Abstract
Introduction
Multiple sclerosis (MS) is a central nervous system disorder and is frequently associated with fatigue. The Fatigue Scale for Motor and Cognitive Functions (FSMC) is an instrument to assess both motor and cognitive dimensions of fatigue in MS.
Methods
The study included 60 MS patients who completed the Persian version of the Modified Fatigue Impact Scale (MFIS) and a translated version of FSMC questionnaire. The FSMC was translated using standardized forward–backward translation, expert review, and patient feedback to ensure linguistic and conceptual validity. To evaluate test–retest reliability, participants completed the translated version of FSMC again after two weeks. Content validity was examined by a panel of eight experts.
Results
Sixty individuals with a mean age of 36.36 ± 9.7 years and an Expanded Disability Status Scale score of 3.00 ± 2.0 were enrolled in this study. Fifty-one (85%) of the participants were female. The Persian FSMC demonstrated strong content validity (Content Validity Index: 0.875–1.00; content validity ratio: 0.75–1.00). Internal consistency was excellent (Cronbach's alpha = 0.964). Test–retest reliability was strong for average scores (intraclass correlation coefficient (intraclass correlation coefficient (ICC) = 0.979), though fair to good for single measures (ICC = 0.540). Convergent validity was supported by strong correlations with MFIS scores (FSMC-total: r = 0.88; motor: r = 0.87; cognitive: r = 0.85; all p < .001). No floor/ceiling effects were observed.
Conclusions
The Persian version of the FSMC is a valid and reliable instrument for assessing fatigue in Iranian patients with MS. With strong psychometric properties, it is well-suited for clinical and research use in Persian-speaking populations.
Introduction
Multiple sclerosis (MS) is an autoimmune disorder of the central nervous system (CNS), primarily characterized by demyelination and neuronal damage. Its symptoms vary widely and may include physical disability, cognitive impairment, and disturbances in gait and balance.1–5 Fatigue—defined as a persistent sense of tiredness during the day that limits both physical and mental activity—is experienced by 75–95% of individuals with MS and is sometimes one of the earliest symptoms to appear.6–9 This fatigue significantly affects patients’ quality of life and overall well-being. 10 Numerous studies have explored various treatments and supplements aimed at alleviating MS-related fatigue.11,12 However, because fatigue is a multidimensional symptom, it is essential to distinguish between its different components when evaluating and managing it.13,14
A wide range of fatigue scales have been developed, each designed to assess different aspects or even distinct theoretical constructs of fatigue.14,15 Every questionnaire is based on its own conceptual framework, making each tool uniquely suited to specific types of research or clinical evaluation. 14
The Fatigue Scale for Motor and Cognitive Functions (FSMC) is widely used to assess both physical and cognitive fatigue, particularly in patients with MS. It is specifically designed to distinguish between these two dimensions of fatigue.14,16–18 The FSMC enables the evaluation of not only overall subjective fatigue but also its motor and cognitive components.16,17 The scale has been validated in large patient cohorts and across various populations.17,19–21 Notably, physical and cognitive fatigue as measured by the FSMC show distinct correlations with quality of life in individuals with MS: FSMC-Motor scores are associated with physical health domains, while FSMC-Cognitive scores are linked to mental health domains. 18
Self-report measures like the FSMC are susceptible to various sources of error and must be validated before being used in new populations or translated into other languages. Therefore, this study aimed to assess the validity of the Persian version of the FSMC as a measure of fatigue in individuals with MS. Additionally, we evaluated the scale's internal consistency, test-retest reliability, and concurrent validity within an Iranian MS population.
Material and methods
Participants
This study included 60 patients with MS, recruited between October 2022 and February 2023 from the Isfahan Multiple Sclerosis Center of Kashani hospital, Iran. All participants provided written informed consent prior to enrollment.
Inclusion criteria were: a confirmed diagnosis of MS based on the 2017 McDonald criteria, 22 Persian as a native language, and the ability to read Persian. Exclusion criteria included: major neurological or psychiatric disorders (e.g. stroke, epilepsy, brain tumor, CNS infections, major depressive disorder, bipolar disorder, schizophrenia, and substance abuse); a history of developmental or learning disabilities; chronic systemic diseases (e.g. diabetes, renal or liver failure, COPD); other potential causes of fatigue (e.g. anemia, thyroid disorders, Vitamin D deficiency, sleep disorders); and any history of MS relapse or corticosteroid treatment within four weeks prior to assessment. Participation was voluntary, and individuals were free to withdraw at any time.
Demographic data including age, gender, marital status, years of education, and employment status were recorded. Clinical information collected from MS patients included disease duration, MS phenotype, and scores on the Expanded Disability Status Scale (EDSS). All neurological assessments were conducted by a qualified neurologist.
This study was done according to Helsinki's declaration and its subsequent amendments. The study was ethically approved by the Ethics Committee of Isfahan University of Medical Sciences, Isfahan, Iran (Ethics code: IR.ARI.MUI.REC.1401.043).
FSMC questionnaire
The FSMC is composed of 20 items—10 assessing cognitive fatigue and 10 assessing motor fatigue—each rated on a scale from 1 to 5. The total score ranges from 20 to 100. Scores between 43 and 53 indicate mild fatigue, 53 to 63 represent moderate fatigue, and scores above 63 reflect severe fatigue. 17
Translation
The translation, cultural adaptation, and validation of the FSMC questionnaire were conducted in accordance with established guidelines. 23 In the first step, two independent translators—each familiar with either the questionnaire's subject matter or the cultural and linguistic intricacies of the Persian language—translated the validated English version of FSMC into Persian. In the second step, a third independent translator reviewed both versions and reconciled any differences to produce an initial Persian draft.
In the third step, this draft was back-translated into English by two independent translators who had no prior exposure to the original FSMC. In the fourth step, a review committee comprising all translators, clinicians, and methodologists assessed the back-translations to evaluate content validity, 24 resolve discrepancies, and finalize a prefinal version.
In the fifth step, the prefinal version was reviewed by 10 MS patients, who provided feedback on the clarity and comprehensibility of the items, suggesting improvements where needed. Based on their input, necessary revisions were made to produce the final Persian version.
In the seventh step, 60 MS patients completed both the Persian FSMC and MFIS questionnaires. Two weeks later, the FSMC was readministered to the same group. Finally, the test–retest reliability and internal consistency of the Persian FSMC were assessed using statistical analyses. 25
Validity
A panel of eight experts—comprising two neuropsychiatrists and six neurologists—was invited to evaluate the validity of the questionnaire. Each item was rated on a four-point scale for relevance by the panel. Each expert assessed the relevance, clarity, and comprehensibility of each item using a 4-point Likert scale. Content validity was assessed using the Item-Level Content Validity Index (I-CVI). The I-CVI for each item was calculated by dividing the number of experts who rated the item as either 3 or 4 by the total number of experts. An I-CVI value of 0.78 or higher was considered acceptable for panels of this size. Items with I-CVI values between 0.70 and 0.77 were identified for potential revision, while those with values below 0.70 were considered for elimination.24,26 For the content validity ratio (CVR), experts were asked to rate each item as “essential,” “useful but not essential,” or “not essential.” A CVR value of 0.75 or higher was deemed acceptable based on the number of participating experts. 27
To assess convergent validity, participants completed the MFIS questionnaire. MFIS has been previously validated in Iranian population.28,29 The relationship between the FSMC and MFIS was examined due to their structural and content similarities. Correlation coefficients were interpreted as weak (< 0.30), moderate (0.30–0.69), or strong (≥ 0.70).
Reliability
To assess the internal consistency of the questionnaire, we used Cronbach's alpha coefficient, a standard measure of reliability. The values were interpreted as follows: ≥ 0.90 reflects excellent consistency, 0.80–0.89 indicates good consistency, 0.70–0.79 is considered acceptable, 0.60–0.69 is questionable, 0.50–0.59 is poor, and values below 0.50 indicate unacceptable consistency. 30 Test–retest reliability was evaluated by having participants complete the questionnaire a second time approximately two weeks after the initial administration. The resulting intraclass correlation coefficient (ICC) was interpreted as follows: values ≥ 0.75 indicated excellent reliability, 0.40–0.74 reflected fair to good reliability, and values below 0.40 were considered poor.
Floor and ceiling effect
We assessed floor and ceiling effects by determining whether over 15% of participants scored at the minimum or maximum possible values, respectively.
Data analysis
Statistical analyses were performed using IBM SPSS Statistics version 26 (IBM Corp., Armonk, NY, USA). Both frequency (percentage) and mean ± standard deviation were used to describe the results.
To evaluate the internal consistency of the FSMC, Cronbach's alpha was calculated, with values above 0.80 interpreted as indicating excellent reliability. The ICC was used to assess test–retest reliability, with an ICC greater than 0.70 considered acceptable. Cronbach's alpha was also computed for each of the three components of the questionnaire to determine internal consistency within each domain.
The Pearson correlation coefficient was used to evaluate the relationship between the FSMC and MFIS scores.
Results
Participants
A total of 60 individuals with MS met the eligibility criteria and enrolled. Among them, 85% were female, with an average age of 36.36 ± 9.7 years and an EDSS score of 3.00 ± 2.0. Demographic and clinical characteristics are summarized in Table 1.
Baseline characteristics of the study participants.

Note. RRMS: relapsing-remitting multiple sclerosis, PMS: progressive multiple sclerosis, EDSS: Expanded Disability Status Scale.
Validity
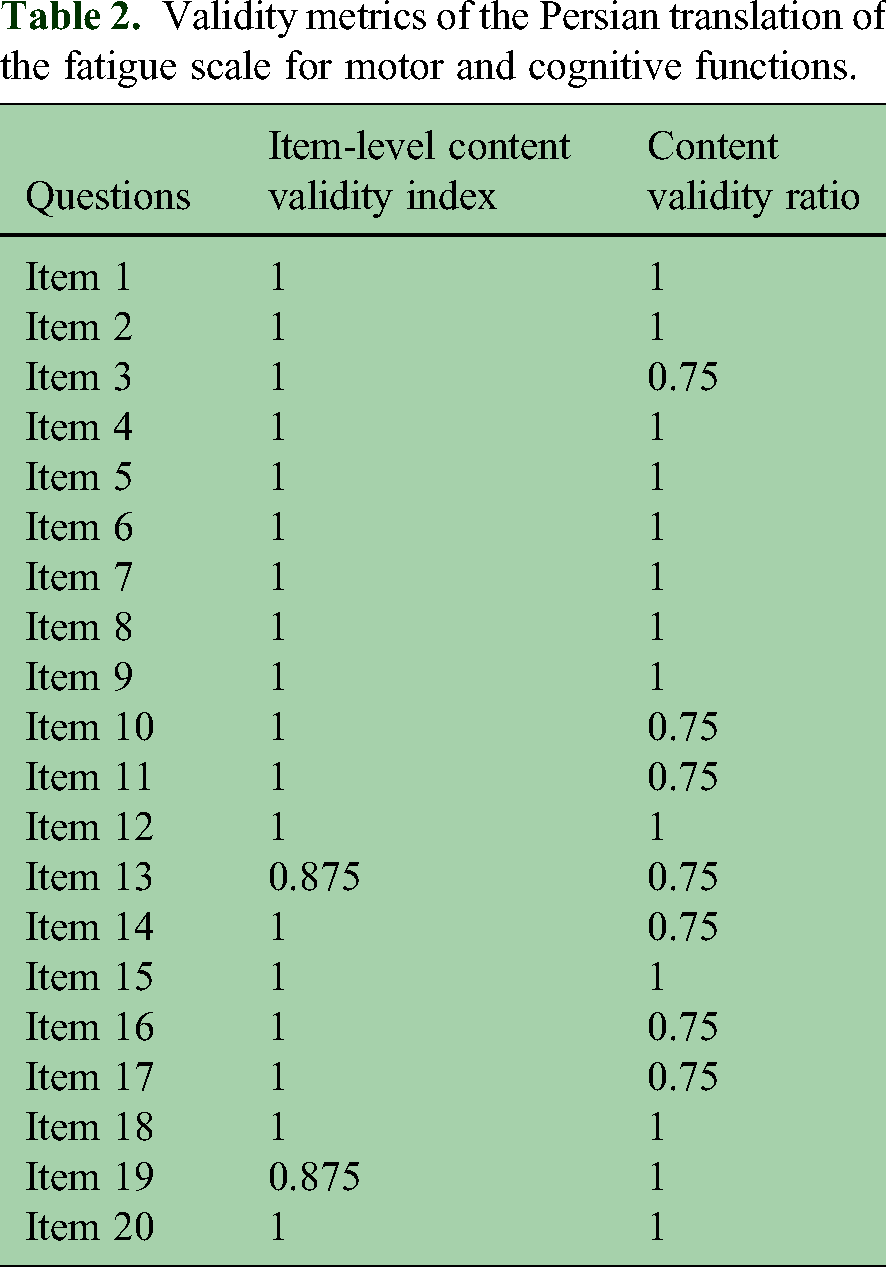
Table 2 displays the I-CVI and CVR for each questionnaire item. The I-CVI values ranged from 0.875 to 1.00, indicating strong expert agreement on item relevance. Additionally, all items demonstrated acceptable CVR values, ranging from 0.75 to 1.00.
Validity metrics of the Persian translation of the fatigue scale for motor and cognitive functions.
A strong correlation was observed between total FSMC and total MFIS scores (r = 0.88, p < .001). The FSMC subscales also showed high correlations with the MFIS total score: r = 0.85 for cognitive function and r = 0.87 for motor function (both p < .001). Specifically, the cognitive subscale of the FSMC was strongly correlated with the cognitive subscale of the MFIS (r = 0.83, p < .001), and the motor subscale of the FSMC was highly correlated with the physical subscale of the MFIS (r = 0.86, p < .001). These findings are detailed in Table 3.
Convergent validity of the Persian version of the fatigue scale for motor and cognitive functions.

FSMC: Fatigue Scale for Motor and Cognitive Functions, MFIS: Modified Fatigue Impact Scale.
The EDSS demonstrated a moderate correlation with the total FSMC score (r = 0.55, p < .001), as well as with its cognitive (r = 0.46, p < .001) and motor (r = 0.57, p < .001) subscales. In contrast, no significant correlation was found between disease duration and the total FSMC score (r = 0.17, p = .20), or its motor (r = 0.21, p = .09) and cognitive (r = 0.16, p = .23) subscales.
Reliability
The Cronbach's alpha coefficient of the current Persian version of the FSMC was 0.964, showing excellent internal consistency. For cognitive domain Cronbach's alpha coefficient was 0.936 and for motor domain 0.942, showing excellent internal consistency. To assess the test–retest reliability of the instrument, the ICC was calculated using a two-way mixed-effects model with a consistency definition (Type C). The ICC for single measures was 0.540 (95% CI [0.448–0.646]), indicating fair to good reliability of individual items. However, the ICC for average measures was 0.979 (95% CI [0.970–0.987]), reflecting excellent reliability when item scores were averaged. The F-test was significant, F(51, 1989) = 47.98, p < .001), confirming that the observed ICC values were significantly greater than zero. These results indicate that the instrument demonstrates strong stability over time, particularly when considering total or subscale scores rather than individual items.
For cognitive domain, the ICC for single measures was 0.554 (95% CI [0.459–0.660]), indicating fair to good reliability of individual items. However, the ICC for average measures was 0.963 (95% CI [0.947–0.976]), reflecting excellent reliability when item scores were averaged. For motor domain, the ICC for single measures was 0.586 (95% CI [0.498–0.683]), indicating fair to good reliability of individual items. However, the ICC for average measures was 0.966 (95% CI [0.952–0.977]), reflecting excellent reliability when item scores were averaged. The reliability indices of questionnaire items are shown in Table 4.
Reliability metrics of the Persian translation of the Fatigue Scale for Motor and Cognitive Functions.

The corrected item–total correlations were examined separately for the first (T1) and second (T2) administrations of the questionnaire. All items showed acceptable to strong item–scale correlations in both rounds. For the first administration (T1), values ranged from 0.588 to 0.825, and for the second administration (T2), from 0.590 to 0.882. These findings indicate that each item was consistently aligned with the overall construct at both time points, supporting the internal reliability and stability of the instrument across administrations. Furthermore, the “Cronbach's Alpha if Item Deleted” values for all items were either lower than or approximately equal to the total scale alpha, indicating that each item contributed positively to the internal consistency of the scale. Therefore, no item appeared to be redundant or detrimental to reliability.
Floor and ceiling effect
In our study group, only one participant (1.67%) achieved the lowest possible score, and none reached the highest score. This confirms the absence of both floor and ceiling effects, as neither exceeded the 15% threshold.
Discussion
Validating questionnaires in the native language of the target patient population is essential, as cultural and linguistic differences can influence understanding and interpretation. This study aimed to validate the FSMC for use in an Iranian population of individuals with MS. The high ICC values demonstrate strong test–retest reliability of the Persian version, while the high Cronbach's alpha coefficients across all three components, including total score, cognitive domain, and motor domain, confirm acceptable internal consistency.
Both the CVR and CVI values for each item met acceptable thresholds, indicating that the Persian version of the FSMC demonstrated satisfactory content validity.
After confirming the two-dimensional structure through factor analysis, Cronbach's alpha and ICC were calculated for both the total score and each subscale. The results demonstrated excellent internal consistency across the total scale and subscales, indicating strong interrelatedness among items and suggesting that both the full scale and its subcomponents effectively measure a common underlying construct. Additionally, the number of items was sufficient to ensure reliability. These findings align with those reported in previous validation studies of the FSMC in German, Danish, and Finnish populations,17,19,20 supporting the cross-cultural robustness of the scale and reinforcing its utility for clinicians and researchers in neurorehabilitation and psychometrics globally.
The strong correlation between FSMC and MFIS was expected, given their similar structures. Both tools assess fatigue severity and include comparable subscales. These results support the FSMC's convergent validity and highlight the need for further research to identify the most effective tool for assessing fatigue. While the high correlation suggests some potential for interchangeability, the instruments may also be complementary. For instance, the FSMC provides a focused assessment of cognitive and motor fatigue, whereas the MFIS offers broader coverage, including psychosocial aspects. This distinction may influence tool selection depending on the clinical or research context. Future studies should investigate which tool is more sensitive to change or better suited for specific patient needs or intervention outcomes.
As EDSS scores increased, there was a significant rise in both the total FSMC score and the cognitive and motor subscales, consistent with previous studies.17,19 However, disease duration did not show a significant correlation with the total score or any of the subscales.
One of the strengths of this study was the inclusion of participants with a range of disease duration, disability and phenotype. Furthermore, the use of the MFIS allowed for a comparative evaluation of scores between the two questionnaires.
However, the study also had several limitations. It was conducted at a single center, which may limit the generalizability of the results to individuals with MS living in other regions of Iran. The relatively small sample size is another limitation that could affect the broader applicability of the findings. Additionally, the absence of an age- and gender-matched healthy control group further restricts the study's comparative scope. Although the Persian version of the FSMC exhibited excellent test–retest reliability at the total score level, the moderate reliability observed at the individual item level warrants consideration. This finding suggests that, while the instrument is highly reliable for evaluating overall fatigue, the variability in individual items may limit its utility for detailed or fine-grained analyses. Furthermore, while the scale performed well within the Iranian context, cultural or linguistic differences may affect its applicability in other Persian-speaking communities, warranting further study.
Conclusion
In conclusion, the Persian version of FSMC demonstrated strong reliability and validity for assessing both physical and cognitive aspects of fatigue, showing excellent internal consistency and precision. It is considered a suitable and recommended tool for measuring fatigue in the Iranian population with MS. Given its strong psychometric properties, the Persian version of the FSMC appears suitable for use in clinical trials and intervention studies aimed at evaluating fatigue management strategies in individuals with MS.
Footnotes
Acknowledgments
We extend our sincere gratitude to the staff and fieldworkers of the Isfahan Multiple Sclerosis Center of Kashani Hospital, as well as to the participants, whose cooperation made this study possible.
Author contributions
F-T, K-A, A-P, and I-A designed the study; F-T and A-M contributed to data collection; A-A analyzed the data; I-A supervised the study; A-A drafted the text. All authors contributed to the manuscript and approved the submitted version.
Declaration of conflicting interests
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Permission to reuse and copyright
No figures were taken from any journals, websites, and other sources.
Declaration of generative AI and AI-assisted technologies in the writing process
During the preparation of this work, the authors used ChatGPT in order to improve readability and language. After using this tool/service, the authors reviewed and edited the content as needed and take full responsibility for the content of the publication.
Data availability statement
The datasets generated and analyzed during the current study are available from the corresponding author (IA) upon reasonable request.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
