Abstract
Background
Cholesterol is an essential and major component of myelin. Brain cholesterol turnover in humans can be studied noninvasively by metabolic labeling of the brain-specific cholesterol metabolite, 24S-hydroxycholesterol (24-OHC), which is released into blood.
Objectives
We examined the effects on brain cholesterol turnover in healthy individuals and in multiple sclerosis (MS) following treatment with placebo or the remyelinating monoclonal antibody, rHIgM22, which binds to oligodendrocytes and myelin.
Methods
In vivo synthesis rates of brain cholesterol were measured by label incorporation and die-away of 24-OHC sampled from blood during and after heavy water (D2O) intake in age- and sex-matched non-MS and clinically stable relapsing-remitting MS subjects.
Results
Incorporation and die-away of labeled 24-OHC revealed biphasic kinetics, with two kinetically distinct pools of brain cholesterol: a large, slow turnover pool and a smaller, metabolically more active pool of newly synthesized cholesterol. The latter showed significantly higher turnover rates in MS compared to non-MS subjects, which was significantly reduced in patients with MS treated with rHIgM22.
Conclusions
Plasma 24-OHC kinetics provide a minimally invasive biomarker of brain cholesterol metabolism and revealed differences between healthy and clinically stable MS subjects, with increased turnover of the metabolically active 24-OHC pool that normalized in response to rHIgM22 therapy.
Introduction
Multiple sclerosis (MS) is the most common autoimmune disorder of the central nervous system and is characterized by demyelinating lesions with neurological deficits. 1 Myelin loss is an early pathological hallmark of MS. Although remyelination is the natural response to damage to the myelin sheath, this endogenous repair process is less than needed in human MS and results in progressive injury. Remyelinating therapies that target myelination, focusing on oligodendrocyte progenitor cells and mature oligodendrocytes, are in development. 2 A recombinant form of a human remyelination-promoting antibody (rHIgM22) increases myelin accrual in animal models and has undergone safety and tolerability studies in clinical trials.3,4
Cholesterol is a major myelin lipid that is synthesized entirely in situ in the brain. 5 Cholesterol synthesis is a rate-limiting stage of myelin formation. 6 Cholesterol synthesis in oligodendrocytes and neurons is required for remyelination following chronic demyelination.7,8 We have shown in animal models that synthesis of brain cholesterol, phospholipids, and myelin-associated proteins can be measured by metabolic labeling with heavy water (2H2O [D2O]) and that rates are decreased during experimental demyelination and increased during remyelination. 9 Importantly, these changes were observed by tracking in blood the labeling kinetics of the cholesterol metabolite, 24S-hydroxycholesterol (24-OHC), that is synthesized exclusively in the brain 10 and released into blood. Plasma 24-OHC kinetics correlated closely with brain cholesterol turnover. 9
Development of remyelinating therapies would benefit from a noninvasive biomarker to track myelin synthesis in vivo. In general, changes in rates of synthesis are more sensitive than changes in pool size. 11 We previously described a “virtual biopsy” technique for measuring labeling kinetics of molecules synthesized in inaccessible tissues, by sampling an accessible body fluid.12–20 Here, we applied the virtual biopsy approach to measure brain myelin turnover in vivo in healthy human subjects and patients with MS, based on labeling kinetics of plasma 24-OHC.
The metabolic mechanisms of proposed remyelinating therapies have not been testable in humans. How the remyelinating monoclonal antibody, rHIgM22, alters brain cholesterol turnover, for example, remains unknown. rHIgM22 binds to both oligodendrocytes and myelin.21,22 and might stimulate production of myelin or block binding of myelin-destructive antibodies to lipoprotein antigens and reduce myelin breakdown.
We used a pulse-chase heavy water administration protocol for a human physiological study comparing MS to healthy non-MS subjects and determining the effects of treating MS subjects with the remyelinating antibody rHIgM22. We report the first biomarker in humans of brain cholesterol synthesis and turnover. We observed significant differences between MS and non-MS subjects and a response to treatment with rHIgM22 that may be informative regarding its therapeutic mechanism of action.
Materials and methods
D2O labeling protocol, rHIgM22 treatment, and sample collection in humans
All human subjects provided written informed consent before participating in the study, which was approved by the institutional review boards for Acorda Therapeutics (trial registration identifier: NCT01803867, published elsewhere 3 ) or the Human Research Protection Office at the Washington University School of Medicine (protocol approval number: 201512086), and followed the guidelines set forth by the Declaration of Helsinki.
Deuterium labeling was evaluated following oral consumption of D2O (70%; Cambridge Isotope Laboratories) as described previously.13,14,23,24 Six healthy subjects consumed 50 mL of 70% D2O 4 times a day for 5 days followed by 2 daily doses of 50 mL of 70% D2O for 37 days. Between 100 and 250 days later, the same 6-week heavy water dosing protocol was repeated. Eighteen patients with stable multiple sclerosis drank 50 mL of 70% D2O twice a day for 2 weeks after which they received placebo or rHIgM22 treatment at 1 or 2 mg/kg (n = 6 per group) and then 2 weeks later again consumed 50 mL of 70% D2O twice a day for 2 weeks. The baseline characteristics of the subjects are shown in Table 1. Blood samples were collected weekly for the 6-week labeling period and for another 4 timepoints during the 200-day study. Plasma or serum were stored frozen. Blood samples were collected to characterize D2O enrichment time course and measure kinetics of 24-OHC.
Baseline demographics and clinical status of participants.

EDSS: Expanded Disability Status Scale; MS: multiple sclerosis; PP: primary progressive; PR: progressive relapsing; rHIgM22: monoclonal recombinant human antibody IgM22; RR: relapsing–remitting; SP: secondary progressive.
Human serum or plasma 24-OHC and cholesterol preparation
Serum or plasma samples were processed as described previously. 9 For analysis of 24-OHC, solid phase extraction was performed as described. 9 24-OHC was collected, dried under nitrogen and the silated samples were analyzed by gas chromatography–mass spectrometry (GC-MS). 9
GC-MS analysis
GC-MS analysis was performed on an Agilent Technologies GC6890N with 5973N mass detector. The GC was equipped with DB-5MS column (30 m × 0.25 mm × 0.25 µm). The mass spectrometer was operated in selected ion-monitoring mode, with selected ions for 24-OHC (m/z 413 and 414) corresponding to M0 & M1 mass isotopomers.
Calculation model for 24-OHC kinetics in human plasma
To quantify independent parameters synthesis (s) and dilution (d) rates for 24-OHC, we modified the model previously described
25
for determining birth and death rates and to capture kinetic heterogeneity of subpopulations of circulating cells in pulse-chase labeling experiments.26,27 Briefly, we modeled the measured total body water precursor pool enrichment, p(t), to characterize D2O exposure. Label enrichment L of the 24-OHC pool in blood was modeled as: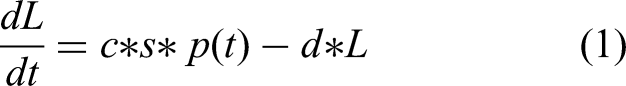
To determine p for equation (1), body water enrichment data were evaluated in a single compartment model. Total body water (TBW) is considered a single, well-mixed pool which turns over at rate kw, into which heavy water is administered in known amounts and intervals, I(t).
The differential equations for body water labeling (equation (2)) and 24-OHC labeling (equation (1)) were implemented in MS Excel, with single day timesteps. The model parameters (TBW, kw, s, and d) were determined by least squares fit to body water and 24-OHC enrichment data.
The two-pool model in humans is described in detail in Supplemental Methods.
Statistical analysis
Unpaired 2-tailed t-tests were used for comparing 24-OHC kinetics in MS versus non-MS subjects. One-way analysis of variance (ANOVA) with post hoc Tukey for pairwise multiple comparisons was used for comparing placebo and rHIgM22 effects in MS subjects. Differences between groups were considered statistically significant at p < 0.05.
Results
Blood 24-OHC label incorporation and die-away kinetics
In healthy non-MS volunteers (n = 6), the D2O protocol consisted of a labeling period of 5 weeks. In patients with relapsing–remitting MS treated with either placebo or rHIgM22 at doses of 1 mg/kg or 2 mg/g (n = 6 in each group), the D2O protocol comprised 2 periods of 2 weeks labeling separated by 2 weeks. Both protocols included a label die-away phase to day 200. These labeling and de-labeling protocols were analyzed by the kinetic incorporation/die-away model. The measured body water enrichment time course was used in the calculation model for incorporation (s), which corrects for any variability or differences in timing of label administration. The die-away curve (d) starts with the label in 24-OHC after labeling is completed, and is therefore not affected by the label administration protocol. Body water enrichments under each protocol are shown in Supplemental Figures 1 and 2 and achieved peak values generally of 1.0% to 2.0%.
The 24-OHC enrichment during the labeling and de-labeling periods in non-MS and MS subjects are shown (Figure 1(a)). The body water 2H enrichment curves and the plasma 24-OHC label incorporation and die-away curve of the individual non-MS and MS subjects are shown in Supplemental Figure 1. The best fit of plasma 24-OHC label incorporation and die-away curves was to 2-pools (Figures 1 and 2, Supplemental Figures 1 and 2 and Supplemental Table 1). The need for more than one pool is visually apparent and by consideration of the rate constant for label incorporation (s, or ks) and the rate constant for dilution (d, or kd). The values of d (kd) are higher than s (ks). A faster kd than ks is common in dynamic systems that functionally include a more rapidly turning over and a less rapidly turning over pool.25,27,29 Comparing s to d also reveals the relative sizes of the two pools. Vrisekoop et al. 27 presented a modeling approach for these systems which we built on and modified in our 24-OHC model. The goodness-of-fit for the model was strong, with R2 = 0.82 ± 0.17 in MS and 0.89 ± 0.07 in non-MS (Supplemental Table 1).
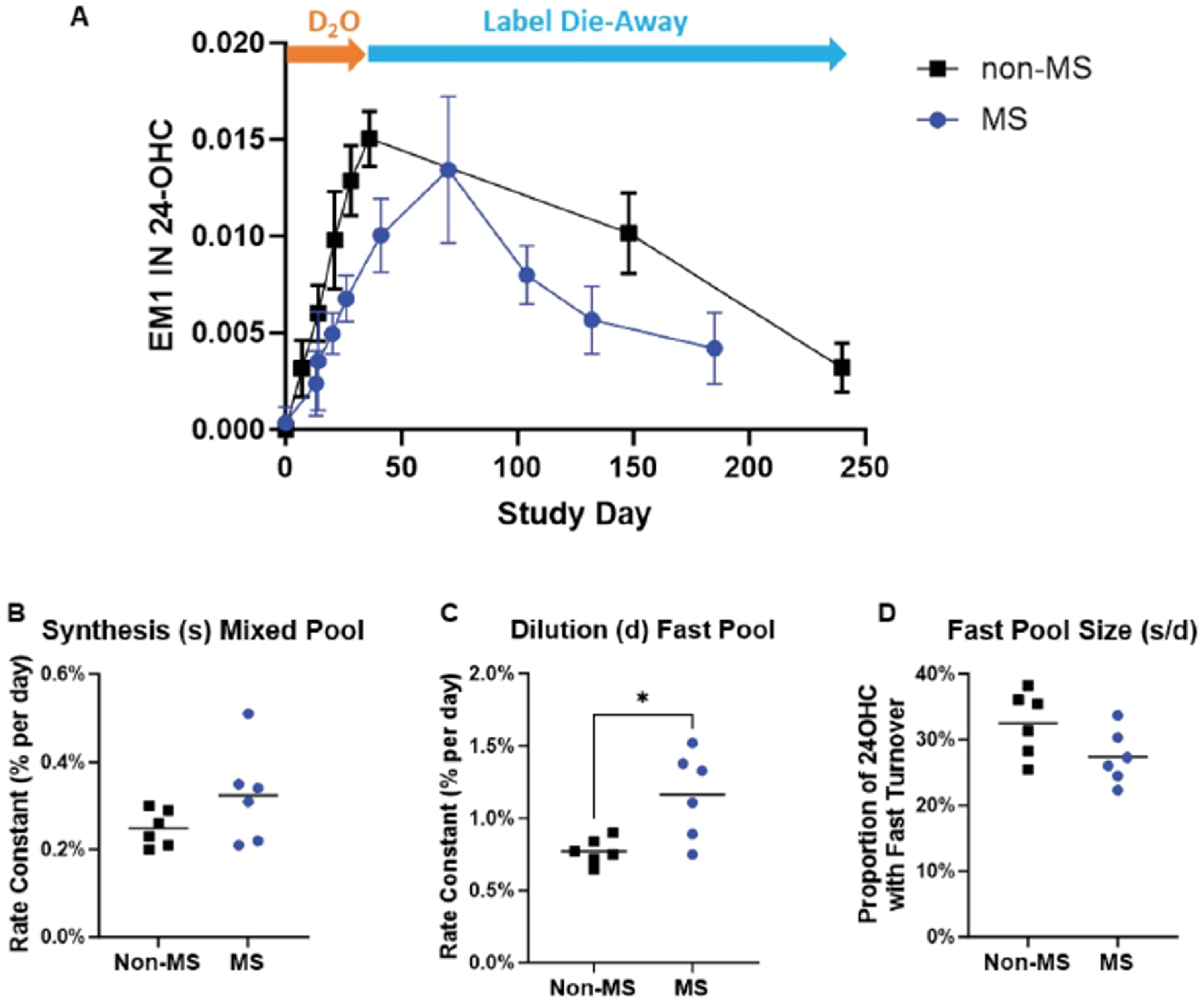
24-OHC kinetics in non-MS and MS human subjects. (a) 2H enrichment of 24-OHC in the blood of 6 non-MS and 6 MS human subjects during D2O incorporation and label die-away. (b) Synthesis rate (s) in mixed 24-OHC pool in non-MS and MS subjects. (c) Dilution rate (d) of 24-OHC in fast pool (higher turnover pool) in non-MS and MS subjects, *p < 0.05 compared to non-MS. (d) Calculated fast pool size of 24-OHC in non-MS and MS subjects. 24-OHC: 24S-hydroxycholesterol; D2O: duration of heavy water label; MS: multiple sclerosis.

24-OHC kinetics in patients with MS treated with placebo or rHIgM22. (a) 2H enrichment in 24-OHC in patients with MS during D2O incorporation and label die-away. (b) Synthesis rate (s) in mixed 24-OHC pool in MS subjects treated with placebo or rHIgM22 (1 or 2 mg/kg). (c) Dilution rate (d) of 24-OHC in fast pool (higher turnover pool) in MS subjects treated with placebo or rHIgM22 (1 or 2 mg/kg) (d) Calculated fast pool size of 24-OHC in MS subjects treated with placebo or rHIgM22 (1 or 2 mg/kg). *p < 0.05 compared to placebo. 24-OHC: 24S-hydroxycholesterol; D2O: duration of heavy water label; MS: multiple sclerosis; rHIgM22: monoclonal recombinant human antibody IgM22.
Similar results and good model fits were obtained for both labeling protocols (Figure 1(a), Supplemental Table 1). In MS subjects, the replacement rate, s, of cholesterol in the mixed 24-OHC pool, taken to represent replacement of the total pool of cholesterol in brain, was 0.35 ± 0.14%/day (Figure 1(b)), with a calculated mean t½ of total brain cholesterol of 198 days. For non-MS subjects, the replacement rate of the mixed 24-OHC pool was 0.25 ± 0.04%/day, reflecting t½ of 277 days for total brain cholesterol (p = 0.15 vs. MS subjects). In contrast, the dilution rate, d, of the metabolically active, more rapid turnover pool of 24-OHC was consistently and significantly higher in MS subjects on placebo (1.17 ± 0.39%/day, t½ = 59 days) compared to non-MS subjects (0.77 ± 0.09%/day, t½ = 90 days, p = 0.012, Figure 1(c) and Supplemental Table 1). This is taken to represent higher turnover rates of the metabolically more active pool of brain cholesterol in MS. The calculated fraction of 24-OHC in the metabolically active pool was not different between MS and non-MS subjects (29.3 ± 2.7% in MS vs 32.5 ± 5.0% in non-MS, p = 0.08, Figure 1(d)).
24-OHC kinetics were also measured in patients with MS administered rHIgM22 at 1 or 2 mg/kg and compared to placebo. 2H enrichments in 24-OHC during the labeling and die-away are shown (Figure 2(a)). The body water 2H enrichment curves and the plasma 24-OHC label incorporation and die-away curves of individual subjects treated with rHIgM22 are shown in Supplemental Figure 2. Treatment with rHIgM22 did not alter the replacement rate, s, of the total cholesterol pool (Figure 2(b)). However, there was a statistically significant (one-way ANOVA, main effect p = 0.03) reduction in the dilution rate of the metabolically active pool, d, by rHIgM22 (Figure 2(c)). A post hoc Tukey test was also performed for subjects treated with 1 mg/kg rHIgM22 (0.74 ± 0.25%/day, t½ = 94 days, post hoc Tukey p = 0.027 vs. placebo) or 2 mg/kg rHIgM22 (0.89 ± 0.20%/day, t½ = 78 days, post hoc Tukey p = 0.188 vs. placebo) with placebo values being 1.16 ± 0.30%/day, t½ = 60 days. The fraction of the metabolically active pool of 24-OHC was significantly (one-way ANOVA p = 0.04 across all three groups) increased by rHIgM22 (Figure 2(d)) treated with 1 mg/kg (32.6 ± 6.03%, post hoc Tukey p = 0.6086) or 2 mg/kg rHIgM22 (42.2 ± 14.4%, post hoc Tukey p = 0.0374) compared to placebo (27.4 ± 4.09%). The goodness-of-fit for the model was strong, with R2 = 0.85 ± 0.19 in the rHIgM22 1 mg/kg group and 0.87 ± 0.09 in the rHIgM22 2 mg/kg group (Supplemental Table 1).
Discussion
We evaluated plasma 24-OHC kinetics as a biomarker of brain cholesterol synthesis and turnover in humans. The data reveal a rapidly turning over pool of 24-OHC and therefore brain cholesterol in both MS and non-MS subjects, with a replacement rate (d) ∼3-times higher than the mixed pool rate (s) of 24-OHC. The turnover rate of the metabolically active pool was significantly higher in MS than in non-MS human subjects and was reduced by treatment with the remyelinating antibody rHIgM22 in MS subjects. The t½ of the mixed 24-OHC pool, representing brain total cholesterol, is ∼ 6 to 10 months with no significant differences between MS and non-MS subjects and no significant effect of rHIgM22 treatment.
Our study was designed, first, to measure incorporation and die-away kinetics of plasma 24-OHC in human subjects during and after metabolic labeling with heavy water and evaluate whether there is more than one kinetically distinct pool of cholesterol in human brain; second, to compare MS to non-MS humans; and, third, to determine whether treatment with rHIgM22 altered brain cholesterol kinetics in MS. We found strikingly different label die-away rates compared to label incorporation kinetics, which is characteristic of dynamic systems with faster turnover of newly made components.20,24,30 This has been called “last-in, first-out,” for pools like glycogen 20 or triglycerides, 24 or “activation-induced cell death” for T-lymphocytes. 30 We observed a considerably faster dilution rate d of labeled 24-OHC (representing replacement of recently synthesized, labeled cholesterol molecules by even more newly synthesized, unlabeled molecules) compared to label incorporation rates s into unlabeled 24-OHC (representing replacement of unlabeled cholesterol molecules by labeled molecules in mixed brain cholesterol). This difference allows calculation of a metabolically more active pool turnover rate (d) and a slower-pool turnover rate (s). The ratio of the two rates (s/d) reveals the proportion of the total pool in each compartment. 25
A discrepancy between s and d represents strong evidence for a kinetically heterogeneous population that feeds the circulating pool of 24-OHC, since there is no known mechanism for 24-OHC to have differential clearance in blood. The pool of 24-OHC that was newly synthesized was replaced more rapidly during the die-away period than the general 24-OHC pool. Although neurons are considered to be the major source of the 24-hydroxylase enzyme, which converts cholesterol to 24-OHC, 31 this enzyme has also been detected in oligodendrocytes and white matter axons in humans. 32 24-OHC has been measured in human brain white matter 33 and 24-OHC colocalizes with cholesterol in myelin. 34 Intercellular transfer of 24-OHC into active, myelin-depositing oligodendrocytes is consistent with cholesterol transfer into oligodendrocytes from other cells and cholesterol transfer among neurons and glial cells have been shown previously.6,35
Interestingly, the metabolically active pool replacement rate was significantly higher in MS than the non-MS subjects, even with a small sample size (n = 6 per group). Although the metabolically active pool of cholesterol in the adult brain has traditionally been thought to be primarily neuronal in origin, 31 oligodendrocyte cholesterol can become dynamic under conditions of demyelination and remyelination. We have previously shown in mice that 24-OHC kinetics in brain white matter and blood respond to cuprizone. 9 Cuprizone acts primarily on oligodendrocytes to cause demyelination through intrinsic and extrinsic cellular mechanisms, 36 but the cuprizone model can also show glial cell activation and changes in neuronal function. 36 It is not known whether 24-OHC kinetics represent more than just oligodendrocyte metabolism in humans. Moreover, the relevance of cuprizone treatment in mice to human demyelination remains unknown.
The different kinetics of the metabolically active pool of cholesterol in MS subjects compared to non-MS subjects may reflect a compensatory increase in remyelination rate of the active pool, replacing myelin breakdown. It is also conceivable that the faster turnover represents excess newly synthesized cholesterol during remyelination that has not been incorporated into mature myelin and is excreted as 24-OHC. 35 Of interest, studies administering neural stem cells into animals with neurological disease suggest that transplanted cells induce repair by increasing effectiveness of endogenous myelinating cells rather than by directly myelinating axons. 37 Failure of surviving oligodendrocytes to respond to tissue injury, revealed by turnover rates of the fast pool, may reflect an unsupportive micro-environment. While some of the mixed pool cholesterol synthesis (s) may reflect neuronal cholesterol dynamics, the parameter d is more plausibly taken to represent breakdown and replacement of newly synthesized and deposited myelin.
We also evaluated the effects of the remyelinating antibody rHIgM22, which binds to both oligodendrocytes and myelin.21,22 rHIgM22 reversed the higher turnover rate d in patients with MS. This may represent increased stability of newly formed myelin, consistent with rHIgM22 blocking the binding of autoantibodies to lipid antigens such as sulfatides 38 in myelin and thereby reducing myelin breakdown without broad immunosuppression. This finding represents a potential in vivo mechanism of action of rHIgM22.
It is worth comparing this approach to previously proposed biomarkers of myelination in humans. Plasma concentration of 24-OHC has been proposed as a marker of myelination.34,39 Concentrations of metabolites in blood, however, represent very different biology than flux rates.13–15 Metabolite concentrations in plasma are influenced by several factors including release rate from brain, distribution volume and clearance efficiency from blood. None of these are of fundamental interest to the physiology of myelination, whereas new synthesis rates represent brain myelin dynamics, as our preclinical data show. 9 Kanhai et al. 40 measured the turnover of myelin breakdown products β-galactosylceramide and N-octadecanoyl-sulfatide in cerebrospinal fluid (CSF) sampled 5 times by lumbar puncture during and after heavy water labeling. They reported fast (non-negligible) and negligible turnover pools, similar to our data. They did not show differences in turnover rates between MS and non-MS subjects, however, but a difference in the fraction of cholesterol in the non-negligible turnover pool. Their non-negligible turnover pools had more variable values for t½ (96–373 days) than our t½ values for blood 24-OHC (90 days in MS subjects). Their kinetic findings generally support the biological model that we present, although the need for repeated CSF samples is less convenient than blood sampling and unlikely to become a routine biomarker in humans.
Our data clearly show that clinically stable patients with relapsing–remitting MS exhibit subclinical abnormalities of myelin turnover and that these patients are not pathophysiologically quiescent, whether the higher turnover rate of the metabolically active pool represents compensatory remyelination or ineffective myelination. Indeed, recent studies of serum neurofilament light chain showed ongoing neuronal pathology in clinically stable MS 41 that may be consistent with our observations here.
Limitations of our study include control subjects being studied from a different cohort than the MS subjects, although matched for age and sex. The small clinical sample sizes will require confirmation in larger cohorts. The labeling protocols were slightly different for the healthy and MS subjects but, importantly, the difference in labeling protocols does not alter the kinetic results. The measured time-course of body water enrichment is taken into account in the calculation of incorporation (s), which corrects for any differences in the timing of label administration. In both labeling protocols (MS and non-MS subjects) the period of labeled body water exposure was 6 weeks followed by a die-away phase (Figure 1). The only difference in the two protocols is that during weeks 3 and 4, the MS subjects did not take heavy water, resulting in a fall then rise in body water enrichments. This was measured and accounted for in the calculation model. The die-away curve calculation (d) starts with the enrichment in 24-OHC after labeling is completed, so d is not affected by the label administration protocol.
In summary, label incorporation and die-away rates in plasma 24-OHC during and after heavy water pulse-chase revealed two kinetically distinct pools of brain cholesterol in human subjects with and without MS, paralleling preclinical results. Significant differences were observed in turnover rate of the more metabolically active pool in relapsing–remitting MS compared to non-MS controls, and these changes were reversed by treatment with the remyelinating antibody rHIgM22. The turnover of the metabolically active pool of brain cholesterol may be an informative flux biomarker for monitoring progression or treatment response of patients with MS.
Supplemental Material
sj-docx-1-mso-10.1177_20552173251344555 - Supplemental material for Metabolic labeling kinetics of brain-derived 24S-hydroxycholesterol in blood in multiple sclerosis: Effects of treatment with the remyelinating antibody rHIgM22
Supplemental material, sj-docx-1-mso-10.1177_20552173251344555 for Metabolic labeling kinetics of brain-derived 24S-hydroxycholesterol in blood in multiple sclerosis: Effects of treatment with the remyelinating antibody rHIgM22 by Mahalakshmi Shankaran, Kelvin W Li, Hussein A Mohammed, Joan Protasio, Mark Fitch, Marcy Matthews, Edna Nyangau, Gordon Smith, Samuel Klein, Andrew Eisen, Scott Turner and Marc K Hellerstein in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Supplemental Material
sj-pdf-2-mso-10.1177_20552173251344555 - Supplemental material for Metabolic labeling kinetics of brain-derived 24S-hydroxycholesterol in blood in multiple sclerosis: Effects of treatment with the remyelinating antibody rHIgM22
Supplemental material, sj-pdf-2-mso-10.1177_20552173251344555 for Metabolic labeling kinetics of brain-derived 24S-hydroxycholesterol in blood in multiple sclerosis: Effects of treatment with the remyelinating antibody rHIgM22 by Mahalakshmi Shankaran, Kelvin W Li, Hussein A Mohammed, Joan Protasio, Mark Fitch, Marcy Matthews, Edna Nyangau, Gordon Smith, Samuel Klein, Andrew Eisen, Scott Turner and Marc K Hellerstein in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Supplemental Material
sj-pdf-3-mso-10.1177_20552173251344555 - Supplemental material for Metabolic labeling kinetics of brain-derived 24S-hydroxycholesterol in blood in multiple sclerosis: Effects of treatment with the remyelinating antibody rHIgM22
Supplemental material, sj-pdf-3-mso-10.1177_20552173251344555 for Metabolic labeling kinetics of brain-derived 24S-hydroxycholesterol in blood in multiple sclerosis: Effects of treatment with the remyelinating antibody rHIgM22 by Mahalakshmi Shankaran, Kelvin W Li, Hussein A Mohammed, Joan Protasio, Mark Fitch, Marcy Matthews, Edna Nyangau, Gordon Smith, Samuel Klein, Andrew Eisen, Scott Turner and Marc K Hellerstein in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Supplemental Material
sj-docx-4-mso-10.1177_20552173251344555 - Supplemental material for Metabolic labeling kinetics of brain-derived 24S-hydroxycholesterol in blood in multiple sclerosis: Effects of treatment with the remyelinating antibody rHIgM22
Supplemental material, sj-docx-4-mso-10.1177_20552173251344555 for Metabolic labeling kinetics of brain-derived 24S-hydroxycholesterol in blood in multiple sclerosis: Effects of treatment with the remyelinating antibody rHIgM22 by Mahalakshmi Shankaran, Kelvin W Li, Hussein A Mohammed, Joan Protasio, Mark Fitch, Marcy Matthews, Edna Nyangau, Gordon Smith, Samuel Klein, Andrew Eisen, Scott Turner and Marc K Hellerstein in Multiple Sclerosis Journal – Experimental, Translational and Clinical
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for the study were provided in part by KineMed Inc, Acorda Therapeutics, UC Berkeley College of Natural Resources, NIH grants DK56341, DK20579, DK52574, RR024992, Academy of Nutrition and Dietetics Foundation and the Pershing Square Foundation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
