Abstract
Background
Multiple sclerosis (MS) involves symptoms that may be impacted by angiogenesis. Vascular endothelial growth factor (VEGF), a potent pro-angiogenic molecule, is elevated in early MS, but its activity in later disease is understudied. [Met5]-enkephalin (ENK) has anti-angiogenic activity and is decreased in persons with MS (PwMS).
Objectives
To determine salient symptoms of MS and evaluate relationships between common MS symptoms and angiogenesis-associated biomarkers.
Methods
PwMS and non-MS control participants were identified for this cross-sectional study. Walking times and self-reported fatigue, anxiety, depression, and pain were measured. Serum VEGF and [Met5] ENK levels were measured in a subset of PwMS.
Results
PwMS (n = 66) had significantly greater fatigue than controls (n = 35). In PwMS, fatigue, anxiety, depression, and pain were positively intercorrelated; fatigue was positively correlated with slower walking. Serum ENK and VEGF had a trending negative relationship. Serum ENK, but not VEGF, had a trending negative relationship with the length of disease. Serum ENK and VEGF were not correlated to walking time or self-report measures.
Conclusion
Fatigue is a salient MS symptom when compared to non-MS controls. Imbalanced pro- and anti-angiogenic signaling may influence fatigue in established MS, but further studies with larger sample sizes are needed to elucidate this potential relationship.
Keywords
Introduction
Multiple sclerosis (MS) is a chronic, immune-mediated neurological disease. MS afflicts approximately 1 million individuals in the United States, with women being diagnosed more than men at a ratio of 3:1 worldwide. 1 Common MS symptoms like fatigue, anxiety, depression, pain, and physical disability reduce the quality of life in persons with MS (PwMS).
Over 80% of PwMS experience fatigue and approximately half of PwMS report fatigue as their most disabling symptom. 2 MS-related fatigue involves anatomical, chemical, electrophysiological, and inflammatory substrates. Magnetic resonance imaging (MRI) data suggest that white and gray matter lesion load may result in globally impaired brain function and metacognition-induced experiences of fatigue. 3 Since inflammation is upstream of numerous hypothesized fatigue-related processes, 2 we focus the present study on angiogenesis, a mechanism that facilitates central nervous system (CNS) inflammation.
In PwMS, increased anxiety and depression have been linked to MRI findings of brain inflammation and increased cerebrospinal fluid (CSF) pleocytosis. 4 The most common types of pain associated with MS are neuropathic pain, muscle spasms, and headache, with approximately 50% prevalence in PwMS. 5 Inflammation may contribute to pain in MS through the sensitization of sensory neurons and axonal injury. 6 Disability progression in MS is also associated with elevated immune, inflammatory, and oxidative stress signaling molecules. 7 Together these findings indicate a role for central inflammation in the symptomatology of MS.
In MS, inflammatory cytokines and antibodies secreted by immune cells target myelin, leading to demyelination and axonal injury. 8 Immune cell infiltration relies on blood–brain barrier (BBB) leakiness and vascular surface area. Angiogenesis, the formation of new blood vessels from existing vasculature, promotes MS inflammatory activity by increasing cellular infiltration across the BBB. 9 Angiogenesis is associated with onset and relapse in MS, and imbalances in angiogenic molecules (e.g. vascular endothelial growth factor; VEGF) have been implicated in chronic inflammation and neurodegeneration in MS.10–12 More research is warranted to determine how angiogenesis might influence the severity of symptoms in established disease.
A potential regulator of angiogenesis is [Met5]-enkephalin (ENK), also termed opioid growth factor (OGF). ENK is an endogenous opioid that has inhibitory growth properties when bound to its receptor, OGFr. 13 Administration of exogenous ENK inhibited angiogenesis in a chick chorioallantoic membrane (CAM) model, and blockade of OGFr increased angiogenesis in CAM and in cutaneous wounds in diabetic rats.14,15 Greater circulating ENK levels are correlated with improved sensorimotor function in the mouse model of MS, experimental autoimmune encephalomyelitis (EAE). 16 Treatment with ENK improves behavioral outcomes, rescues spinal cord myelination, and delays the onset of EAE.16–18 Serum ENK levels are decreased in PwMS compared to non-MS controls. 19
In this cross-sectional study, we measured the prevalence and intensity of fatigue, anxiety, depression, and pain in PwMS compared to non-MS controls. To begin to determine the potential impacts of angiogenic signaling on symptoms of MS, we investigated the utility of serum ENK and VEGF as biomarkers for alterations in walking ability, fatigue, anxiety, depression, and pain.
Methods
Study design and participants
The clinical study 9784 was approved by the Institutional Review Board at Pennsylvania State University. Participants were recruited from the Penn State Neurology Clinic from April 2023 to September 2024. Female and male patients aged 18–76 years old with a confirmed MS diagnosis meeting the 2017 McDonald criteria were interviewed to obtain informed consent at the time of their scheduled neurology appointment. 20 Female and male control participants, also aged 18–76 years old, were guests of MS patients or patients of the Neurology Clinic undergoing unrelated procedures. Potential control participants with a history of autoimmune disease and any participants with cognitive deficits affecting informed consent were excluded.
MS participants optionally provide a blood sample and medical record access for their most recent timed 25-foot walk (T25-FW). Confidentiality was maintained by assigning de-identified study participant numbers used for data entry, storage, and analysis. A sample size power analysis for significant differences in Patient-Reported Outcomes Measurement Information System (PROMIS)-FatigueMS scores between MS and control participants was performed to determine a minimum sample size of N = 29 MS participants with an effect size of Cohen's d = 0.75, based on a study that found the mean PROMIS-FatigueMS T-score for PwMS in the US to be 57.7 ± 10.5 and the mean T-score for the general US population to be 50.21,22 An additional sample size power analysis was performed to determine a minimum sample size of N = 19 for serum biomarker correlations with an effect size of Pearson's r = 0.6 based on previous work from our laboratory.
Demographics and characteristics
Recruited participants completed surveys on demographics, fatigue, anxiety, and depression. Relevant demographics and characteristics for control participants included age, sex, pain, and current use of medications to treat fatigue, anxiety, depression, or pain. Pain was self-reported by participants using the numeric pain scale from 0 to 10. 23 Relevant demographics for MS participants included those for the controls, in addition to MS characteristics: Length of disease (LOD), type of MS, and use and type of disease-modifying therapy (DMT).
Fatigue
The PROMIS-FatigueMS survey is an eight-item short-form survey that assesses a range of symptoms, from mild tiredness to debilitating, chronic exhaustion.
22
The PROMIS-FatigueMS has excellent internal consistency (Chronbach's alpha > 0.96), strong test-retest reliability (intraclass correlation coefficient
Anxiety
The PROMIS-Anxiety survey is a seven-item short-form survey that assesses the severity of anxiety and associated symptoms like nervousness and tenseness. 24 The PROMIS-Anxiety has demonstrated higher internal consistency (Chronbach's alpha = 0.95) than other anxiety measures in MS. 25 The PROMIS-Anxiety demonstrated better sensitivity (0.73) and comparable specificity (0.83) to these measures at published cut-point scores. Each survey item is scored 1–5 on a Likert scale, with total raw scores ranging from 7 to 35 for completed surveys. Higher scores equal a greater experience of anxiety.
Depression
The PROMIS-Depression survey is an eight-item short-form survey that assesses characteristics of depression, such as feelings of helplessness and worthlessness. 26 The PROMIS-Depression has demonstrated higher internal consistency (Chronbach's alpha = 0.95) than other depression measures in MS. 25 The PROMIS-Depression demonstrated comparable sensitivity (0.69) and specificity (0.85) to these measures at published cut-point scores. Each survey item is scored 1–5 on a Likert scale, with total raw scores ranging from 8 to 40 for completed surveys. Higher scores equal a greater experience of depression.
The Multiple Sclerosis-Specific Beck Depression Inventory (MS-BDI) is a 12-item survey that assesses symptoms of depression that are determinant for depression in individuals with MS. 27 The MS-BDI has comparable sensitivity (0.73) and specificity (0.96) to other depression measures. 27 Each survey item is scored 0–3 on a Likert scale, with total raw scores ranging from 0 to 36 for completed surveys. Higher scores equal a greater experience of depression. The MS-BDI was used as an ancillary measure to discriminate between depression-mimicking MS symptoms and actual symptoms of depression in MS participants.
Walking ability
T25-FW data was obtained from the medical record of MS participants who provided consent and was recorded using de-identified study participant numbers. The T25-FW test measures walking ability in ambulatory MS patients. 28 The test involves the patient walking a straight 25-foot course as quickly as possible. The walking time is measured for two trials and averaged for a final score. Greater walking times indicate greater ambulatory dysfunction. In this study, MS participants who were non-ambulatory due to MS-related disability were omitted from analysis.
Sample collection and analysis
Participants who consented to blood collection had their blood drawn by hospital laboratory personnel. Samples were kept at room temperature for 1–3 h and kept at 4 °C until retrieved by a research team member and centrifuged at 2500 r/min for 10 min at 4 °C. Sera were aliquoted and kept for long-term storage at −80 °C until used for enzyme-linked immunosorbent assay (ELISA). Sera were assayed using commercially produced sandwich ELISA kits for VEGF (#ELH-VEGF, RayBiotech, Peachtree Corners, GA) and ENK (#MBS269873, MyBioSource, San Diego, CA) according to manufacturer recommendations. Standards and samples were run in duplicate within-assay, and each assay was performed twice. Duplicate and repeated concentration values were averaged before data analyses.
Statistical analysis
Data were analyzed using GraphPad Prism version 10.2 for MacOS (Boston, MA). Normality of continuous variables was assessed visually with histograms and Shapiro-Wilk tests. For pairwise comparisons of parametric data, two-tailed t-tests were performed. Mann-Whitney U-tests were used for pairwise comparisons of nonparametric data. Two-tailed two-sample independent proportions tests were used for cohort proportion differences. Spearman correlation coefficient (rho) was computed for relationships between variables. To reduce bias, potential covariates were assessed for significant relationships that would necessitate stratification before further analysis. For all tests, statistical significance was determined at P < 0.05.
Results
MS and control cohort demographics and characteristics
Of 141 participants who provided informed consent, 62 completed and returned surveys at the time of the interview, 40 mailed back completed surveys, and 39 did not return surveys. The response rate for participants who mailed surveys back was 50.6%. The MS cohort was composed of 51 female and 15 male participants with a mean age (±SD) of 53 ± 11.6 years for combined sexes (Table 1). The Control cohort was composed of 22 female and 13 male participants with a mean age of 51 ± 13.7 years. There was no significant difference in the mean age or proportion of female participants between the MS and control cohorts.
MS and control cohort demographics and characteristics.

Values represent means ± SD. P values represent comparisons with non-MS controls. MS: multiple sclerosis; RRMS: relapsing-remitting MS; PPMS: primary progressive MS; SPMS: secondary progressive MS; DMT: disease-modifying therapy.
The mean LOD for the MS cohort was 16.5 ± 9.5 years with a range of 1–42 years. Of the 66 MS participants, 59 had a diagnosis of relapsing-remitting MS (RRMS), 5 had secondary progressive MS (SPMS), and 2 had primary progressive MS (PPMS). DMT information was provided by 57 MS participants. Twelve participants reported taking infusion therapies (8 ocrelizumab, 4 natalizumab), 13 reported taking an injectable drug (8 glatiramer acetate, 3 interferon beta-1a, 1 peginterferon beta-1a, 1 interferon beta-1b), 21 reported taking oral medication (8 teriflunomide, 10 dimethyl fumarate, 2 cladribine, 1 fingolimod), and 11 reported taking no DMT.
A subset of the MS cohort provided blood samples for serum VEGF and ENK analyses (Table 2). Of the 23 MS participants who provided blood samples, 3 did not return surveys and were excluded from analyses. The final subset included 15 female and 5 male participants, with a mean age of 52 ± 12.3 years. All 20 participants had RRMS and the mean LOD was 14.5 ± 8.1 years. The mean age of the MS cohort subset (52 ± 12.3) was no different from the mean age of the control cohort (51 ± 13.7; P = 0.63). The proportion of the MS subset that was female (75%) was no different from that of the control cohort (62.9%; P = 0.36).
MS cohort biomarker subset demographics and characteristics.

Values represent means ± SD. MS: multiple sclerosis; RRMS: relapsing-remitting MS; DMT: disease-modifying therapy.
Self-report measures for MS and non-MS control participants
Incomplete (partially filled) surveys were excluded from analysis, resulting in different N values for the MS cohort: PROMIS-FatigueMS (N = 64), PROMIS-Anxiety (N = 65), PROMIS-Depression (N = 65), MS-BDI (N = 62). The 20 MS cohort subset participants completed the surveys in full except for the PROMIS-FatigueMS and PROMIS-Anxiety (both N = 19). All 35 control participants completed each survey in full.
The proportion of all MS participants taking medications for fatigue (24%) was significantly greater than controls (3%) at P = 0.007 (Table 3). The proportion of all MS and control participants taking medications for anxiety, depression, or pain were not significantly different. The mean PROMIS-FatigueMS score for the MS cohort (22.1 ± 7) was significantly greater than the Control cohort (17.7 ± 7.7) at P = 0.003. Mean PROMIS-Anxiety and PROMIS-Depression scores were not different between the MS and Control cohorts. There was an insignificant difference in MS-BDI scores for MS (6.1 ± 5.6) vs. Controls (4.7 ± 5.1, P = 0.23). The proportion of both cohorts exceeding the clinically relevant cut-point score of 8 was not different between the MS (17%) and Control cohorts (20%; P = 0.76).
All MS, MS biomarker subset, and control cohorts self-reported fatigue, anxiety, depression, and pain.

Values represent means ± SD. P values represent comparisons with non-MS controls. MS-BDI: Multiple Sclerosis-Specific Beck Depression Inventory; PROMIS: Patient-Reported Outcomes Measurement Information System.
The proportion of MS participants with high fatigue (PROMIS-FatigueMS raw score
Correlation of self-report measures and walking ability in MS participants
Seven participants had missing T25-FW scores due to non-MS reasons and four participants had missing scores due to MS-related pathology, so walking time data was obtained for 55 MS participants. Correlation analyses for walking times and survey scores for fatigue (N = 53), anxiety (N = 54), depression (N = 55), and pain (N = 46) showed that fatigue, anxiety, and depression had very strong positive intercorrelations (Figure 1). Pain had strong positive correlations to fatigue, anxiety, and depression. Walking times were weakly positively correlated to fatigue and were not significantly correlated to anxiety, depression, or pain.

Relationships between fatigue, anxiety, depression, pain, and walking time in MS participants. Fatigue, anxiety, depression, and pain were strongly to very strongly intercorrelated, and fatigue was weakly correlated to slower walking times. Numbers in boxes denote Spearman rho; red color and positive values denote a positive correlation; blue color and negative values denote a negative correlation. P < 0.05 (*), P < 0.01 (**), P < 0.001 (***).
Correlation of serum VEGF and ENK in MS cohort subset
ELISA revealed a mean serum VEGF concentration of 244.7 ± 198 pg/mL with a range of 10.3–738.6 pg/mL and a mean serum ENK concentration of 87.5 ± 47.1 pg/mL with a range of 2.9–179.4 pg/mL in the MS cohort subset. Five samples had nondetectable ENK values. Serum VEGF and ENK showed a strong but statistically insignificant negative association (Spearman rho = −0.48, P = 0.07) (Figure 2). There was no significant difference in mean VEGF or ENK values between females (N = 15) and males (N = 5); therefore, males and females were not separated for further analyses. There were no significant correlations between VEGF and age or LOD. ENK values were not significantly correlated to age or LOD, but there was strong negative association between ENK and LOD (Spearman rho = −0.5, P = 0.06) (Figure 3). Due to low N values for specific DMTs, groupwise comparisons based on DMT were not performed in the subset.
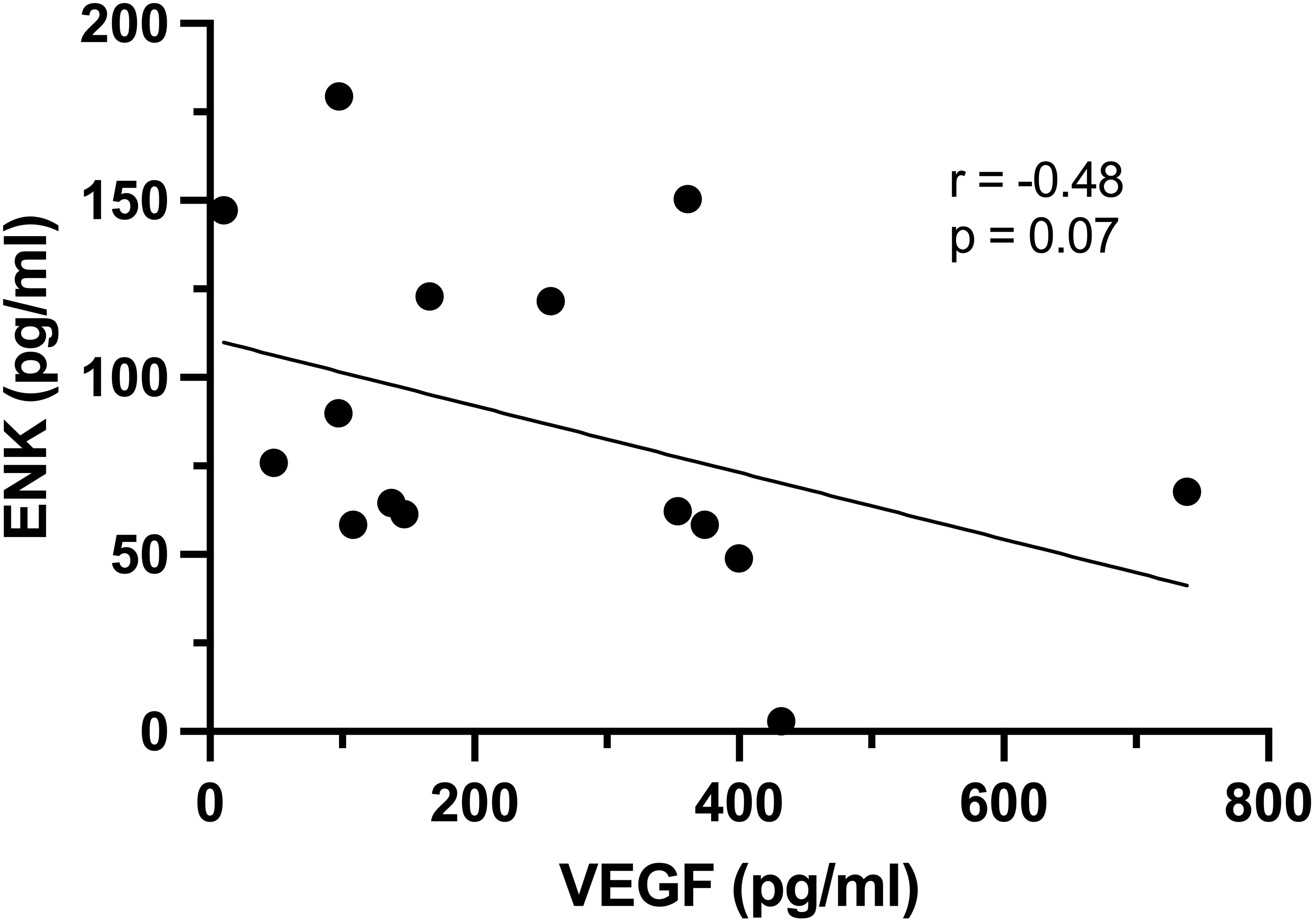
Correlation analysis of serum concentrations of ENK and VEGF in MS cohort subset. Data revealed a strong but statistically insignificant trending negative association between serum ENK and VEGF.

Correlation analyses in the MS cohort subset between (a) VEGF and age, (b) ENK and age, (c) VEGF and length of disease, and (d) ENK and length of disease. There were no statistically significant correlations, but ENK had a strong trending negative association with length of disease.
One MS cohort subset participant did not finish the PROMIS-FatigueMS, and one participant did not finish the PROMIS-Anxiety. VEGF and ENK were not correlated to anxiety, depression, or pain (data not shown). VEGF and ENK were not significantly correlated to fatigue; however, both biomarkers showed potential moderate positive and negative associations, respectively, with fatigue (VEGF: Spearman rho = 0.32, P = 0.19; ENK: Spearman rho = −0.36, P = 0.2) (Figure 4). Neither biomarker was correlated to walking time.
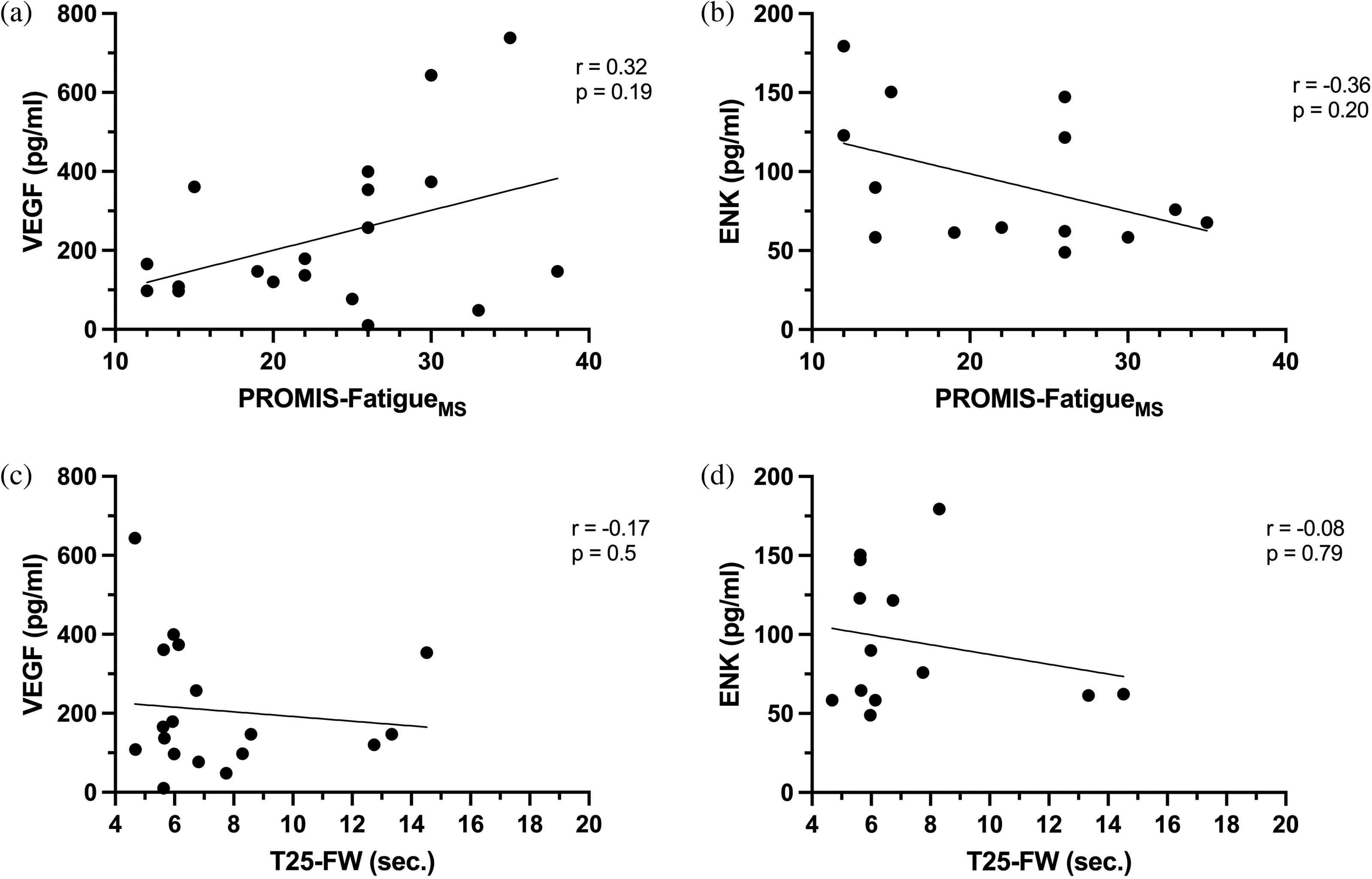
Correlation analyses in the MS cohort subset between (a) VEGF and fatigue, (b) ENK and fatigue, (c) VEGF and walking time, (d) ENK and walking time. There were no statistically significant correlations.
Discussion
Data revealed that PwMS experienced greater fatigue, but not anxiety, depression, or pain, compared to non-MS controls. PwMS experience greater anxiety and depression than the general population, 29 and the mean PROMIS-Anxiety and PROMIS-Depression scores for both cohorts in this study were greater than the US average. This study's Control cohort does not represent the general population, possibly due to the impacts of chronic pain on anxiety and depression. This poses a potential limitation; however, comparing PwMS to non-MS controls with similar pain, anxiety, and depression provides unique insights to discerning MS symptoms representing illness intrusiveness. The greater proportion and intensity of fatigue in the MS cohort supports fatigue as a salient MS feature.
The observed intercorrelation of fatigue, anxiety, depression, and pain in the MS cohort was expected, as these symptoms are comorbid in MS.30,31 MS symptoms are classified into clusters that aid in predicting the occurrence of related symptoms. “Emotional/cognitive” symptoms (depression, anxiety, cognitive impairments) and “physical” symptoms (pain, fatigue, sleep disorders) have been shown to co-occur and predict worsened quality of life in PwMS. 32 To further investigate symptom clusters, future studies would benefit from more robust cognitive and sleep health assessments.
Fatigue was the only variable that was positively correlated with walking time. These results highlight a need for the consideration of how fatigue might influence T25-FW times, since these times are used clinically to determine worsening physical disability in PwMS.
A trending strong negative association between serum VEGF and ENK was observed. This result was expected due to the proposed role of ENK as a modulator of angiogenesis and the role of VEGF as the most potent pro-angiogenic factor.14,15,33 VEGF promotes endothelial cell proliferation and shifts the cytokine profile of T cells to the pro-inflammatory Th1 phenotype. 34 Conversely, ENK reduces number of infiltrating CD4+ T cells in EAE mice. 18 These results warrant future studies on how ENK treatment might limit angiogenesis in MS and the EAE model.
In this study, serum VEGF was not correlated with age or LOD. These findings support previous studies that found VEGF to be more involved in disease onset and relapse instead of later disease progression. 11 We observed a potential positive association between fatigue and serum VEGF, possibly supporting the role of VEGF in inflammation and subsequent fatigue in MS. 11 The observed potential negative relationship between serum ENK and fatigue may support the role of ENK as a modulator of angiogenesis-associated inflammation and subsequent fatigue. However, given the tenuousness of these biomarker-fatigue relationships, it is possible that angiogenic signaling does not underlie fatigue in established MS. The pathophysiology of MS-related fatigue is complex, and it is likely that angiogenesis-induced CNS inflammation is only one of numerous factors.
Serum ENK had a trending negative association with LOD, but not age. These results differ from a previous study that found a positive correlation between ENK and age, but not LOD. 35 However, the previous study's sample consisted of only PwMS taking dimethyl fumarate, glatiramer acetate, low-dose naltrexone (LDN), or no DMT. The present study includes data from PwMS taking a variety of DMTs, including those from the previous study. More research is needed on how age and LOD influence the balance of different pro- and anti-angiogenic factors in MS.
Limitations of this study include potential self-report bias and subjective survey questions. Outside factors that might influence fatigue, anxiety, depression, and pain could not be controlled for, except for those in the exclusion criteria. As the correlation matrix analysis consists of multiple correlations, there is a risk of type 1 error in our findings; such an error could produce a “significant” or “trending” result from random data. However, given that comparisons produced very significant results (P < 0.01, P < 0.001), we are confident that we observed real relationships between these variables. Regardless, correlation data should be an indication of relationships to explore further. For example, the trending relationship we discovered between VEGF and ENK warrants future studies that quantify VEGF in PwMS taking LDN, a drug that functions to increase ENK production.
To the best of our knowledge, the present study is the first to examine the relationships between the pro-angiogenic factor VEGF and the anti-angiogenic factor ENK in MS. Our data corroborate reports that fatigue is a salient MS symptom. Furthermore, we propose that imbalanced angiogenic signaling may contribute to fatigue in PwMS, and that these relationships should be explored further in the context of MS symptom clusters.
Footnotes
Acknowledgments
The authors acknowledge Shannon Orehek, L.S.W. at the Penn State Hershey Neurology Clinic for assisting with data collection. The authors dedicate this paper to the memory of Chirag Patel, M.D., PhD, whose previous work as a doctoral student in the McLaughlin-Zagon laboratory was foundational to the conceptualization of the present study.
Declaration of conflicting interests
The authors declare the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: L.B.O., I.S.Z., and P.J.M. have nothing to declare. G.A.T. is a board member of the National Multiple Sclerosis Society but receives no financial support. J.J.Z. is an independent consultant and is part of a speaker's bureau for Sanofi, Bristol Myers Squibb, EMD Serono, and TG Therapeutics.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by a generous gift from the Paul K. and Anna E. Shockey Family Foundation and funds from the Penn State LDN Now Fund.
Ethical considerations
The study #9784 was reviewed and approved by the Pennsylvania State University Institutional Review Board.
Consent to participate
Written informed consent was obtained from participants after the study was explained and all questions were answered.
Data availability statement
The non-identifying participant data that were used in this study are available from the corresponding author upon reasonable request.
