Abstract
We present real-world data on patients switching from anti-CD20s to fumarates for various motivations in this retrospective observational study of 43 patients from three multiple sclerosis centers. Recurrent infections on anti-CD20s were the most common reason for switching to fumarates. Patients experienced limited disease activity on fumarates (83.7% were free from relapse and new MRI lesions), suggesting effectiveness was maintained. Of the 16.3% with disease activity on fumarates, 57.1% also had disease activity on anti-CD20s. Tolerability was the main reason for discontinuing fumarates. Future studies will provide additional insight into how to effectively and safely transition from anti-CD20s to fumarates.
Introduction
Selecting a disease-modifying therapy (DMT) for patients living with multiple sclerosis (MS) can be challenging given the varying efficacy and safety profiles with over 20 available options. Proposed treatment strategies include escalation, induction, high efficacy, and de-escalation with little consensus on the best approach. Increasing evidence suggests that utilizing high efficacy treatment early, including B-cell depletion, may result in improved long-term outcomes compared to using an escalation approach. 1
Treatment decisions may be complicated as risks and benefits change with age. Clinical relapses and MRI activity generally decrease with age, possibly due to immunosenescence. 2 Additionally, observational studies suggest an increased risk of infections with age and disability among MS patients with long-term anti-CD20 use.3,4 As patients age, the risk of infectious complications may outweigh the advantages of high efficacy therapies. Therefore, de-escalation may help achieve a more favorable benefit-risk profile. Fumarates may offer a safe transition from anti-CD20 therapy, however, evidence examining this switch is limited. The aim of this observational study was to explore the characteristics, disease activity, and reasons for discontinuation among patients with MS who transition from anti-CD20 to fumarate therapy.
Methods
Eligible patients for this study met the following inclusion criteria: (a) had a diagnosis of MS, (b) were most recently treated with an anti-CD20 within 12 months before switching to a fumarate therapy prior to July 2023, and (c) were 18 years or older at time of de-escalation. Patients were excluded if they had other chronic neurological illnesses that might mimic MS. We conducted a retrospective chart review to identify eligible patients at three MS. Each site obtained Institutional Review Board approval.
Patients were followed from anti-CD20 (ocrelizumab or rituximab) start until fumarate (dimethyl, diroximel, or monomethyl fumarate) discontinuation or until most recent visit prior to data extraction. Demographics, reasons and time to discontinuation, and disease activity outcomes (clinical relapse, contrast enhancing lesions, new T2 lesions, or a combined composite measure) were retrospectively collected from electronic medical records (EMRs) for anti-CD20 and fumarate therapy. The form of MS was collected from diagnoses by the treating clinicians for each patient at the time of fumarate initiation. If there was no evidence for disease activity recorded the EMR, patients were considered to have no disease activity for the purpose of this study analysis. Expanded Disability Status Scale was not examined as it was not regularly captured in EMRs at the time of this study. Descriptive statistics were used to characterize the cohort. Unless specified, percentages are reported using the entire cohort as the denominator. Sensitivity analyses were conducted to further explore outcomes among those with relapsing-remitting MS (RRMS) only, and those with at least one year of treatment on both anti-CD20 and fumarate therapy.
Results
Forty-three patients were identified (30 from Rocky Mountain MS Center at the University of Colorado, 5 from the Rocky Mountain MS Clinic at Salt Lake, and 8 from New York University Langone MS Comprehensive Care Center). Patients were predominantly female, white, non-Hispanic, with relapsing-remitting MS (Table 1).
Patient characteristics who transitioned from anti-CD20 to fumarate (N = 43) therapies.
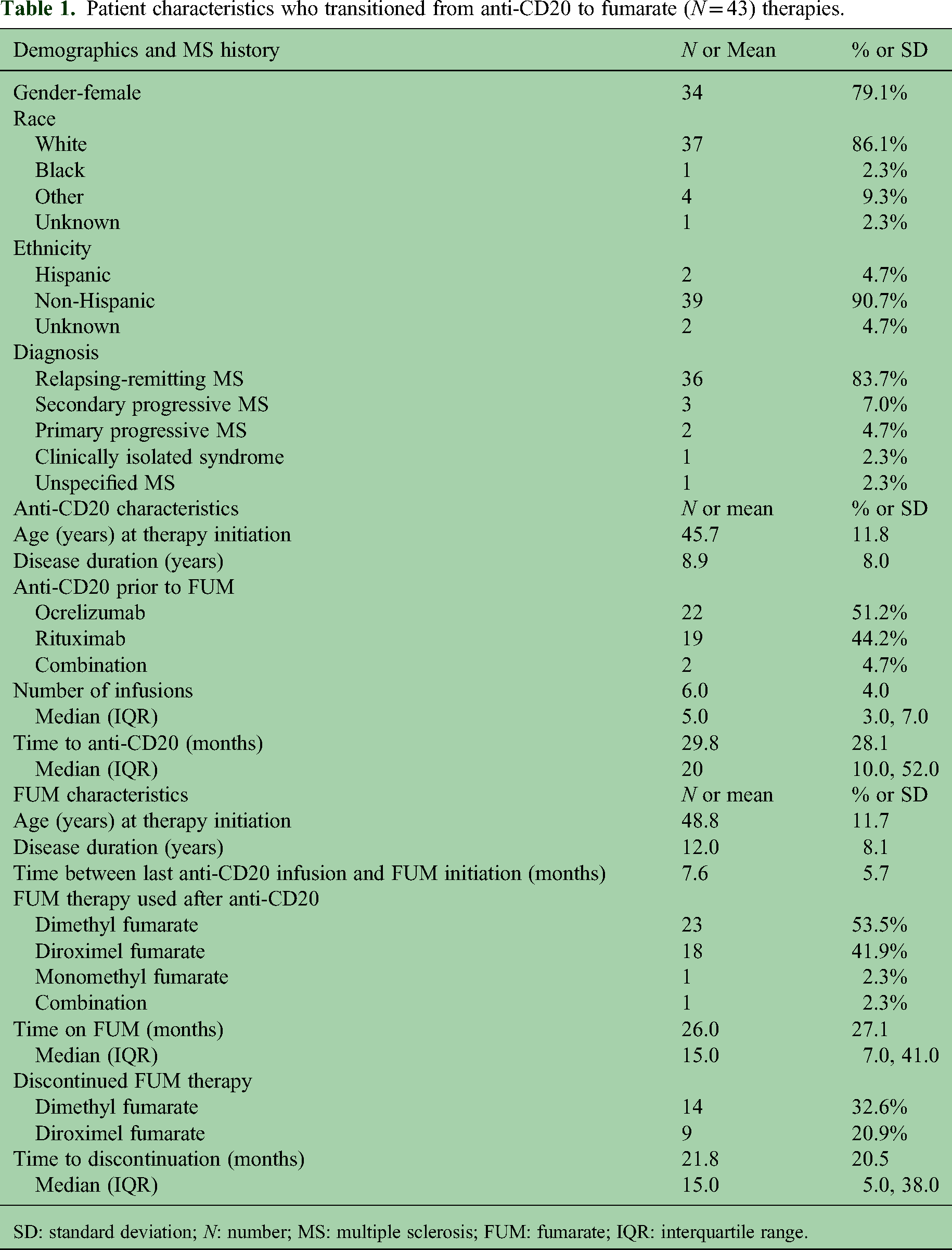
SD: standard deviation; N: number; MS: multiple sclerosis; FUM: fumarate; IQR: interquartile range.
Approximately half (48.8%) initiated anti-CD20 due to having disease activity on their prior DMT (Supplementary Table S1). At anti-CD20 initiation, patients had a mean age of 45.7 and had been diagnosed with MS for 8.9 years. A total of 22 and 19 patients initiated ocrelizumab and rituximab respectively, with two patients having received both treatments. Patients received anti-CD20 treatment for a mean of 29.8 months. Most patients discontinued anti-CD20 therapy due to recurrent infections (Figure 1(a)). Adverse events, excluding infections, leading to discontinuation occurred in 25.8% of patients on anti-CD20s and were commonly infusion reactions (14.0%). While on anti-CD20 therapy, 74.4% had no disease activity, while 11.6%, 16.3% and 2.3% of patients experienced a clinical relapse, new T2 lesion or enhancing lesion, respectively (Figure 1(b) and Supplementary Table S2). The mean time from anti-CD20 initiation to disease activity was 13.18 months (SD = 14.25).

Bar graphs demonstrating proportions for (a) reasons for discontinuing anti-CD20 therapy, (b) disease activity outcomes experienced on anti-CD20 therapy, (c) reasons for discontinuing fumarate therapy, and (d) disease activity outcomes experienced on fumarate therapy after anti-CD20 treatment.
Patients initiated fumarate therapy on average 7.6 months after their last anti-CD20 infusion. At the time of fumarate initiation, patients had a mean age of 48.8 (79.1% of patients were age 40 or older) and disease duration of 12.0 years. Patients were treated with dimethyl fumarate (53.5%), diroximel fumarate (41.9%), monomethyl fumarate (2.3%), or a combination of fumarates (2.3%). Patients were followed for a mean of 26.0 months (range: 0–122 months) on fumarate therapy with 53.5% discontinuing treatment after a mean of 21.8 months (median = 15). Adverse events led to discontinuation in 34.9% of patients (Figure 1(c)).
While on fumarate therapy, 4.65%, 11.63% and 0.0% patients experienced a clinical relapse, new T2 lesion or enhancing lesion, respectively (Figure 1(d) and Supplementary Table S2). Disease activity on average occurred 3.6 months (SD = 2.57) after fumarate initiation, and 7.1 months (SD = 2.73) after last anti-CD20 infusion. The mean time from fumarate initiation and disease activity was 3.6 months (SD = 2.57). Overall, 83.7% of patients remained free of relapses and MRI lesions after switching to fumarates. Of the 7 patients with disease activity on fumarates (2 with a relapse and 5 with new T2 lesion on MRI), mean age was 42.8, mean disease duration was 10.9 years, and most (57.1%) also had disease activity on anti-CD20 therapy; both of the patients who had a clinical relapse on fumarates also had new T2 lesions on anti-CD20s with one also having a clinical relapse. None of the patients who were clinically stable on anti-CD20s relapsed after switching to fumarates. When restricting analyses to RRMS patients only, trends were similar (Supplementary Tables S3 and 4). All patients with disease activity while on either anti-CD20s or fumarates had RRMS. When restricting analyses to those with a minimum of 12 months on both anti-CD20s and fumarates (Supplementary Tables S5 and 6), the only disease activity while on fumarate therapy were new T2 lesions.
Discussion
We present real-world data on the patient's experience in switching from anti-CD20 to fumarate therapies. Given that older age, disability, and duration of treatment have been associated with serious infections in patients on anti-CD20s, it is not surprising that infections were the most common reason for discontinuation of anti-CD20s,. 4 The mean washout duration of 7.6 months plus the mean fumarate treatment of 26 months led to almost 3 years of follow-up after the last anti-CD20 infusion. Overall, 53.5% discontinued fumarate therapy, comparable to prior studies with 47% discontinuation within 2 years, primarily due to tolerability issues from gastrointestinal symptoms.5,6
Efficacy was maintained on fumarate therapy (16.3% composite disease activity vs. 25.6% on anti-CD20s). Subjects with disease activity on fumarates tended to have had disease activity on anti-CD20 therapy (57.1%), emphasizing the importance in selecting stable patients. When restricting analyses to patients who maintained at least one year of treatment on both anti-CD20 and fumarate therapy, none experienced a clinical relapse on fumarate therapy, though three patients did have new T2 lesions. Age may also help inform when to de-escalate. 7 This study included younger patients (48.8 years) than DOT-MS (52.8 years), which was a discontinuation study that was stopped early due to 26.7% of subjects developing disease activity, 8 supporting a de-escalation approach in these younger patients. In another study, all of the 25 MS patients, who switched from ocrelizumab to diroximel fumarate, remained free of relapse and MRI activity. 9 Notably, only the first year of fumarate therapy was examined and participants were required to be stable on ocrelizumab prior to switching. Another discontinuation study, DISCOMS, included older patients that mostly stopped platform medications and saw 12.2% disease activity in the discontinuation arm. 10
Limitations include some DMTs, such as dimethyl fumarate, were available as both generic and brand forms which may have an influence on tolerability or disease activity. Unfortunately, however, we were not able to differentiate between these forms of treatment in this study. Further, MRIs were not re-baselined after starting either anti-CD20 or fumarate therapy, limiting the ability to determine the timing of new T2 lesion formation. Importantly, this study was retrospective and utilized convenience sampling approaches. These results may not be generalizable to younger patients considering switching from anti-CD20s to fumarate therapy but appears more efficacious than stopping treatment. Additionally, no measure of disability was assessed as it was not reliably captured in EMRs, limiting our understanding of effectiveness of these therapies among those with progressive forms of disease. Future clinical studies will provide additional insight into patient selection (age, disease stability duration) who may benefit the most in transitioning from anti-CD20 therapy to fumarates.
Supplemental Material
sj-docx-1-mso-10.1177_20552173251371808 - Supplemental material for Transition from anti-CD20 therapies to fumarates as a treatment strategy: A multicenter, retrospective observational experience
Supplemental material, sj-docx-1-mso-10.1177_20552173251371808 for Transition from anti-CD20 therapies to fumarates as a treatment strategy: A multicenter, retrospective observational experience by Brandi L Vollmer, Tammy Hoyt, Timothy W West, Josef Gutman, Monica Benitez, Evan L Riddle, James B Lewin, Jason P Mendoza and Enrique Alvarez in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
TWW has received compensation for activities such as advisory boards, lectures and consultancy with the following companies and organizations: Biogen, EMD Serono/Merck, Genentech/Roche, Horizon/Amgen and TG Therapeutics.
ELR, JBL, and JPM are employees of Biogen.
EA has received compensation for activities such as advisory boards, lectures and consultancy with the following companies and organizations: Biogen, Celgene/BMS, EMD Serono/Merck, Genentech/Roche, Horizon/Amgen, Novartis, Sanofi, Cionic, and TG Therapeutics and research support from: Atara, Biogen, BMS, Genentech/Roche, Novartis, Sanofi, TG Therapeutics, Patient-Centered Outcomes Research Initiative, National Multiple Sclerosis Society, National Institutes of Health, and Rocky Mountain MS Center. Remaining authors does not have a conflict of interest.
Ethical approval
Institutional Review Board approval was obtained from each site, including Rocky Mountain MS Center at the University of Colorado, the Rocky Mountain MS Clinic at Salt Lake, and New York University Langone MS Comprehensive Care Center.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by Biogen.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
