disease progression, supporting our findings.
PRL groups in past studies often had higher lesion volumes,9,11,15,17,33 and relapse rates 11 or more advanced disease at baseline.17,33 We found that total lesion load strongly correlated with PRL volume and count. This suggests high T2 lesion volume may contribute to disease progression, as seen in Reeves et al., 11 where PRL–disability associations disappeared after adjusting for baseline T2 lesion volume. While baseline mean parenchymal volumes between PRL+ and PRL− subjects showed borderline statistical significance, the magnitude of the difference was minimal, and its clinical relevance remains unclear.
Are paramagnetic rims present in all recent lesions?
All 32 lesions developed during main study period (i.e. 60 months) displayed paramagnetic rims, a finding validated in two independent cohorts. Prior studies show a strong link between lesion size and rim formation, with larger (>100 mm3) lesions consistently displaying rims. However, this volume threshold reflects the detection capabilities of our imaging protocol and should not be interpreted as a universal minimum; rim visibility is known to vary depending on magnetic field strength, spatial resolution and the specific susceptibility-weighted sequence used.
While PRLs can form at lesion onset, the proportion of new lesions developing PRLs remains debated. 6
Multiple studies20,21,23,24 confirm that a significant proportion of new or recent MS lesions develop a paramagnetic rim, with lesion volume being the strongest predictor. Among newly formed or recent lesions, those that develop a rim are typically 3–4 times larger than rim-negative ones, which are often smaller than 100 mm3. As our study excluded lesions below this threshold, our findings align with these observations, reinforcing lesion size as a key determinant in paramagnetic rim formation.
Conversely, two studies2,9 reported no PRLs in newly formed lesions, though neither provided volumetric measurements, making it difficult to assess their findings in the context of lesion size.
A plausible explanation for the link between lesion size and PRLs, first proposed by Absinta et al., 23 suggests that larger acute lesions, combined with patient-specific factors such as age, tissue energetics and vascular status trigger a stronger inflammatory response at the lesion edge. This immune response aims to isolate the lesion from the surrounding unaffected parenchyma and involves the subtle opening of the blood–brain barrier in capillaries at the lesion edge. This phenomenon, manifesting as a centripetal pattern of contrast enhancement, is associated with the infiltration of blood-derived monocytes/macrophages and the activation of resident microglia, 18 which is reflected in the appearance of a phase rim. In contrast, smaller lesions experience less intense inflammation and are more conducive to ‘repair-promoting’ mechanisms, such as remyelination and new synapse sprouting. 37 These mechanisms could prevent a significant immune reaction at the lesion edge and reduce iron accumulation. Moreover, detecting PRLs requires a sufficient iron concentration within cells, which may not be reached in smaller lesions, reducing rim formation or MRI visibility. 2 Therefore, the smaller lesion size reduces the likelihood of paramagnetic rim formation or detection.
The lesion age-dependent nature of the paramagnetic rim is also supported by its evolution over time. Studies have observed that the rim gradually fades, 38 with reports suggesting a median disappearance time of approximately 7 years. 39 This is consistent with our findings, which show evidence of rim fading and a significant reduction in the proportion of PRL-positive lesions as lesion age surpasses 5 years.
The association of PRLs with recently formed lesions may explain their link to disease progression observed in multiple studies. Clinical decline and brain atrophy are more pronounced in patients with recent lesions, where inflammation remains active and drives ongoing damage. 40 This is further supported by the younger age of patients with PRLs. Consequently, PRLs could serve as an indicator of acute disease activity, underscoring the importance of timely detection and targeted therapeutic interventions.
A crucial question for future studies is whether PRLs eventually lose their rims 24 and transition into chronic inactive lesions or whether they continue to represent active chronic inflammation in the form of slowly expanding lesions. If the former, the use of PRLs as a biomarker for CALs in longitudinal studies and clinical trials will need to account for their natural decay over time. If the latter, distinguishing PRLs from recently formed lesions in cross-sectional or short-term longitudinal studies, including clinical trials, may be challenging. Understanding this distinction is vital for PRLs’ clinical utility as biomarkers for disease progression and therapeutic targeting.
Study limitations
This study has several limitations. The small sample size limits generalisability and excluding small lesions may bias results by overlooking their role in PRL formation. The 5-year follow-up, while informative, may not fully capture the long-term impact of PRLs on atrophy and clinical outcomes, especially since lesion age beyond 5 years remains uncertain.
The effect of DMT on PRL dynamics is difficult to assess due to the cohort's heterogeneous treatment regimens and durations. Larger, treatment-stratified studies with standardised imaging intervals are needed to clarify these interactions. Additionally, PRLs’ temporal evolution, particularly their fading over time, raises questions about their stability as markers of chronic active lesions, complicating their use in longitudinal studies and clinical trials.
In conclusion, we observed that within our study cohorts paramagnetic rims were consistently detected in all recently formed MS lesions exceeding a specific size threshold and that in some lesions the rim gradually diminishes as the lesion ages. While these observations reflect the constraints of our imaging protocol and lesion inclusion criteria, they provide insight into the temporal dynamics of PRLs and their potential role in tracking lesion evolution.
Supplemental Material
sj-docx-1-mso-10.1177_20552173251378788 - Supplemental material for The presence of a paramagnetic phase rim in multiple sclerosis is linked to lesion age: An exploratory study
Supplemental material, sj-docx-1-mso-10.1177_20552173251378788 for The presence of a paramagnetic phase rim in multiple sclerosis is linked to lesion age: An exploratory study by Samuel Klistorner, Tatiana Usnich, Margareta A Clarke, Deborah Pareto, Àlex Rovira, Friedemann Paul, Michael Barnett and Alexander Klistorner in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Data availability
The data that support the findings of this study are available from the corresponding author, upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: SK is the recipient of PhD scholarship from MS Research Australia. MAC is a 2023 ECTRIMS Postdoctoral Research Fellow and a recipient of the Skills Development Award from the UK MS Society. ÀR receives research support from Fondo de Investigación en Salud (PI19/00950 and PI22/01589) from Instituto de Salud Carlos III, Spain. DP receives research support from Instituto Carlos III, Spain (PI18/00823, PI22/01709) co-funded by the European Union. AK receives research support from MS Research Australia.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
