Abstract
Background
This study aimed to evaluate the impact of ellagic acid (EA) on fatigue, depression, and anxiety in patients with multiple sclerosis (MS) who have moderate disability.
Methods
A triple-blind, placebo-controlled clinical trial was conducted. Fifty-eight MS patients were randomly allocated to receive EA or placebo. Measurements of fatigue, depression, and anxiety were taken at the beginning and end of the study. Data analysis was performed via SPSS.
Results
Significant improvements were observed in the EA group across several measures: the State-Trait Anxiety Inventory (STAI), the Quick Inventory of Depressive Symptomatology (QIDS), the Hospital Anxiety and Depression Scale (HADS) for both depression and anxiety subscales, and the Modified Fatigue Impact Scale (MFIS), which includes total, cognition, psychosocial, and physical scores (P < 0.001). At the end of the study, significant differences between the EA and placebo groups were noted. Within the EA group, significant changes from baseline were found in EDSS, STAI (p = 0.003), QIDS (p = 0.041), HADS-D (p = 0.032), HADS-A (p = 0.012), total MFIS (p = 0.004), MFIS-Cognition (p = 0.001), MFIS-Psychosocial (p = 0.049), and MFIS-physical (p = 0.001) scores. In the EA group, significant changes from baseline were observed in EDSS, STAI (p = 0.003), QIDS (p = 0.041), HADS-D (p = 0.032), HADS-A (p = 0.012), total MFIS (p = 0.004), MFIS-Cognition (p = 0.001), MFIS-Psychosocial (p = 0.049), and MFIS-physical (p = 0.001) scores.
Conclusions
EA appears to significantly alleviate fatigue, depression, and anxiety in MS patients.
Introduction
Depression affects more than half of multiple sclerosis (MS) patients, significantly diminishing their quality of life. Prior studies have shown that more than 50% of individuals with multiple sclerosis (MS) suffer from depression.1,2 This mental health condition has a profound impact on overall well-being and quality of life. In MS patients, depression may arise as a reaction to their circumstances, be endogenous, or stem from the disease itself, and it can also lead to increased fatigue. 3 Furthermore, anxiety is a significant contributor to suicidal thoughts, impacting approximately 25% of those with MS. Anxiety is another prevalent issue among MS patients, affecting an estimated 19% to 45% of this population. It can worsen MS symptoms and is frequently regarded as a factor that heightens the likelihood of disease relapse.4,5 The presence of anxiety symptoms can worsen and lead to the recurrence of MS symptoms, thereby complicating patient recovery. 6
Fatigue is among the most common and debilitating symptoms in MS patients and is reported by more than 70% of individuals as a significant problem. This symptom is an important and influential factor for quality of life, social isolation and lack of employment in patients with MS. 7 Therefore, reducing and controlling anxiety, fatigue and depression in these patients is very important, and since MS medications have many side effects, the use of nondrug treatments to control these diseases and patients’ problems seems reasonable.
Currently, various methods are used to address and reduce depression, fatigue and anxiety. One of these solutions is the use of food supplements. Ellagic acid (EA) is a strong and polyphenolic antioxidant that is abundant in fruits and seeds, such as red raspberries, wild raspberries, strawberries, red grapes, pomegranates, almonds, and walnuts. On the basis of animal models and laboratory studies, EA has been identified as an effective substance for treating chronic diseases such as neurodegenerative diseases. 8 Vallarino and colleagues (2022) demonstrated that EA and pomegranate peel extract reduced clinical symptoms, microglial activity and astrogliosis, and improved spinal cord demyelination and inflammation in EAE mice. 9 In other study, Shen et al. (2021) reported that EA metabolites reduced inflammatory cells, demyelination, dendritic cell activity and Th17 cell differentiation in EAE and subsequently improved clinical symptoms. 10 Busto et al. (2018) showed that EA delays disease onset and reduces clinical symptoms in mice while protecting myelin-associated sphingolipids in EAE. 11 Also, Sanadgol et al. (2017) found that a high dose of EA (80 mg/kg body weight) significantly increased the population of mature oligodendrocytes and reduced apoptosis in cuprizone-induced demyelination. 12
In other study, Han et al. (2024) showed that EA halted disease progression and improved symptoms by modulating the microbiota-metabolite immunity axis in MS mice by altering gut bacteria, increasing short-chain fatty acid-producing bacteria and improving the secretion of inflammatory cytokines via histone deacetylase inhibition and acetylation. 13
Because many animal studies have investigated the efficacy of EA in animal models of MS and reported improvements in the clinical symptoms of the disease, but no such study has been conducted in MS patients, and because depression, anxiety and fatigue are important factors affecting the quality of life of individuals with MS, this study focused on the effects of pure EA on these factors.
Methods
Types of study and study population
This study was a multicenter, randomized, triple-blind, placebo-controlled clinical trial. The MS patient individuals were recruited from the neurology clinics at Rasoul Akram and Firoozgar hospitals, Tehran, Iran. The study protocol received approval from the Iran University of Medical Sciences ethics committee (ethics code IR.IUMS.REC.1399.1000) and was registered with the Iranian Registry of Clinical Trials (identifier: IRCT20120415009472N22, dated 19/12/2020).
Inclusion and exclusion criteria
The study involved 58 patients diagnosed with MS who gave their informed consent and expressed a willingness to take part in the study. The diagnosis was validated via McDonald's criteria and MRI, with an EDSS score ranging from 4.5 to 5.5. The participants included both males and females aged between 18 and 55 years, with a minimum of six months since diagnosis. The exclusion criteria involved participants who previously consumed nutritional or antioxidant supplements, except for vitamin D, had autoimmune or inflammatory diseases, had chronic illnesses, had substance or tobacco abuse, were pregnant, or were breastfeeding. Patients who experienced changes in the severity of the disease, medication dosage, diet, or physical activity or who consumed less than 90% of the prescribed supplements were removed from the study.
Sampling method and calculation of sample size
Considering the inclusion and exclusion criteria, convenience sampling or sampling based on availability was used in this study. The participants in the study were randomly assigned into two groups—those receiving EA and the placebo group—using balanced block randomization based on blocks of 4. The random allocation was performed using a sequence of 4-block randomization generated with the Blockrand statistical package in the R software, and it was conducted by the statistical consultant.
The supplements distributed between the two groups were completely similar in terms of packaging, color, and odor, and all supplement and placebo packages were labeled by an independent statistical consultant. The aforementioned statistical consultant had no involvement in data collection or analysis, and all researchers and the statistical consultant responsible for data analysis were unaware of the groups under study until the end of the study.
The sample size was 21 per group considering the previous studies 14 and the severity of the disease (EDSS), with Z1-α/2 equal to 1.96 and Z1-β equal to 1.28, S1 = 0.3, S2 = 0.3, and μ1-μ2 = 0.3, using the following formula: n = (Z1−α/2+Z1−β)2(S12+S22)/(μ₁-μ₂)². A total of 29 samples were determined per group to tailor possible sample attrition.
Implementation of the intervention
EA supplements, each containing 90 mg, were obtained from Axenix in Australia. The placebo capsules, which were indistinguishable in appearance and size from the EA capsules, consisted of maltodextrin. The participants were instructed to consume two capsules daily with their meals, resulting in a daily intake of 180 mg of EA. Adherence to the regimen was controlled through phone calls and text messages. The participants were advised to follow their usual dietary and physical activity patterns throughout the study and to declare any alterations in their medication dosages. Compliance was evaluated by counting the remaining capsules at the conclusion of the study.
Information gathering instruments
General information questionnaire
This questionnaire included questions about sex, age, occupation, contact information, period of illness, type and number of medications, food supplements, and history of disease.
24-h 3-day food recall questionnaire
Information on people's food intake was collected through a 24-h food recall questionnaire.
In both groups, a 24-h food recall questionnaire was completed at the beginning of the study and at the end of week 12 for 3 nonconsecutive days (one weekend day and two working days). The listed amounts of each food were subsequently converted to grams via the home scale guide and analyzed with Nutritionist 4 software.
International physical activity questionnaire (IPAQ)
The IPAQ comprises seven concise questions that inquire about the duration and frequency of heavy or moderate physical activities, as well as walking, sitting, and duration of physical activities beyond daily routines over the past week. The participants completed this questionnaire both before and after the intervention.
Anthropometric measurements
Anthropometric data, such as height (measured to 1 mm accuracy) and weight (measured to 100 grams accuracy), were collected. BMI was calculated as weight (kg) divided by height squared (m²).
Measurement of anxiety, depression and fatigue
Anxiety was quantified through the application of the Spielberger State-Trait Anxiety Inventory (STAI). Spielberger's State Anxiety Questionnaire consists of 40 questions, the first 20 of which measure the state of anxiety, and the second 20 of which measure the trait of anxiety. The Quick Inventory of Depressive Symptomatology (QIDS) questionnaire, which has 16 items and a range of scores from 0 to 27, was used to measure depression. The HADS comprises 14 items and is divided into two parts. The HADS-A was used to assess anxiety, and the HADS-D was used to assess depression. A questionnaire (MFIS) was used to evaluate fatigue. The MFIS questionnaire consists of 20 items and 5 subscales, namely, physical fatigue (range 0–36), psychosocial fatigue (range 0–8), cognitive fatigue (range 0–40) and total fatigue (range 0–84).
Statistical analysis
Data analysis was performed via SPSS version 24. Descriptive statistics were used to describe the samples. The normality of data distribution was checked using Kolmogorov‒Smirnov test and all variables showed a normal distribution, so parametric statistical tests were used for data analysis. Independent t tests were used to compare quantitative variables at the beginning of the study and their changes over time. Paired t tests were used to compare within-group changes before and after the intervention and linear regression was used to adjust for confounding factors, p-values of less than 0.05 were considered statistically significant.
Results
Patient ådemographic information
Among the 192 patients with multiple sclerosis (MS) who were referred between January 2019 and September 2020 to the neurology clinics of the Rasoul Akram and Firoozgar Hospitals, 58 patients were eligible to participate in the study. Regarding the changes in disease progression and medications, 4 patients (3 women) from the EA group and 4 patients (4 women) from the placebo group were included in the study. The adherence rates to the study were 92.23% for the EA group and 92.42% for the placebo group. Patient demographics, such as age, BMI, and disease duration, were recorded to evaluate their potential influence on the outcomes. With the exception of age and disease duration, there is no difference between two groups regarding all other baseline variables (Table 1, Figure 1).

Flow diagram of participant recruitment based on the consolidated standards of reporting trials (CONSORT) statement.
Baseline characteristics of participants in the ellagic acid and control groups.

The data are presented as the mean ± SD or number (%).
BMI: body mass index; EDSS: Expanded Disability Status Scale.
Chi-square test.
P value < 0.05 was considered to indicate statistical significance.
Patient food intake
Study participants were appraised regarding their dietary information. No significant disparity in food intake was found between the groups, as revealed by the independent t test. Furthermore, an analysis of intragroup variations revealed no notable differences within any of the groups throughout the duration of the study (Table 2).
Dietary intake of participants in the ellagic acid and control groups.

The data are presented as the mean ± SD.
Independent sample t test.
Paired sample t test.
P value < 0.05 was considered to indicate statistical significance.
Disease severity, fatigue, depression and anxiety
A comparison between groups at the end of the study revealed that there was a significant difference between the two groups in terms of EDSS (1.01 [0.358, 1.779], p = 0.001; STAI (−5.09 [-7.711, −2.475], p = 0.001; QIDS (−2.68 [-3.195, −2.177], p = 0.001); HADS-D (−2.45 [-3.225, −1.692], p = 0.001); HADS-A (−15.75 [-22.29, −9.20], p = 0.001); MFIS-total (−12.51 [-13.895, −11.131], p = 0.001; MFIS-Cognition (−4.72 [-5.886, −3.573], p = 0.001); and MFIS-Psychosocial (−1.91 [-3.326, −0.493], p = 0.001]; and MFIS-physical (−6.60 [-8.712, −4.494], p = 0.001) scores at the end of the intervention. No notable intragroup differences were observed at the beginning of the study. However, significant changes were noted in the EA group compared with the control group from the beginning of the study. The EDSS scores showed a change of −1.06 ± 0.09 compared with 0.02 ± 0.02 (p = 0.001), the STAI score (−5.03 ± 0.01 vs. −0.03 ± 0.05, p = 0.003), the QIDS score (−2.84 ± 08 vs. 0.02 ± 0.01, p = 0.041), the HADS-D score (−2.55 ± 0.08 vs. −0.13 ± 0.06, p = 0.032), the HADS-A score (−3.65 ± 0.04 vs. 0.02 ± 0.05, p = 0.012), the MFIS-total score (−12.26 ± 0.14 vs. −0.17 ± 0.07, p = 0.004), the MFIS-Cognition score (−13.99 ± 0.32 vs. 0.01 ± 0.02, p = 0.001), the MFIS-Psychosocial score (−0.82 ± 0.01 vs. −0.04 ± 0.02, p = 0.049), the MFIS-physical score (−6.94 ± 0. No significant change was observed in the placebo group (Table 3).
EDSS, STAI, QIDS, HADS-d, HADS-a, total MFIS, total MFIS-cognition, and total MFIS-psychosocial and MFIS-physical status in the ellagic acid and control groups.
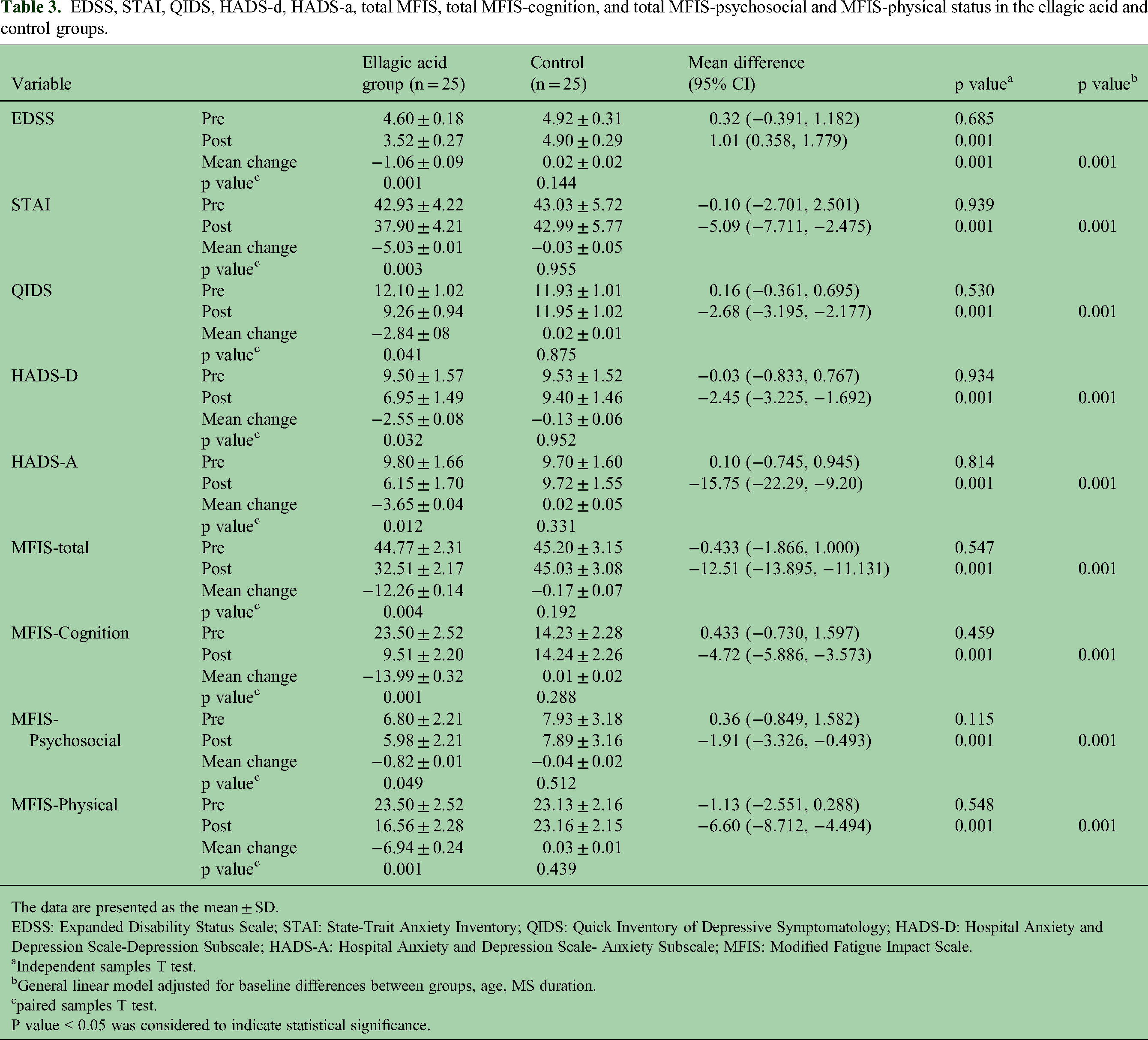
The data are presented as the mean ± SD.
EDSS: Expanded Disability Status Scale; STAI: State-Trait Anxiety Inventory; QIDS: Quick Inventory of Depressive Symptomatology; HADS-D: Hospital Anxiety and Depression Scale-Depression Subscale; HADS-A: Hospital Anxiety and Depression Scale- Anxiety Subscale; MFIS: Modified Fatigue Impact Scale.
Independent samples T test.
General linear model adjusted for baseline differences between groups, age, MS duration.
paired samples T test.
P value < 0.05 was considered to indicate statistical significance.
Discussion
In the present study, we investigated the effect of a three-month intake of EA on MS patients with anxiety, fatigue and depressive symptoms in a randomized clinical trial.
We found a statistically significant reduction in HADS-A, MFIS and STAT-Y scores compared to the control group. In terms of clinical significance, the minimum clinically important difference (MCID) for the disability severity variable in MS patients with an EDSS score of less than 5.5 is a one-unit reduction 15 ; in the present study, a 1.06 reduction in EDSS score was observed in patients receiving EA. The MCID for the STAI variable is 10%, 16 and in our study a decrease in STAI score of more than 10% was observed. The MCID for the HADS variable is 1.4–4.4. 17 In the present study, a decrease of 2.55 points in the HADS-D variable and a decrease of 3.65 points in the HADS-A variable was observed in the group receiving EA. The MCID for the MFIS variable is also between 3.86and 8.11, 18 whereby in the present study a decrease in the MFIS score of 12.26 points was observed after supplementation with EA. Therefore, the changes achieved are also clinically significant.
The efficacy of EA has been demonstrated in previous studies. Lu et al. (2020) showed in their study that pomegranate peel extract at a dose of 100 mg/kg body weight (each gram of extract contains 97.5 mg EA) for sixteen days improved disease severity in animal model of MS, and this effect was achieved by modulating the gut microbiota. 19 In another study, Petrou et al. (2021) showed that a nano-formulation of pomegranate seed oil improved cognitive function in MS patients over three months. 20 In another study, Kiasalari et al. (2021) achieved a significant reduction in IL-17 levels after supplementation with 10 or 50 mg/kg body weight of EA for twelve days in an animal model of MS. 21 Also, in an animal model of MS and type 1 diabetes, pomegranate peel extract improved disease symptoms, which was due to inhibition of immune cell infiltration into pancreatic islet cells and reduction in the production of inflammatory cytokines involved in the pathogenesis of MS, including IL-17 and interferon-gamma (IFNγ). 22 In another study, EA at a dose of 4 mg/kg body weight for ten days caused preventive and therapeutic effects in an animal model of MS, which were achieved by inhibiting the infiltration of peripheral inflammatory cells into the CNS by reducing CD4 + IL-17 and CD4 + IFNγ cells. 23
As for the mechanism of the antidepressant, anti-anxiety and anti-fatigue effects of EA, several pathways are involved. The first pathway is the effect of EA on the regulation of inflammation and the inhibition of microglial activation. In depression, microglia is activated and cause the production of pro-inflammatory cytokines, including IL-17. EA inhibits nuclear factor kappa B (NF-κB) and mitogen-activated protein kinase (MAPK) signaling, reduces the production of inflammatory cytokines, including IL-17, and inhibits microglial activation, and by improving neuronal function, it has antidepressant, anti-anxiety, and anti-fatigue effects.10,12 By reducing inflammation and inflammatory factors, it also increases the activity of regulatory T cells and improves the function of the hypothalamic-pituitary-adrenal axis, improving psychological problems and fatigue in MS patients. 24 In humans, the effects of IL-17 on the demyelination process of nerve cells in MS patients are known to cause the onset of psychological consequences of the disease, including depression, anxiety and fatigue. In addition, the worsening of patients’ condition is associated with an increase in the number of Th17 cells in patients’ blood. 25
Th17 cells are reportedly the first cells to come into contact with myelin antigens presented by antigen-presenting cells (APCs) in the subcranial space. Upon antigen recognition, Th17 cells release several proinflammatory mediators such as IL-17A and create an inflammatory environment that can cause tissue damage in the CNS. 26 IL-17A causes the production of reactive oxygen species (ROS) in the endothelial cells of the blood-brain barrier, leading to activation of the contractile mechanism and rupture of the intercellular junctions, resulting in the breakdown of the blood-brain barrier, which is a very important early event in the pathogenesis of MS and is associated with depression and fatigue from MS. 26 EA inhibits the production of reactive oxygen species through its antioxidant effect and prevents damage to membrane lipids by reducing oxidative stress and maintains the integrity of the neuronal membrane. EA also inhibits neuronal apoptosis and, by preventing neuronal death, improves the psychological state of patients and reduces fatigue caused by MS. 27
Furthermore, by stimulating the production of matrix metalloproteinase (MMP) enzymes, IL-17A causes the destruction of tight junction proteins and increases the permeability of the blood-brain barrier, which increases the amount of inflammatory cytokines in the CNS environment and causes tissue and neuronal damage. By lowering IL-17 levels, EA reduces inflammation and maintains neuronal integrity, and neurochemical changes associated with depression, anxiety and fatigue are improved. By reducing inflammation, EA also facilitates the process of remyelination, removing barriers to the maturation of oligodendrocytes and increasing their survival. 28
According to the available studies, EA can reduce inflammation and oxidative stress in MS patients. However, the available studies have investigated short- and medium-term effects, and further research is needed to determine the long-term effects.
This research has several strengths. To date, our study is the first to investigate the effects of EA, an important component of pomegranate, on psychological impairments such as anxiety, fatigue and depression in MS patients. In addition, the HADS-A, MFSIS and STAT-Y2 questionnaires are the most accurate assessment methods for evaluating pathological symptoms in MS patients. The strengths of this study include our analyses that took into account patients’ dietary intake, physical activity and baseline measurements. The assessment of confounding factors such as food intake and physical activity is strength of our study. It is important to be aware of the limitations of our study. These include the administration of a single dose of EA, financial limitations in the assessment of inflammatory factors and gene expression of these factors. Further studies in humans are needed to facilitate the clinical application of EA supplements.
Conclusion
Notably, this research is unique in that it is the first to discuss the prominent effect of pure EA on MS patients with psychological impairments. EA supplementation led to a marked decline in psychiatric issues, including fatigue, anxiety, and depression, in patients with MS. Compared with those in the control group, we demonstrated prominent improvements in HADS-A, MFSIS, and STAT-Y2 scores. Although additional investigations are needed to determine how to improve psychological symptoms in MS patients, mechanisms such as the anti-inflammatory properties of EA can be considered. Additionally, EA has a main effect on modulating immune system activity by increasing Th2/Treg function and decreasing Th1/Th17 function, and these increases have been proposed to have beneficial effects on psychological symptoms in MS patients.
Footnotes
Author contributions
S.J.K: Developed the hypothesis, drafted the manuscript, conducted laboratory experiments, and handled sampling and data analysis. N.A, A.D, B.H.A: Provided supervision and manuscript editing. Gh.H, F.SH: Reviewed and edited the manuscript. M.M: Performed native editing of the manuscript and contributed to conceptualization. Sh.A, Sh.Kh, M.K and M.A: Conducted sampling. A.J.K: Analyzed the data. A.D: Conducted laboratory experiments. A.M: Supplements coding, supervision and manuscript editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Iran University of Medical Sciences (project number: 17787).
Iran University of Medical Sciences, (grant number 17787).
Ethics statement
The study received approval from the Ethics Committee of Iran University of Medical Sciences (Ethics code: IR.IUMS.REC.1399.1000) and was registered on the Iranian Registry of Clinical Trials (identifier: IRCT20120415009472N22, dated 19/12/2020). All participants provided written informed consent.
Data availability
Data can be made available upon reasonable request from the corresponding author.
