Abstract
Background
Mood-behavioral symptoms, fatigue and pain are frequent among people with multiple sclerosis (pwMS). Music therapy (MT) is a non-pharmacological option for symptomatic treatment in neurological diseases.
Objectives
To assess effects of 6-week-outpatient MT on anxiety (primary outcome) as well as: depression, fatigue, pain and body perception (secondary outcomes) in pwMS.
Methods
We randomized pwMS 1:1 to music therapy group (MTG) and control group (CG). Both had one 45-min session per week, MTG with a monochord, CG without music. A blinded rater assessed endpoints at baseline and week 6 with standardized questionnaires (e.g. hospital anxiety and depression scale, HADS) and quantitative sensory testing (QST). Immediate session effects were also assessed. The analysis included linear mixed models, adjusted for pwMS's characteristics and baseline scores.
Results
Fifty-seven pwMS (age: 50.1 ± 12.4 years, 47 women, MTG: n = 30, CG: n = 27) were included. In MTG, anxiety levels (HADS) did not differ from CG at week 6 (p = 0.109). Among secondary outcomes, psychosocial fatigue was reduced (p = 0.029), QST heat pain thresholds were higher (p = 0.024) and immediate subjective effects stronger in MTG (e.g. feeling balanced: p < 0.001, relaxed: p < 0.001, less pain: p < 0.001).
Conclusion
Despite no difference in anxiety, we observed effects of receptive MT on fatigue, pain and body perception.
Introduction
Multiple sclerosis (MS) is an inflammatory and neurodegenerative disease of the central nervous system, with a tremendous impact on functionality and quality of life. 1 Mood-behavioral and cognitive symptoms are common in people with MS (pwMS). 2 Moreover, fatigue is a common and disabling symptom, which can appear early in the course of the disease. 3 Next to pharmacological treatments, several non-pharmacological interventions—like mindfulness training 4 —were reported to reduce these symptoms in pwMS. In this regard, there is data suggesting a role of music therapy (MT) as a complementary therapeutic approach in MS. 5
MT is one branch of art therapy, where direct effects of music and a variety of music-based interventions (MBI) are used for therapeutic purposes. It aims at accomplishing individualized goals, 6 related to physical, psychological and social needs of patients, and is a valuable asset for the treatment of several neurological disorders. For example, MT was shown to improve anxiety in patients with dementia, 7 speech and social skills in people with autism, 8 or motor function in patients with Parkinson's disease. 9 Moreover, MT could reduce pain and depression in patients suffering from chronic pain conditions.10–12
Still, the majority of previously published data in MT on pwMS are based on studies with uncontrolled designs, 13 low number of participants14,15 or use of various MBIs, 15 which hampers their comparison and does not allow to draw definite conclusions about exact effects. In January 2023, the National Institute of Health (NIH) published a call for controlled studies in the field of MT and MBIs, to promote clearer research designs. 16 Furthermore, many prior MT-studies were conducted in an “inpatient” setting,17–19 although most pwMS are regularly followed-up in outpatient settings.
We aimed at examining the effects of a specific MBI in pwMS, using a randomized, rater-blinded, controlled trial design. We focused on the effects of receptive MT with an instrument called “treatment monochord.” Our primary outcome was reduction of anxiety symptoms, which is a frequent and disabling symptom among pwMS. 2 We chose the monochord as it is described to have a relaxing effect. 20 Our secondary outcomes were reduction in depressive symptoms, fatigue, stress, health-related quality of life, and pain thresholds. Moreover, we investigated potential immediate effects of the MBI on pain levels and body perception.
Methods
Study participants
We recruited pwMS prospectively from our MS center in the Outpatient Clinic of Neurology, University Hospital of Basel. The recruitment period lasted from May 2021 to May 2022.
The inclusion criteria were
diagnosis of MS, according to 2017 Mc Donald Criteria,
21
both relapsing-remitting and progressive MS allowed age ≥ 18 years expanded disability status scale (EDSS) score ≤ 6.5
The exclusion criteria were
reported or medically recorded diagnosis of serious psychiatric disorders, including acute and severe depression, suicidality and severe anxiety syndromes other currently life-threatening or severely disabling physical disorders >2 MS relapses within the last year occurrence of MS relapse within 3 months before recruitment change in disease modifying treatment (DMT) within 3 months before recruitment change in symptomatic MS medication (e.g. for depression, anxiety, fatigue, pain) within 3 months before recruitment pregnancy inability to lie supine for 15 min severely compromised hearing (unless participant has sufficient hearing aid) numbness on the back of both hands which would affect assessment of pain thresholds
Musical background knowledge or previous experience with MT was not necessary (but also not an exclusion criterion) for participation. The flow of study participants including screening process and dropouts is depicted in Figure 1.
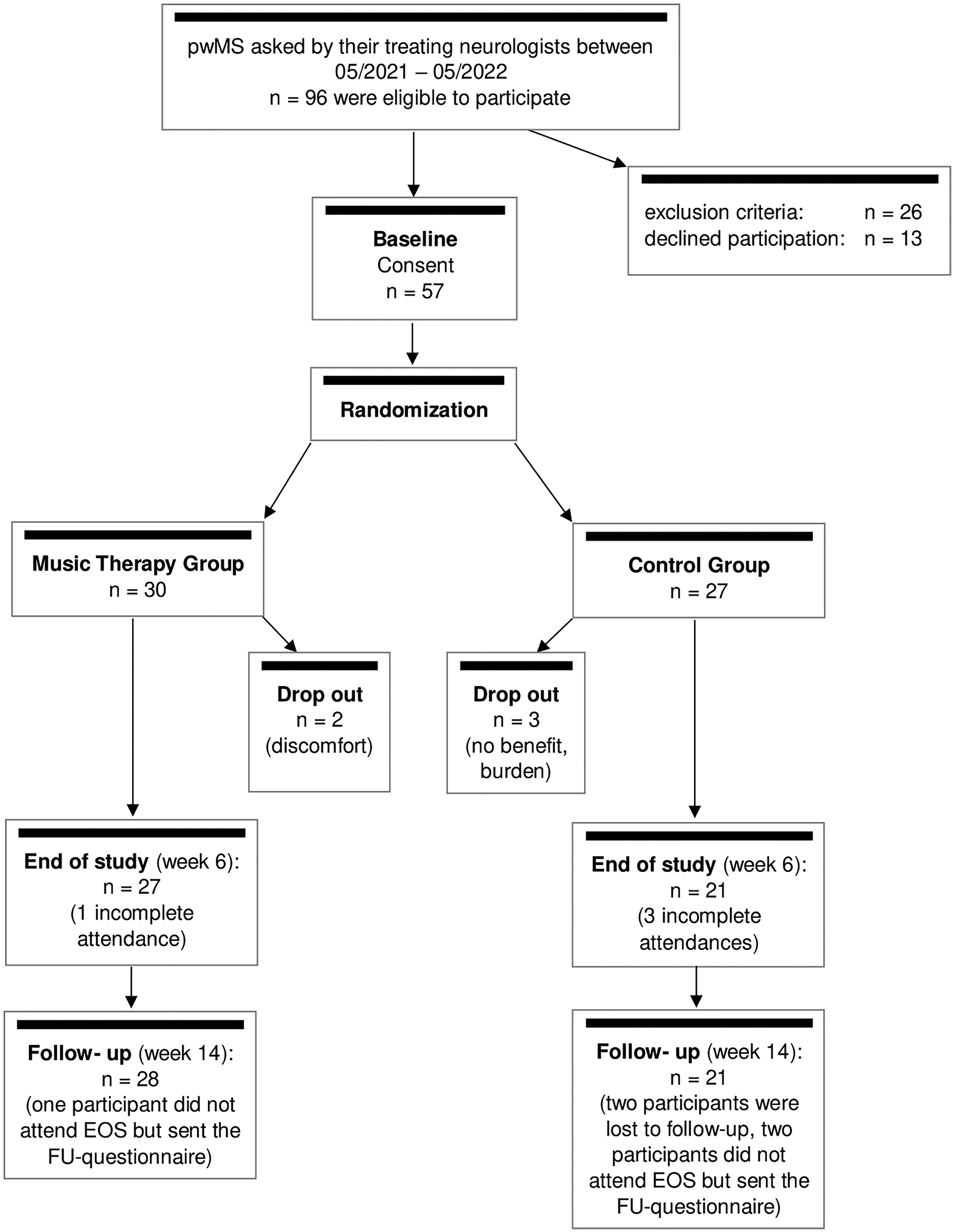
Flow chart of the study. Note that we performed an intention-to-treat analysis, including participants that dropped out.
Study design
“Music Therapy with a Monochord in Multiple Sclerosis (MUTIMS)” was a monocentric, rater-blinded, randomized-controlled study. Participants were randomized 1:1 to music therapy group (MTG) and control group (CG) using sealed envelopes (extensive description provided in the supplementary material).
Participants of both MTG and CG took part in six outpatient sessions, once per week, with the same music therapist (JK). Participants could not be blinded to the treatment group, but the raters who obtained all outcome measures at baseline and at end-of-study (EOS) visit were fully blinded (KE, NCF).
Study intervention
In MTG, participants received a MBI with the “treatment monochord” (Figure 2). After a short introduction, the participant laid down on top of the monochord and remained in a relaxed supine position. The music therapist played the 55 strings attached below the body of the monochord. The tuning was in two different tones (cis and gis). The sound is transmitted via air and via resonance, making it possible to listen to the music and feel its vibration. After 15 min of playing, the music therapist guided the participant back out of relaxation and ended with a closing conversation, so each session lasted approximately 45 min.

The “treatment monochord” that was used in the study. It was designed by music therapist Joachim Marz in Switzerland in the 1980s. Its strings are attached below the instrument's “body” and the participants are lying on the instrument during the treatment.
In CG, participants received a similar intervention with one difference: Instead of perceiving the music, CG relaxed on top of the monochord in silence. The music therapist, monochord and therapy room (with stable temperature of 22° Celsius) were the same for each group to minimize confounding. We selected this controlled design to detect specific effects of the MBI and control for effects due to relaxation while lying on the instrument or contact to the therapist.
Assessment of primary and secondary outcomes
At baseline, participants underwent a standardized clinical neurological examination (Neurostatus EDSS). In case the EDSS had been obtained less than 3 months before inclusion, we used this as baseline score. The following validated questionnaires were used to assess primary and secondary outcomes: hospital anxiety and depression scale (HADS-A/D), 22 for assessment of anxiety and depressive symptoms, modified fatigue impact scale (MFIS), 23 a self-reported measure of fatigue in daily life with different subscales, Perceived Stress Questionnaire (PSQ20) 24 and Short Form Health Survey (SF-36). 25 The HADS questionnaire was performed every 2 weeks during the study and at follow-up, 2 months after the last session (week 14). All other questionnaires were performed at baseline and EOS (week 6).
Quantitative sensory testing (QST) 26 was performed at baseline and EOS, for assessment of pain thresholds. It was performed according to the standardized protocol described by the German Research Network for Neuropathic pain, 26 using standardized instructions for participants (German language). For extensive description of QST see supplementary material, documents. We assessed immediate session effects using questionnaires “A” and “B” (Q A&B, see supplementary material) before and after every session, based on a previous study with the monochord. 27 Participants had to rate statements from 0 to 10, using a visual analog scale (VAS).
Statistical analysis
All participants who had measurements of the outcomes of interest available were included in the analyses. The study protocol and statistical analysis plan are available as supplementary material. The sample size estimation was conducted for the primary research question, the effect of MT versus CG on anxiety (HADS-A) at the end of six sessions. The sample size was estimated using simulations based on an internal data sample from the Swiss multiple sclerosis cohort study (SMSC), a large observational study of pwMS in our MS center. 28 The relevant improvement in HADS-A was set at 2.5 points and chosen as relevant treatment effect of MT. The baseline mean HADS-A score was assumed to be 4.7 with standard deviation 3.3 for both groups, in line with the SMSC data. The correlation between baseline and post-treatment HADS-A score was estimated to be 0.63 based on the same data. An ANCOVA model was fit in each of the simulated data sets of different sizes, and the p-value for the effect of group was determined. To achieve a power of 80% at alpha level 0.05, we determined that 60 pwMS should be included.
An intention to treat analysis was performed using the available data. For the analysis of the primary outcome, we used a linear regression model to assess whether the HADS-A score after six sessions is different in participants in MTG compared to participants in CG. The intervention (MTG or CG) was the explanatory variable of interest and the model was adjusted for the baseline HADS-A score. We report the estimate with the Wald 95% confidence interval (CI) and p-value for each fixed-effect estimate. For the secondary outcomes assessed at baseline and after the sessions, we used the same linear model as in the primary analysis. Each model was adjusted for the baseline score of the corresponding endpoint. The immediate treatment effects, assessed with Q A&B, were assessed using a linear mixed model, acknowledging clustering of data within sessions and participants. We used the interaction term between treatment and time (after vs. before session) and treatment group as fixed effect of interest, and the continuous VAS-score as outcome. We also performed a subgroup analysis (for primary- and all secondary outcomes) in participants with higher and lower EDSS scores at baseline (median split at 3.5). To investigate the potential influence of cognition on the findings of our study, we repeated the primary analysis adding the interaction: effect of treatment and symbol digit modalities test (SDMT)—score at baseline.
All analyses were performed in R version 4.3.1. We did not correct the presented p-values of secondary analyses for multiple testing; these should be interpreted as hypothesis generating, rather than confirmatory. Finally, a post-hoc power analysis was performed for the primary analysis.
Standard protocol approvals, registrations, and participant consents
The study was approved by the local ethics committee (Ethikkommission Nordwest- und Zentralschweiz, BASEC 2021-00067) and conducted in accordance with the declaration of Helsinki. All participants gave written informed consent before inclusion. The study was registered at the Swiss National Clinical Trials Portal (SNCTP000004292) and the German Clinical Trials Register (DRKS00024549).
The corresponding author (AP) declares that she takes full responsibility for the data, analyses, interpretation and conduct of the research, that she has full access to all data and has the right to publish all data. Coded data could be made available within the next 10 years upon reasonable request to the corresponding author. We report according to the CONSORT reporting guidelines. 29
Results
From the 57 participants randomized, there were five dropouts, two in MTG (Figure 1). The participants’ characteristics are summarized in Table 1. Overall, there were no group-differences at baseline.
Baseline characteristics of the study participants stratified by treatment group.
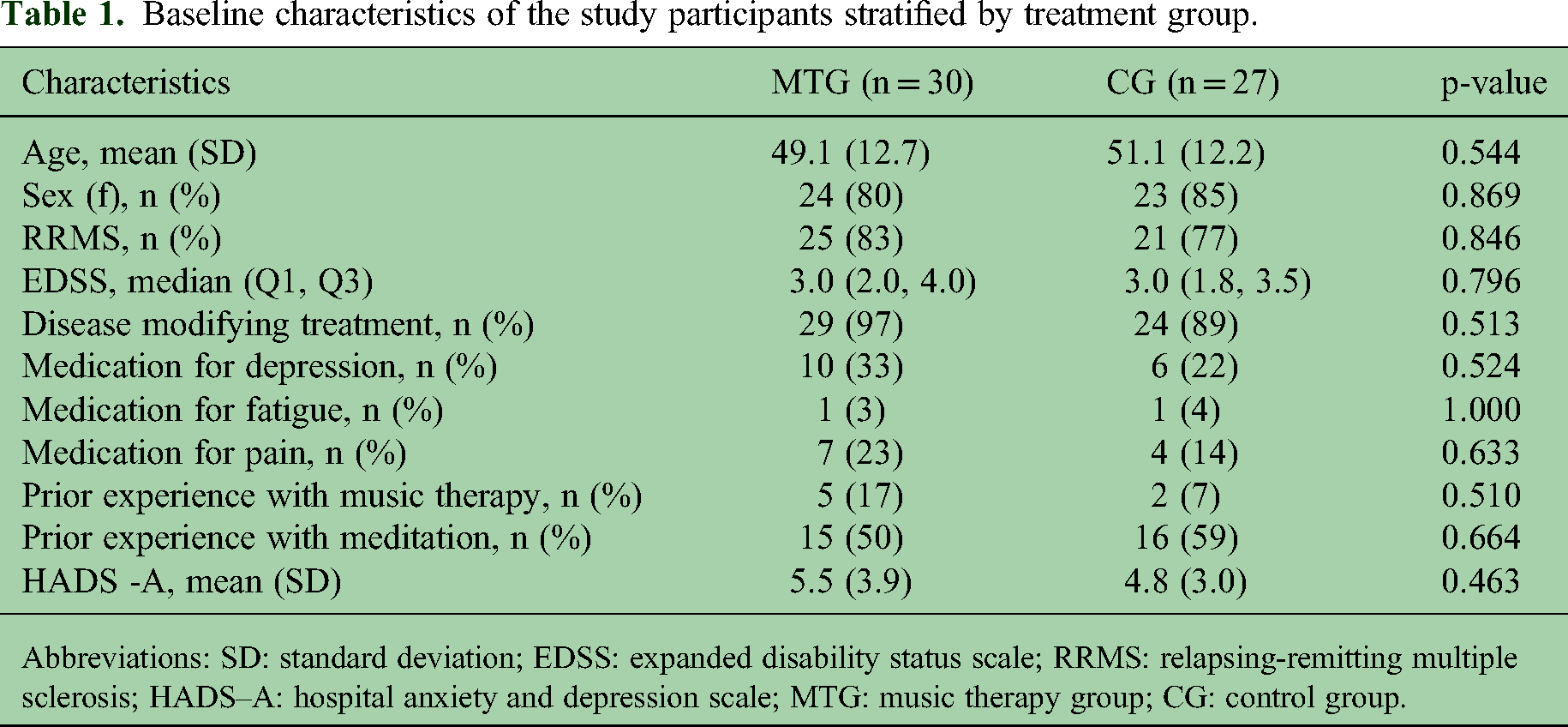
Abbreviations: SD: standard deviation; EDSS: expanded disability status scale; RRMS: relapsing-remitting multiple sclerosis; HADS–A: hospital anxiety and depression scale; MTG: music therapy group; CG: control group.
Effect of music therapy on primary outcome (HADS-A)
At EOS (week 6), MTG showed a mean HADS-A score of 5.1 ± 4.1 (mean at baseline being: 5.5 ± 3.9), and CG: 3.3 ± 2.6 (mean at baseline being 4.8 ± 3). While MTG had an HADS-A score that was on average 1.01 point higher, this was not significant in the adjusted model (Beta = 1.01, 95% CI [−0.23, 2.24], p = 0.109) (Figure 3).

Course of HADS-A scores in the two study groups. Note that the last session was performed at week 6, there was no intervention between week 6 and week 14.
At follow-up (week 14), anxiety levels were 0.77 points lower in MTG compared to CG (Beta = −0.77, 95% CI [−2.23, 0.69], p = 0.295) (Figure 3).
The post-hoc power analysis revealed a power of 37% for the primary outcome.
Effect of music therapy on secondary outcomes
The results are summarized in Table 2. There was no group-difference in depression, total fatigue score, stress level, or overall health-related quality of life at week 6. We found an average 0.47-point higher improvement on the psychosocial fatigue subscale of the MFIS in MTG compared to CG (95% CI [−0.88, −0.05]). Moreover, heat pain thresholds measured with QST were 1 degree Celsius higher in MTG vs. CG at EOS (95% CI [0.14, 2.00]). We did not find group-differences in other pain thresholds and QST parameters (data not shown). Immediate effects on body perception after the sessions were stronger in MTG for feeling balanced, relaxed, in touch with the body and for feeling less pain (Table 3). The subgroup analysis in pwMS with higher disability (EDSS > 3.5, n = 15) did not show differences between MTG and CG regarding primary- and secondary outcomes (data not shown; course of primary outcome HADS-A in Figure 4).

Course of HADS-A in the two study groups, stratified for disability level at baseline. Although there were no significant group-differences, participants with higher disability (B) showed a tendency for continuously dropping anxiety levels only in the MTG and not in the CG.
Results regarding the secondary outcomes.
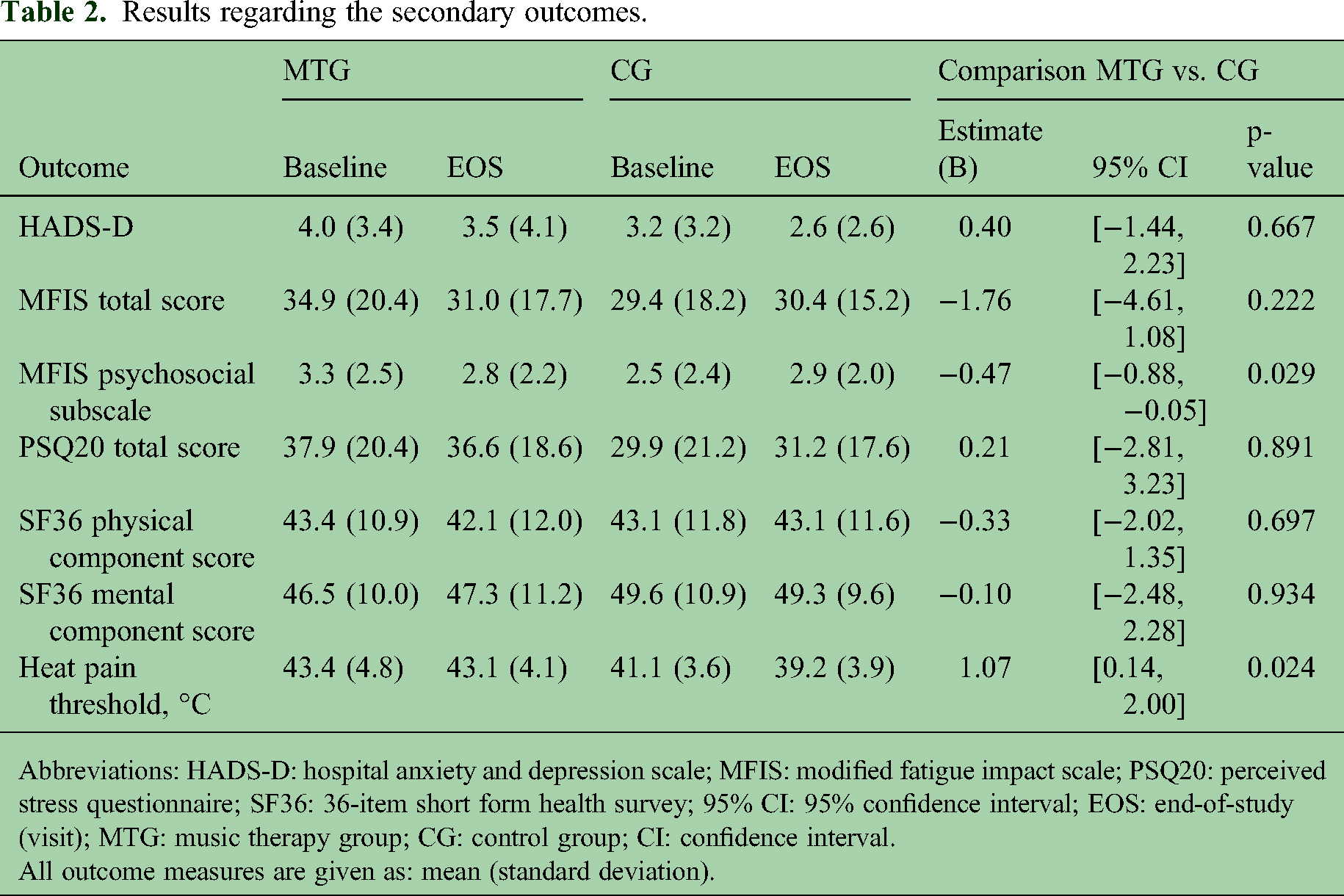
Abbreviations: HADS-D: hospital anxiety and depression scale; MFIS: modified fatigue impact scale; PSQ20: perceived stress questionnaire; SF36: 36-item short form health survey; 95% CI: 95% confidence interval; EOS: end-of-study (visit); MTG: music therapy group; CG: control group; CI: confidence interval.
All outcome measures are given as: mean (standard deviation).
Immediate effects after the sessions expressed as change on the visual analog scales (after vs. before the sessions).

The results (“effects”) are given as estimates (B) with [95% CI], for the analysis of the intermediate treatment effect (i.e. the questionnaires “B” score after each session minus “A” score before each session). They represent the effect after adjusting for the baseline value of each outcome. Note that positive numbers (e.g. “I feel balanced”) correspond to increase and negative numbers (e.g. “I feel pain) to a decrease on the VAS.
Abbreviations: MTG: music therapy group; CG: control group; VAS: visual analog scale; CI: confidence interval.
We did not identify an interaction between SDMT and group on our primary outcome (Beta = 0.02 [95% CI: −0.08, 0.11]), nor a main effect of baseline SDMT score (Beta = −0.04 [95% CI: −0.1, 0.02]).
Discussion
Recently, the NIH published a call for controlled, highly structured studies with MT in people with neurological diseases. 16 There is an unmet need for data in the field of MT in pwMS as the type of intervention, its duration and administration vary 18 and previous published data about MT in pwMS came from small and uncontrolled studies. 17 Therefore, it proves difficult to form specific conclusions about its efficacy in MS. 14 Thus, we chose to examine one specific, structured MBI with a “treatment monochord” in a randomized, controlled, rater-blinded trial. Since whole body vibration may positively influence several MS-associated symptoms, 30 we considered the monochord as particularly interesting for our study.
After six MT sessions, we did not find a difference in anxiety, depression, overall fatigue and perceived stress, compared to CG. However, we identified clear immediate MBI-effects on participant's body perception. In a previous study with active, outpatient MT in pwMS, improvement on anxiety and self-esteem appeared after a much longer period of time (12 months) of continued MT intervention. 14 In our study, participants in MTG reported immediate positive effects, which however did not result in a sustainable improvement over 6 weeks. Based on previous own data with the monochord in post-traumatic psychological disorders, 27 we initially hoped, that the 6-week-design would be sufficient to detect treatment effects, but the post-hoc power analysis showed that our study was apparently underpowered to find a significant group-difference for anxiety in pwMS. Perhaps longer duration and/or more frequent sessions might be needed to achieve sustained improvement of anxiety in outpatient MT. Moreover, a therapeutic effect of contact with the music therapist and/or relaxation on the monochord could be partially responsible for our results.
As depicted in Figures 3 and 4, CG also showed a tendency for reduced anxiety levels at week 6 compared to baseline. A previous study using a mindfulness-based intervention revealed positive effects on anxiety, depression and fatigue in pwMS; 4 however, the CG in that study received only standard care. We used a stricter design, since we were specifically interested in the exact impact of the monochord (through its sound and vibration) on MS-associated symptoms.
The monochord was previously shown to promote relaxation and less exhaustion in cancer patients.20,31 In line with this, pwMS in our MTG reported a more relaxed state and showed less psychosocial fatigue compared to CG. Furthermore, the monochord was shown to reduce pain symptoms in chronic pain conditions 32 and in patients with post-traumatic psychological disorders. 27 Effects of MT on pain have also been found in prior studies in palliative care and cardiovascular diseases.11,12 In our study, we found a reduction in pain levels after the sessions in MTG. Moreover, our standardized QST showed higher heat pain thresholds in MTG. Although these results must be interpreted with caution (exploratory outcomes), they may suggest a relaxing and analgesic effect. Further studies are needed to confirm that MT with the “treatment monochord” may be beneficial in pwMS who also suffer from chronic pain.
When interpreting the findings of our study, it has to be noted that in a “regular” therapeutic MT- interaction the music therapist can choose from a wide range of musical instruments and MBIs, according to the patient's individual situation, needs and demands. 6 Some studies have examined active forms of MT in pwMS to improve motor symptoms and could show effects on right-hand movement 33 and ambulation. 34 Interestingly, in people with dementia, receptive MT could reduce anxiety and disruptive behavior but active MT did not, 7 pointing to the possible relevance of targeted therapy. To our knowledge, there are no studies comparing receptive and active MT in MS.
Our subgroup analysis gave comparable results to the overall group. In pwMS with higher disability (EDSS >3.5), we observed a tendency for dropping anxiety levels until week 14 only in MTG, as depicted in Figure 4. The reasons for stronger effects on this subgroup are speculative. It could be that patients with higher burden of symptoms can profit more from a symptomatic treatment. Moreover, social/psychological factors might play a role, since these patients are often more isolated, with less social interactions and indeed more motivated to participate in complementary therapies.
Of note, our study had overall a good adherence with totally five dropouts, of which only two were in MTG (2/30 = 6.7% of participants that received MT). Since many pwMS—particularly with progressive course—suffer from bothering symptoms that can often be only partly addressed by medications, non-pharmacological treatments like MT are typically well accepted. 35 This should encourage clinicians, therapists and researchers to further use and investigate such treatments to understand which patients profit most.
Main limitation of our study is the fact that participants could not be blinded to treatment assignment. We had a high percentage of female participants, which limits the generalizability of our results to male MS patients. Moreover, the VAS-based measures used to assess immediate session effects were previously described but not validated in pwMS. The main strength of our study lies in its prospective, randomized-controlled design and blinded raters. This contrasts with many previous uncontrolled studies with MT and other non-pharmacological interventions.
As a conclusion, receptive MT with a “treatment monochord” showed good adherence on an outpatient basis and favorable effects on psychosocial fatigue and pain, as well as several immediate subjective effects. No group-difference in anxiety was observed. Studies considering more frequent sessions and longer duration as well as comparison of active and receptive MBIs are needed to further explore the benefits of MT in pwMS.
Supplemental Material
sj-docx-1-mso-10.1177_20552173251352712 - Supplemental material for Music therapy with a monochord in multiple sclerosis (“MUTIMS”): A randomized, controlled, rater-blinded trial
Supplemental material, sj-docx-1-mso-10.1177_20552173251352712 for Music therapy with a monochord in multiple sclerosis (“MUTIMS”): A randomized, controlled, rater-blinded trial by Jenni Kuhlmann, Katarina Ebner, Andrea Zimmer, Nikki Rommers, Nuria Cerdá-Fuertes, Bettina Fischer-Barnicol, Lisa Dinsenbacher, Joachim Marz, Marcus D’ Souza, Katrin Parmar and Jens Kuhle, Ludwig Kappos, Athina Papadopoulou in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Supplemental Material
sj-docx-2-mso-10.1177_20552173251352712 - Supplemental material for Music therapy with a monochord in multiple sclerosis (“MUTIMS”): A randomized, controlled, rater-blinded trial
Supplemental material, sj-docx-2-mso-10.1177_20552173251352712 for Music therapy with a monochord in multiple sclerosis (“MUTIMS”): A randomized, controlled, rater-blinded trial by Jenni Kuhlmann, Katarina Ebner, Andrea Zimmer, Nikki Rommers, Nuria Cerdá-Fuertes, Bettina Fischer-Barnicol, Lisa Dinsenbacher, Joachim Marz, Marcus D’ Souza, Katrin Parmar and Jens Kuhle, Ludwig Kappos, Athina Papadopoulou in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Supplemental Material
sj-docx-3-mso-10.1177_20552173251352712 - Supplemental material for Music therapy with a monochord in multiple sclerosis (“MUTIMS”): A randomized, controlled, rater-blinded trial
Supplemental material, sj-docx-3-mso-10.1177_20552173251352712 for Music therapy with a monochord in multiple sclerosis (“MUTIMS”): A randomized, controlled, rater-blinded trial by Jenni Kuhlmann, Katarina Ebner, Andrea Zimmer, Nikki Rommers, Nuria Cerdá-Fuertes, Bettina Fischer-Barnicol, Lisa Dinsenbacher, Joachim Marz, Marcus D’ Souza, Katrin Parmar and Jens Kuhle, Ludwig Kappos, Athina Papadopoulou in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Supplemental Material
sj-docx-4-mso-10.1177_20552173251352712 - Supplemental material for Music therapy with a monochord in multiple sclerosis (“MUTIMS”): A randomized, controlled, rater-blinded trial
Supplemental material, sj-docx-4-mso-10.1177_20552173251352712 for Music therapy with a monochord in multiple sclerosis (“MUTIMS”): A randomized, controlled, rater-blinded trial by Jenni Kuhlmann, Katarina Ebner, Andrea Zimmer, Nikki Rommers, Nuria Cerdá-Fuertes, Bettina Fischer-Barnicol, Lisa Dinsenbacher, Joachim Marz, Marcus D’ Souza, Katrin Parmar and Jens Kuhle, Ludwig Kappos, Athina Papadopoulou in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Supplemental Material
sj-docx-5-mso-10.1177_20552173251352712 - Supplemental material for Music therapy with a monochord in multiple sclerosis (“MUTIMS”): A randomized, controlled, rater-blinded trial
Supplemental material, sj-docx-5-mso-10.1177_20552173251352712 for Music therapy with a monochord in multiple sclerosis (“MUTIMS”): A randomized, controlled, rater-blinded trial by Jenni Kuhlmann, Katarina Ebner, Andrea Zimmer, Nikki Rommers, Nuria Cerdá-Fuertes, Bettina Fischer-Barnicol, Lisa Dinsenbacher, Joachim Marz, Marcus D’ Souza, Katrin Parmar and Jens Kuhle, Ludwig Kappos, Athina Papadopoulou in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Acknowledgments
We want to thank all people with MS who participated in our study.
Data availability statement
The corresponding author (AP) declares that she takes full responsibility for the data, the analyses and interpretation, and the conduct of the research, that she has full access to all the data and that she has the right to publish all data, separate and apart from the guidance of any sponsor. Coded data that are not shown within this article could be made available within the next 10 years upon reasonable request to the corresponding author from qualified investigators.
Conflict of interests and disclosures of all authors
This study was funded by research fonds from Jens Kuhle, Athina Papadopoulou, Kathrin Parmar in the University Hospital of Basel, CH and from the Swiss National Science Foundation (to AP; Project Number: PZ00P3_216468).
Jenni Kuhlmann received speaker fees for the MS Society of Switzerland and the MS Society of Germany.
Katarina Ebner served on an advisory board from Lundbeck.
Bettina Fischer-Barnicol served on advisory boards from Biogen and Merck.
Marcus D'Souza is an employee of the University Hospital Basel and the CEO of Neurostatus-UHB Ltd He has received research support from the University Hospital Basel.
Katrin Parmar holds a grant of the Baasch-Medicus foundation, her former institution (University Hospital Basel) received speaker's honoraria from Novartis and ExceMED and travel support by Novartis, Switzerland.
Jens Kuhle received speaker fees, research support and travel support and/or served on advisory boards for ECTRIMS, Swiss MS Society, Swiss National Research Foundation (grant no. 320030_189140/1), University of Basel, Bayer, Biogen, Celgene, Genzyme, Merck, Novartis, Roche, Sanofi and Teva.
Ludwig Kappos’ institutions (University Hospital Basel and RC2NB) have received research support and payments that were used exclusively for research support for Dr Kappos’ activities as principal investigator and member or chair of planning and steering committees or advisory boards in trials sponsored by Actelion, Addex, Almirall, Bayer HealthCare, Celgene, CLC Behring, Genentech, GeNeuro, Genzyme, Merck Serono, Mitsubishi Pharma, Novartis, Octapharma, Ono, Pfizer, Receptos, F. Hoffmann-La Roche, Sanofi-Aventis, Santhera, Siemens, Teva, UCB, and XenoPort; licence fees for Neurostatus products; research grants from the Swiss Multiple Sclerosis Society, the Swiss National Research Foundation, the European Union, and the Roche Research Foundation.
Athina Papadopoulou received speaker fees/fees for advisory boards/for consulting from Sanofi-Genzyme, Eli Lilly, AbbVie, Lundbeck, Pfizer and TEVA (all used for her research in the University of Basel) and travel support from Bayer AG, Abbvie, Teva and Hoffmann-La Roche. Her research was supported by the University- and University Hospital of Basel, the Swiss Multiple Sclerosis Society, the “Stiftung zur Förderung der gastroenterologischen und allgemeinen klinischen Forschung sowie der medizinischen Bildauswertung,” the “Freie Akademische Gesellschaft Basel” and the Swiss National Science Foundation (Project numbers: P300PB_174480 and currently: PZ00P3_216468).
Andrea Zimmer, Nikki Rommers, Nuria Cerdá-Fuertes, Lisa Dinsenbacher, and Joachim Marz have nothing to disclose.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Swiss National Science Foundation (grant number PZ00P3_216468, to Athina Papadopoulou).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
